BioImpacts. 8(3):211-221.
doi: 10.15171/bi.2018.24
Original Research
A cancer vaccine with dendritic cells differentiated with GM-CSF and IFNα and pulsed with a squaric acid treated cell lysate improves T cell priming and tumor growth control in a mouse model
Ananda Mookerjee 1, , $ &, Michele Graciotti 2, $, Lana E. Kandalaft 1, 2, *
Author information:
1Ovarian Cancer Research Center, University of Pennsylvania, Philadelphia, USA
2Ludwig Cancer Research Center, University of Lausanne, Lausanne, Switzerland; Department of Oncology, University Hospital of Lausanne, Lausanne, Switzerland
&Currently at: Cardiovascular Research Center, Icahn School of Medicine, Mount Sinai, New York, USA
$These authors equally contributed.
Abstract
Introduction:
Ovarian cancer is one of the most lethal gynecologic cancers. Relapses after remission are common, hence novel strategies are urgently needed. Our group has previously developed a vaccination approach based on dendritic cells pulsed with HOCl-oxidized tumor lysates. Here we investigate the improvement of this vaccine strategy using squaric acid treatment of cancer cells during tumor lysate preparation and by differentiating dendritic cells in the presence of GM-CSF and IFNα.
Methods:
Induction of cell death by squaric acid treatment was assessed with propidium iodide (PI) and Annexin V in ID8 tumor cells. High mobility group box 1 (HMGB1) immunogenic status was analyzed using a western blot-based method, as previously described. For immunological tests, ID8 cells expressing ovalbumin (ova-ID8) were treated with squaric acid before cell lysis. DCs prepared with the canonical GM-CSF and IL-4 differentiation cocktail or IFNα and GM-CSF were pulsed with tumor cell lysates and further matured in the presence of IFNγ and LPS (4-DCs and α-DCs respectively). DCs were then used in co-culture assays with ova-specific T cells and IFNγ and IL-4 secretion measured by ELISA. DC phenotypes were characterized by FACS. Finally, DCs were tested in an ovarian cancer mouse model measuring body weight and animal survival.
Results:
Squaric acid treatment of mouse ovarian cancer cells induced tumor cell death as well as preserve HMGB1, a crucial Damage-associated molecular pattern (DAMP) signal, in its active reduced form. Squaric acid treatment of ID8-ova cells increased IFNγ and decreased IL-4 production from ova-specific T cells in co-culture experiments, promoting a more immunogenic cytokine secretion pattern. DCs differentiated in the presence of IFNα induced a considerable decrease in IL-4 production compared to canonical 4-DCs, without affecting IFNγ release. DC phenotyping demonstrated a more mature and immunogenic phenotype for IFNα-differentiated DCs. Vaccination in tumor-bearing mice showed that IFNα-differentiated DCs pulsed with squaric acid-treated lysates were the most potent at delaying tumor growth, improving animal survival.
Conclusion:
We identified squaric acid as a novel immunogenic treatment of tumor cells for cancer vaccines particularly efficient in prolonging animal survival when used in combination with IFNα-differentiated DCs. These promising results support future efforts for the clinical translation of this approach.
Keywords: Cancer vaccine, Dendritic cells, IFNα, Ovarian cancer, Squaric acid
Copyright and License Information
© 2018 The Author(s)
This work is published by BioImpacts as an open access article distributed under the terms of the Creative Commons Attribution License (
http://creativecommons.org/licenses/by-nc/4.0/). Non-commercial uses of the work are permitted, provided the original work is properly cited.
Introduction
Ovarian cancer is one of the most lethal gynecologic cancers with a dismal prognosis.
1
Over 230 000 women are diagnosed with ovarian cancer worldwide each year, and about 140 000 women die from the disease.
2
Unfortunately, most ovarian cancer cases are diagnosed at a late stage and have a 5-year survival rate of only 39%.
2
Accumulating evidence shows that tumors can be spontaneously recognized and attacked by the immune system a fact that is associated with improved clinical outcomes.
3,4
Hence, it is postulated that immunotherapeutic vaccines could potentiate the anti-tumor immune response in ovarian cancer, especially when administered in the minimal residual disease setting.
Dendritic cells (DCs) are the most potent antigen-presenting cells in the immune system. Due to their unique ability to prime and stimulate both CD8+ and CD4+ T cells, DCs loaded with tumor antigens have been investigated in several clinical trials for their ability to induce therapeutic anti-tumor T cell responses.
5,6
Historically, DCs used for vaccination purposes have mostly been differentiated from monocytes in the presence of GM-CSF and IL-4. More recently, different cytokine cocktails have been developed that could generate alternative DCs with enhanced potency to induce tumor-specific immune responses.
7-12
In particular, tumor vaccines have been developed using either single antigens or whole tumor lysate. The use of the whole tumor approaches for vaccination purposes has several advantages over the use of defined tumor antigens. Tumor cells express a whole array of tumor-associated antigens (TAAs) that can be defined or undefined, shared or patient-specific (e.g. neoantigens arising from somatic mutations in individual tumors). Although it is currently unknown for most tumor types which TAAs mediate tumor regression, mounting evidence indicates that these neoantigens are important in the response to immune checkpoint blockade.
13-16
Another important feature of whole tumor approaches is the fact that they contain both MHC Class I and II restricted epitopes, allowing antigen presentation by DCs to both CD4+ and CD8+ T cells to generate stronger primary immune responses and to prevent the emergence of tumor escape variants.
3
The use of the whole tumor approaches also obviates the need for HLA selection of patients since it is not restricted to the specific epitope(s) contrary to vaccination with defined peptide antigen(s).
17
Interestingly, different methods have been employed to increase the immunogenic potential of whole tumor lysate as a source of antigens.
18
Our group previously developed a potent vaccination approach consisting of autologous DCs pulsed with hypochloric acid (HOCl)-oxidized autologous tumor lysate against ovarian cancer in humans.
19,20
However, HOCl is a rather unstable molecule characterized by a short half-life.
In the current study, we sought to alternate mild acid treatments to prepare tumor lysates for vaccine applications. We present a new immunogenic treatment of cancer antigens consisting in the use of squaric acid (SqA), a more stable molecule compared to HOCl and which is already clinically approved for the treatment of skin papillomas (so-called verrucae or warts) caused by an active skin infection by the human papilloma virus, a DNA virus. Given that HPV induced papillomas can be cured by the local application of SqA,
21
we theorized that the use of SqA for killing tumor cells could induce immunogenic cell death and thereby elicit a potent antitumor immune response. Furthermore, given that the successful therapeutic application of SqA occurs in the context of an active DNA viral infection, we theorized that the use of interferon-alpha (IFNα) to differentiate DCs could provide an important additional stimulus aiding the generation of an antitumor immune response. Results presented here both in vitro and in vivo in ovarian cancer mouse models suggest that both SqA treatment of cancer antigens and DC differentiation in the presence of IFNα are more immunogenic than a previously tested tumor lysate-pulsed DC vaccine and efficiently prolong animal survival supporting the future clinical translation of this approach.
Materials and Methods
Tumor cell lines and mice
The ID8 cell line was a generous gift from Dr. Paul F. Terranova, University of Kansas. The ID8-ova cell line was generated by stable transfection of ID8 cells using Express-in technology (Thermo Scientific, USA) with a construct of chicken ovalbumin (a kind gift from Dr. Y. Patterson, University of Pennsylvania) cloned in the pcDNA3.1-zeocin mammalian expression vector (Life Technologies, Carlsbad, USA). Tumor cells were cultured in complete DMEM (Cellgro, New York, USA) containing 10% heat-inactivated fetal bovine serum (FBS, Life Technologies) and antibiotics (Penstrep (Gibco, Gaithersburg, USA) at 10 U/mL culture medium and normocin, (Invitrogen, Waltham, USA) at 0.1 mg/mL culture medium). Cells were regularly tested for mycoplasma. Specific pathogen-free grade 6-8 week-old female C57BL/6, OT-I (C57BL/6-Tg(TcraTcrb)1100Mjb/J) and OT-II (B6.Cg-Tg(TcraTcrb)425Cbn/J) mice were purchased from the Jackson Laboratories, Sacramento, USA. Animals were maintained according to the institutional guidelines.
Preparation of tumor lysate
To prepare freeze-thaw tumor lysates, ID8-ova cells were harvested, washed and resuspended at 108 cells/ml in Mg+2/Ca+2-free DPBS containing the protease inhibitors complete mini tablet (Roche, Switzerland) and phosphatase inhibitors (PhosStop, Roche). Cells were then subjected to 6 cycles of freeze and thaw (snap freezing in liquid nitrogen and quick thawing at 37°C), followed by sonication (3 cycles of 5 W output for 15 seconds).
The HOCl-oxidized tumor lysate (ID8-ova-HOCl) was prepared as previously reported.
20
Briefly, ID8-ova cells were resuspended in 60 µM HOCl (Sigma-Aldrich, Germany) solution at a cell density of 108 cells/mL and incubated for 1 hour at 37°C and 5% CO2, with gentle agitation every 30 minutes. Subsequently, HOCl-treated cells were centrifuged at 600 x g for 6 minutes, the supernatant was discarded and the cell pellet washed twice with DPBS, resuspended in 1 mL DPBS and then subjected to 6 freeze-thaw cycles followed by sonication, as described above. For the preparation of SqA-treated tumor lysate (ID8-ova-Sq), ID8-ova cells were incubated with the indicated concentrations of SqA (Sigma-Aldrich, Germany) for 1 hour at 37°C and 5% CO2 after which cells were washed and subjected to the 6 freeze-thaw cycle followed by sonication process described above.
Detection of cell death
To detect cell death, cells were first washed with DPBS and subsequently with 1X binding buffer (Biolegend, USA). Cells were then resuspended in 1X binding buffer at a 2x106 cell/mL concentration. 5 µL of annexin V-FITC (Biolegend, USA) were added to 100 µL of cell suspension, and incubated for 15 minutes at room temperature (RT) in the dark. Cells were then washed with 1X binding buffer and resuspended in 250 µL of 1X binding buffer. Five µL of a propidium iodide (Sigma-Aldrich, Germany) solution (0.1 mg/mL) was added. After 5 minutes, 1 mL of pre-warmed DPBS containing 2% BSA was added and the cells were subsequently analyzed by FACS BD Canto-II (BD Biosciences, USA).
Determination of HMGB1 oxidation
The oxidation status of the high mobility group box 1 (HMGB1) protein in response to HOCl or SqA treatment of ID8 cells was determined using the methoxypolyethylene glycol-maleimide (MAL-PEG) conjugation method as described by Makmura et al
22
with minor modifications. Briefly, the ID8-Sq lysate was treated with 40 mM N-ethylmaleimide (Sigma-Aldrich, Germany) at RT with vigorous shaking to quench free sulfhydryl groups. After 2 hours incubation, the mixture was dialyzed against DPBS overnight at 4°C using a 10 kDa cut-off. DTT (Sigma-Aldrich, Germany) was then added at a 20 mM final concentration and samples incubated at RT for 30 minutes. Finally, MAL-PEG 20 000 (Sigma-Aldrich, Germany) was added at final 4 mM concentration and incubated for 1 hour at RT under constant shaking. β-mercaptoethanol-containing Laemmli buffer NuPAGE (Thermo Fisher, USA) was added to the samples which were then run on a 4%-12% gradient SDS-PAGE NuPAGE (Thermo Fisher, USA). Proteins were then transferred onto a PVDF membrane for 4 hours; the membrane was blocked with 2% BSA (Sigma-Aldrich, Germany) for 1 hour and sampled first with the anti-HMGB1 antibody (Abcam, UK) followed by goat anti-rabbit-HRP (Abcam, UK). Bands were revealed by the chemiluminescence Pierce ECL kit (Thermo Fisher, USA).
Detection of ova protein by western blot and ELISA
One hundred microliters of the indicated cell lysate were mixed with 900 µL of the carbonate-bicarbonate buffer, pH 9.5 and used to coat (100 µL/well) a Nunc maxisorp ELISA plate (Sigma-Aldrich, Germany). An ELISA assay was then performed using anti-ovalbumin-HRP polyclonal antibody (Abcam, UK) and TMB (BD Biosciences, USA) according to manufacturer instructions. For western blot, the indicated cell lysate samples were loaded on a 4%-10% gradient SDS-PAGE NuPAGE gel (Invitrogen, USA). Proteins were then transferred onto a PVDF membrane for 4 hours; the membrane was blocked with 5% fat-free milk (Sigma-Aldrich, Germany) for 1 hour and sampled with anti-ova-HRP antibody (Abcam, UK). Bands were revealed by the chemiluminescence Pierce ECL kit (Thermo Fisher, USA).
Mouse bone marrow-derived DC preparation
DCs were generated from murine bone marrow cells, as described by Garrigan et al
23
with minor modifications. Briefly, bone marrow was flushed from the long bones of C57BL/6 mice. A single cell suspension was cultured in RPMI 1640 (CellGro, USA) supplemented with 10% heat-inactivated FBS (Life Technologies, USA), 2 mM L-glutamine (Gibco, USA), 100 U/mL penicillin (Gibco, USA), 100 µg/mL streptomycin (Gibco, USA), 10 mM HEPES pH 7.4 (Gibco, USA), 0.5 mM sodium pyruvate (Cellgro, USA), 0.5% MEM non-essential amino acids (Cellgro, USA), 0.1 mg/mL Normocin (InVivogen, USA), along with 20 ng/mL GM-CSF (Peprotech) and 10 ng/mL IL-4 (Peprotech, USA) for 4-DCs or 20 ng/mL GM-CSF and 250 U/mL IFNα (AbD Serotec, USA) for α-DCs. On day 2, the medium was replaced with fresh medium containing the respective cytokines. On day 4, 100 µL of the indicated tumor lysate preparations were added to 10 mL culture (~1 tumor cell equivalent per DC) and incubated overnight at 37°C and 5% CO2. Unpulsed DCs were also cultured as negative controls. On day 5, the medium was removed and fresh medium containing 100 ng/mL LPS (Sigma-Aldrich, Germany) and 100 ng/mL IFNγ (Peprotech, USA) was added. DCs were then cultured overnight at 37°C and 5% CO2.
In vitro stimulation of T cells and cytokine analysis
T cells from spleens of OT-I and OT-II animals were purified by negative selection with Dynabeads (Invitrogen, USA) according to manufacturer protocol. 1x105 T cells were then plated in 96-well flat-bottom plates with 1x104 DCs and cultured for 24 hours in a culture volume of 200 µL. Cell-free culture supernatants were then harvested to measure IFNγ and IL-4 cytokine levels by ELISA using OptEIA ELISA sets (BD Biosciences, USA).
Dendritic cell phenotyping
Mature lysate-pulsed DCs were harvested and blocked with DPBS containing 2% FBS and anti-mouse CD16/CD32 monoclonal antibody Fc Block (BD Biosciences, USA). Cells were subsequently stained for 15 minutes at 4°C with the following antibodies (Biolegend, USA): CD14-APC, F4/80-BV421, MHC-I (H-2Kb/H-2Db-PE), MHC-II (I-A/I-E-AF488), CD86-PE/Cy7, CD83-FITC or with the appropriate isotype-matched controls (Biolegend, USA). After staining, the cells were washed and fixed with 2% w/v paraformaldehyde. For intracellular cytokine staining, cells were treated with brefeldin A (Sigma-Aldrich, Germany) at 10 µg/mL for 4 hours at 37°C. Cells were then blocked, stained for appropriate surface markers as described above, washed twice and then fixed and permeabilized using BD fixation/permeabilization kit (BD Biosciences, USA). Cells were subsequently incubated for 2 hours in blocking solution (1X BD Perm-wash containing 1 µg/mL Fc Block), centrifuged and then stained for cytokines with the following antibodies: IFNγ-AF488 (eBioscience, USA), IL-12p70-biotin (Invitrogen, USA) and streptavidin-APC (Invitrogen, USA), TNFα-PE/Cy7 (eBioscience, USA), TFGβ1-LAP-PerCP/Cy5.5 (Biolegend, USA), IL-10-PE (eBioscience, USA); or with appropriate isotypes controls in BD perm-wash. Cells were then washed twice and resuspended in DPBS containing 2% FBS. Cells were analyzed by FACS BD-Canto II (BD Biosciences, USA).
Mice tumor vaccination
C57BL/6 mice were inoculated intraperitoneally (i.p.) with 1 mL of DPBS containing 1x106 ID8-ova cells irradiated 24 hours previously with a sublethal irradiation dose (2.5 Gy). Five days following tumor inoculation, animals were injected intravenously (i.v.) with OT-I CD8+ T cells (2.5 × 105cells/animal) and OT-II CD4+ T cells (5×105 cells/animal). Vaccination with DCs (1×106 DCs/shot/animal) was performed i.p. on days 7, 14 and 21 following adoptive T cell transfer. Animals were monitored regularly for tumor growth. Mice attaining 30 g body weight were sacrificed (~40% increase in body weight over age/sex-matched normal mice).
Statistical analysis
SAS software (version 9.3) and StatXact Procs 9 for SAS were used for statistical analysis. In particular, a two-tailed Student’s t-test was used to compare means of continuous measurements between two groups. The analysis of variance (ANOVA) was used to compare means among more than two groups; differences were considered statistically significant when P<0.05. For animal survival, statistical analysis was performed with GraphPad Prism software using the Log-rank (Mantel-Cox) test.
Results
Squaric acid induces immunogenic necroptosis in tumor cells
In order to assess the role of SqA treatment in cancer vaccination strategies, we first set out to investigate the ability of SqA treatment to induce necrotic cell death or apoptosis in cancer cells. To this aim, we incubated ID8 tumor cells with increasing concentrations of SqA for 1 hour at 37°C. We observed a clear dose-dependent effect on the induction of cell death by increasing SqA concentrations, with 100% cell death at 0.3% SqA (Fig. 1A). To determine the type of cell death induced, we measured PI and annexin V staining in treated cells at 1 hour. As expected, treatment of ID8 cells with SqA resulted in dose-dependent necrosis within 1 hour, with nearly complete necrotic cell death observed at 0.06% SqA. Interestingly, a higher dose of 0.3% SqA resulted in ~100% cell necroptosis, with nearly 100% cells expressing surface annexin V (Fig. 1B).
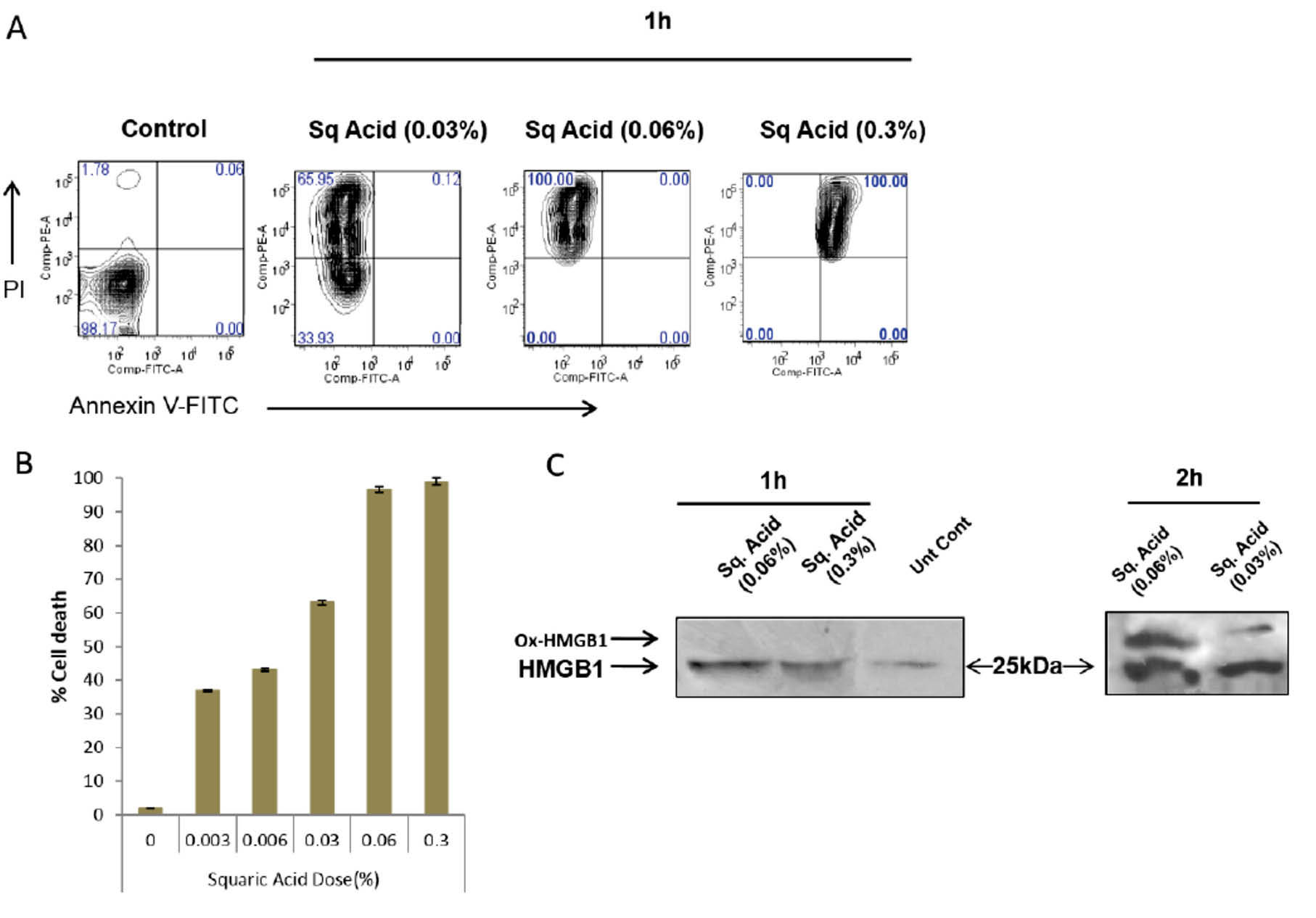
Fig. 1.
Squaric acid induces immunogenic cell death in ID8 cells. (A) ID8 cells were treated with the indicated stimuli for 1 h at 37°C. Cells were then washed and stained for Annexin V and PI. Annexin positive cells were classed as apoptotic while necrotic cells were only PI positive and healthy cells were double negative. Data are representative of 3 independent experiments. (B) ID8 cells were treated with the indicated doses of squaric acid for 1 h and cell death was measured by PI staining. (C) ID8 cells were treated with the indicated stimuli for 1 h or 2 h at 37 °C. The HMGB1 oxidation status in the cell lysate was determined by western blot using the method developed by Makmura et al.
22
Data are representative of 3 independent experiments.
.
Squaric acid induces immunogenic cell death in ID8 cells. (A) ID8 cells were treated with the indicated stimuli for 1 h at 37°C. Cells were then washed and stained for Annexin V and PI. Annexin positive cells were classed as apoptotic while necrotic cells were only PI positive and healthy cells were double negative. Data are representative of 3 independent experiments. (B) ID8 cells were treated with the indicated doses of squaric acid for 1 h and cell death was measured by PI staining. (C) ID8 cells were treated with the indicated stimuli for 1 h or 2 h at 37 °C. The HMGB1 oxidation status in the cell lysate was determined by western blot using the method developed by Makmura et al.
22
Data are representative of 3 independent experiments.
Based on the observation that SqA is able to kill tumor cells, we next tested whether SqA induces immunogenic cell death (ICD), a special type of cell death that through the release of damage-associated molecular patterns (DAMPs) can induce an effective antitumor immune response.
24
One of the markers of ICD is the release of high-mobility group protein B1 (HMGB1).
25
However, a recent study showed that HMGB1 is immunogenic only in its reduced form, but not in its oxidized state.
26
In particular, the study showed that oxidation of Cysteine106 is necessary and sufficient to inactivate such immunostimulatory activity.
26
Based on this, we decided to quantify the levels of HMGB1 cysteine oxidation using a previously established protocol based on SDS-PAGE mobility.
22
In this assay, cysteine residues present in the protein sample in their oxidized sulfenic acid forms are covalently tagged with a ~5 kDa tag, causing a shift towards higher molecular weights in the SDS-PAGE gel which can then be revealed through western blot analysis. We, therefore, used this assay to measure HGMB1 oxidation levels in the ID8 cells treated with increasing concentration of SqA. Interestingly, we observed that 1h treatment with 0.06% or 0.3% SqA did not result in detectable levels of HMGB1 oxidation (no band shift towards higher molecular weights; Fig. 1C). However, prolonged SqA treatment for 2 hours induced detectable levels of cysteine oxidation to sulfenic acid of HMGB1 at both 0.06% and 0.3% tested concentrations (Fig. 1C). Thus, cell treatment for 1 hour with 0.3% or 0.06% SqA efficiently induced tumor cell death while preserving the reduced state of HMGB1. Due to the reported immunogenicity of HMGB1 in its reduced form
26
we, therefore, restricted SqA treatments to 1 hour in the following experiments.
DCs pulsed with SqA-killed tumor cells stimulate antigen-specific T cells
To test the immunogenicity of SqA-killed tumor lysate, we used ovalbumin (ova) as a model antigen. To this aim, we generated an ova expressing ID8 cell line (ID8-ova) by stably transfecting ID8 cells with the chicken ovalbumin gene. Importantly, preliminary western blot and ELISA analyses confirmed that ova was expressed to detectable levels and stable throughout multiple cell passages in this cell line, validating the use of ID8-ova in subsequent immunological tests following ova antigen-specific responses (Fig. S1A and B, Supplementary Data). Next, ID8-ova cells were killed with 0.06% SqA for 1hr (ID8-ova-Sq) followed by 6 freeze-thaw cycles. As a control lysate, ID8-ova cells were killed with 60 µM HOCl followed by 6 freeze-thaw cycles (ID8-ova-HOCl). The so-generated lysates were then used to pulse DCs differentiated from bone marrow cells with GM-CSF and IL-4 (4-DCs), followed by DC maturation with LPS and IFNγ for ~18 hours. We compared the above DCs pulsed with tumor lysate to DCs differentiated from bone marrow cells with GM-CSF and IFNα instead of IL-4 (α-DCs), which were pulsed with ID8-ova-HOCl or ID8-ova-Sq tumor lysate and matured with LPS and IFNγ for ~18 hours. Antigen-pulsed mature DCs (4-DCs and α-DCs) were incubated with ova-specific CD8+ T cells (OT-I) and CD4+ T cells (OT-II) in co-culture assays for 24 hours, and IFNγ and IL-4 secreted levels were measured in the supernatant.
DCs pulsed with tumor lysate prepared with SqA or HOCl elicited significantly higher IFNγ production in both OT-I and OT-II co-cultures compared to DCs pulsed with freeze-thaw lysate, extending our previous observation for HOCl
20
also to the newly tested SqA treatment (Fig. 2A). Furthermore, relative to DCs pulsed with HOCL lysate, DCs pulsed with SqA lysate elicited significantly higher IFNγ responses in OT-1 cells, and among them, α-DCs appeared significantly superior to 4-DCs. The two lysates were similar in terms of OT-II IFNγ responses, but again α-DCs elicited significantly higher responses than 4-DCs. Thus, α-DCs appeared superior to 4-DCs when pulsed with oxidized tumor lysate, and α-DCs pulsed with SqA lysate elicited stronger class I responses.

Fig. 2.
Squaric acid antigen treatment and differentiation of DCs in the presence of IFNα lead to favorable cytokine secretion by antigen-specific T cells in DC-T cells co-cultures. (A, B) DCs were pulsed with the indicated tumor antigens overnight and subsequently matured with LPS and IFNγ overnight. DCs were then co-cultured with OT-I or OT-II cells at a 1:10 ratio at 37°C for 24 h; levels of IFNγ (A) and IL-4 (B) were measured by ELISA. Results are represented as mean ± SD of at least 3 independent experiments. Significant differences were assessed with unpaired Student’s t test and indicated with asterisks: * P < 0.05; ** P < 0.01.
.
Squaric acid antigen treatment and differentiation of DCs in the presence of IFNα lead to favorable cytokine secretion by antigen-specific T cells in DC-T cells co-cultures. (A, B) DCs were pulsed with the indicated tumor antigens overnight and subsequently matured with LPS and IFNγ overnight. DCs were then co-cultured with OT-I or OT-II cells at a 1:10 ratio at 37°C for 24 h; levels of IFNγ (A) and IL-4 (B) were measured by ELISA. Results are represented as mean ± SD of at least 3 independent experiments. Significant differences were assessed with unpaired Student’s t test and indicated with asterisks: * P < 0.05; ** P < 0.01.
SqA lysate-pulsed DCs elicited significantly lower IL-4 production in OT-I cells relative to HOCL lysate-pulsed DCs (Fig. 2B). Furthermore, α-DCs elicited significantly less IL-4 in all conditions relative to 4-DCs, and specifically, when α-DCs were pulsed with SqA lysate we detected no IL-4 in output OT-I cells, indicating a very strong type I polarization.
Hence, results from these in vitro experiments not only confirmed previous evidence for improved immune responses elicited by HOCl-oxidized antigens but highlighted also squaric acid as an alternative and even more immunogenic treatment of antigens worthy of further study and characterization. Concerning the different stimuli tested for DC preparation, our data importantly demonstrated the superiority of α-DCs compared to the “canonical” 4-DCs, at least in terms of favorable in vitro cytokine production.
α-DCs pulsed with SqA lysate are more immunogenic
Based on the above results we investigated further the effects of the different stimuli on DC maturation. To this aim, we measured levels of common markers for DC maturation such as CD14, F4/80, MHC-II, CD86 and CD83 by FACS. Interestingly, when comparing 4-DCs to α-DCs pulsed with the same antigen source (or kept unpulsed), a general trend was observed with a marked decrease in CD14 and F4/80 expression and increase in MHC-I, CD83 and CD86 in α-DCs, all indicative of a more mature and immunogenic DC phenotype (Fig. 3A). On the other hand, we also observed a lower expression of MHC-II molecules in α DCs compared to 4-DCs, which may lead to impaired CD4+-dependent immune responses (Fig. 3A). Instead, no significant differences were recorded between α-DCs pulsed with tumor lysates prepared with SqA or HOCl compared to α-DCs pulsed with freeze-thaw lysate. The same was true also for 4-DCs, except for a significant increase in MHC-I expression levels in 4-DCs pulsed with SqA or HOCl lysate compared to freeze-thaw lysate (P ≤ 0.05, Fig. 3A). Thus, these results confirm the previously published observation that HOCl antigen treatment do not contribute to DC maturation
27
(notably no effect on CD83 and MHC-II levels) and that a second maturation stimuli (such as LPS and IFNγ in our case) is necessary to reach a full maturation status, extending this observation also to SqA. On the other hand, these results importantly demonstrate that HOCl or SqA antigen treatments do not minimally interfere with DC maturation but actually improve their immunogenic phenotype in the few instances indicated.
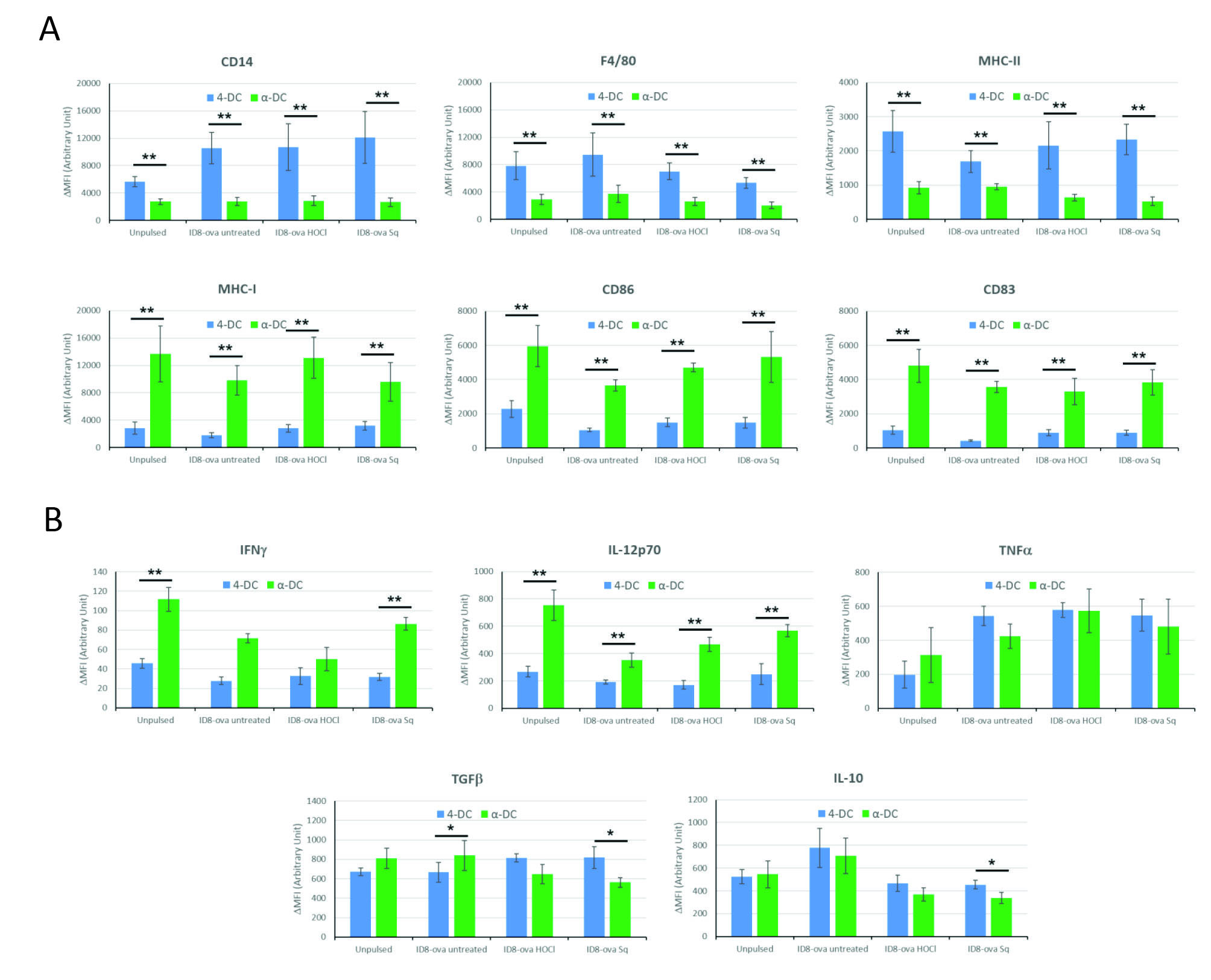

Fig. 3.
Effects of different antigens and differentiation stimuli used to prepare DCs on their mature phenotype and intracellular cytokine profile. (A) DCs were prepared as indicated in Fig. 2. The expression levels of indicated markers were then assessed by antibody staining followed by FACS analysis. The net mean fluorescence intensity (ΔMFI = Raw MFI-MFI of Isotype) for each marker is reported in the graph. Data are representative of 3 independent experiments. Student’s t test results comparing α-DCs to 4-DCs pulsed with the same antigen are indicated with asterisks: ** P < 0.01. (B) DCs were incubated with brefeldin A for 4 h at 37°C. Cells were then stained for CD14, F4/80, MHC-I, MHC-II, CD86 and CD83; subsequently permeabilized and stained for IFNγ, IL-12p70, TNFα, TGFβ and IL-10. Cells were analyzed by flow cytometry and results reported as net MFI for each cytokine. Data are representative of 3 independent experiments. Student’s t test results comparing α-DCs to 4-DCs pulsed with the same antigen are indicated with asterisks: * P < 0.05, ** P < 0.01.
.
Effects of different antigens and differentiation stimuli used to prepare DCs on their mature phenotype and intracellular cytokine profile. (A) DCs were prepared as indicated in Fig. 2. The expression levels of indicated markers were then assessed by antibody staining followed by FACS analysis. The net mean fluorescence intensity (ΔMFI = Raw MFI-MFI of Isotype) for each marker is reported in the graph. Data are representative of 3 independent experiments. Student’s t test results comparing α-DCs to 4-DCs pulsed with the same antigen are indicated with asterisks: ** P < 0.01. (B) DCs were incubated with brefeldin A for 4 h at 37°C. Cells were then stained for CD14, F4/80, MHC-I, MHC-II, CD86 and CD83; subsequently permeabilized and stained for IFNγ, IL-12p70, TNFα, TGFβ and IL-10. Cells were analyzed by flow cytometry and results reported as net MFI for each cytokine. Data are representative of 3 independent experiments. Student’s t test results comparing α-DCs to 4-DCs pulsed with the same antigen are indicated with asterisks: * P < 0.05, ** P < 0.01.
To further assess the immunogenicity of the different DC preparations in the study, we then also measured the in vitro production of the immunostimulatory cytokines IFNγ, IL-12p70 and TNFα and immunoregulatory cytokines TGFβ and IL-10 by antibody intracellular staining (Fig. 3B). Importantly, results showed that α-DCs always produced significantly higher IL-12p70 than 4-DCs (comparing same antigen source), and also more IFN-γ, less IL-10 and less TGFβ in the case of SqA lysate; suggesting once again a more immunogenic phenotype.
In terms of comparisons between different antigen sources, the analyses here presented showed that both 4-DCs and α-DCs produced significantly less IL-10 when pulsed with HOCl or SqA lysates, compared to their freeze-thaw pulsed counterparts (P < 0.05, Fig. 3B). A significant increase in IL-12p70 production was also observed in α-DCs stimulated with HOCl or SqA lysates, compared to freeze-thaw lysate (P < 0.05), while the same was not true in the case of 4-DCs (no significant differences, Fig. 3B). On the other hand, no significant differences were recorded instead between the HOCl and the SqA lysate conditions, suggesting a similar effect at least on the expression of the tested cytokines by 4-DCs or α-DCs.
Taken altogether, the results here presented demonstrated therefore that treating antigens with HOCl or SqA improves the immune fitness of DC vaccine formulations without affecting patterns of expression of important immunostimulatory DC surface markers. Moreover, these analyses also suggested that DCs differentiated in the presence of IFNα (α-DCs) possess advantageous DC phenotypes compared to more “classical” 4-DCs, supporting, therefore, their potential use in in vivo cancer vaccine approaches. As in the previous case of DC-T cell co-cultures (Fig. 2A, B) the combination of ID8-ova-Sq as antigen source with α-DCs overall showed the best immune fitness profile for the tested biomarkers.
Vaccination with α-DCs pulsed with SqA lysate prolongs animal survival in the ID-8 tumor model
We tested whether the considered DC types pulsed with the different tumor lysate preparations were able to control ID8-ova tumor growth and increase animal survival in mice. To this aim, we used ID8-ova cells to establish intraperitoneal orthotopic tumors in sublethally irradiated wild-type mice (day 0) and performed an intravenous adoptive transfer of OT-I CD8+ and OT-II CD4+ cells on day 5, prior to vaccination. Animals were then vaccinated 3 times with the indicated DC vaccine inoculated i.p. every 7 days from initial tumor inoculation (Fig. 4A), and were followed for up to 120 days post-tumor inoculation. Tumor evolution was assessed by body weight, which in this model captures ascites fluid accumulation, and by overall survival.
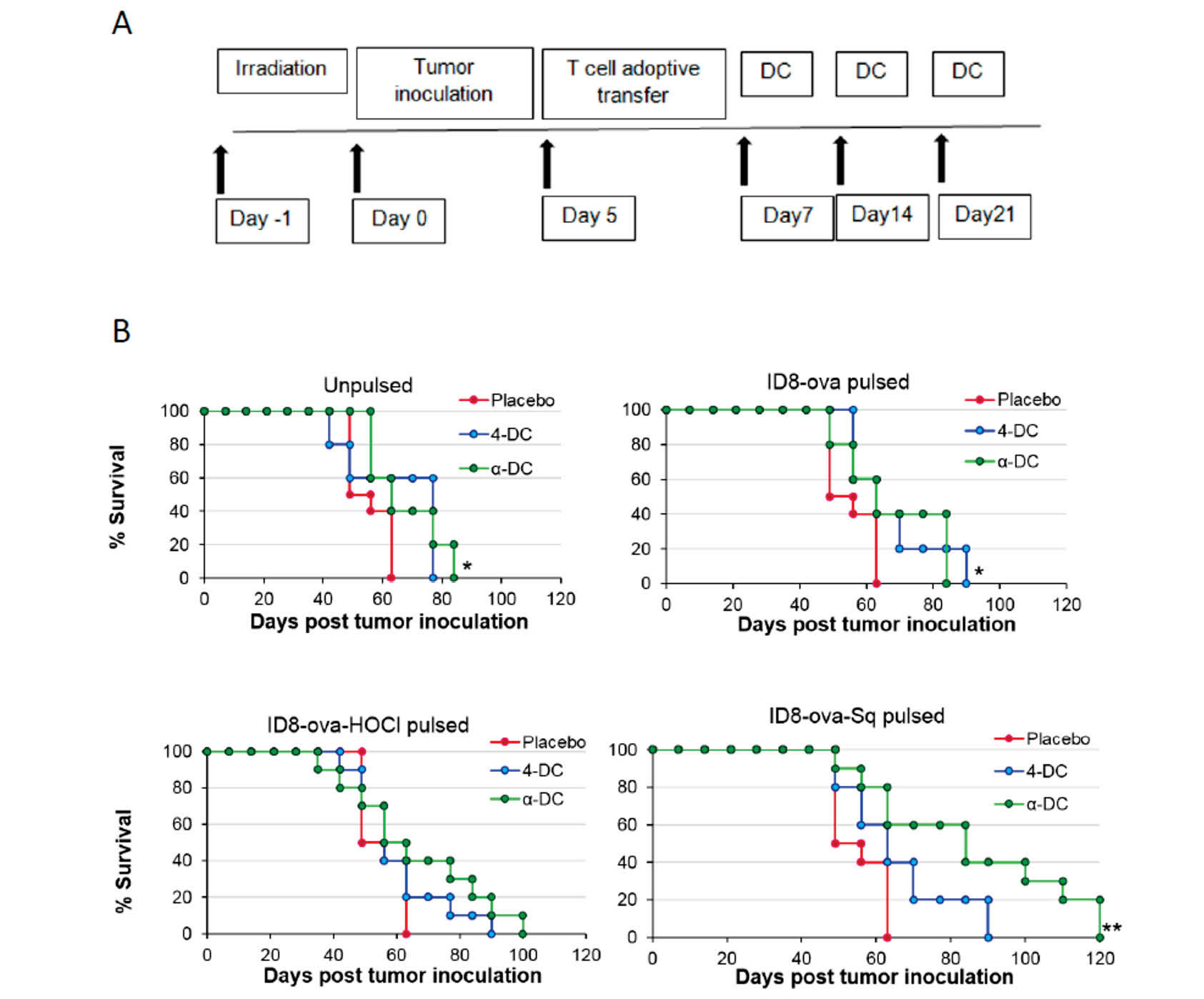
Fig. 4.
Squaric acid antigen treatment and differentiation of DCs in the presence of IFNα prolongs animal survival in a vaccination mouse model of ovarian cancer. (A) Vaccination scheme: mice were inoculated with irradiated ID8-ova cells at day 0, followed by adoptive transfer of OT-I and OT-II cells at day 5. Subsequently mice were vaccinated with the indicated DCs on days 7, 14 and 21 following adoptive T cell transfer (n= 20 for each group). (B) Animal survival was evaluated until body weight increased up to 30g and plotted in Kaplan–Meier cumulative survival plots. Student’s t test results (compared to placebo) are indicated with asterisks: * P < 0.05, ** P < 0.01.
.
Squaric acid antigen treatment and differentiation of DCs in the presence of IFNα prolongs animal survival in a vaccination mouse model of ovarian cancer. (A) Vaccination scheme: mice were inoculated with irradiated ID8-ova cells at day 0, followed by adoptive transfer of OT-I and OT-II cells at day 5. Subsequently mice were vaccinated with the indicated DCs on days 7, 14 and 21 following adoptive T cell transfer (n= 20 for each group). (B) Animal survival was evaluated until body weight increased up to 30g and plotted in Kaplan–Meier cumulative survival plots. Student’s t test results (compared to placebo) are indicated with asterisks: * P < 0.05, ** P < 0.01.
Body weight assessment showed that vaccination with ID8-ova-Sq pulsed α-DCs resulted in a slower increase in body weight (slower accumulation of ascites) in approximately 40% of the vaccinated animals compared to the other groups, while no significant differences were observed among the rest of the groups (Fig. S2, Supplementary Data). Vaccination with either 4-DCs or α-DCs pulsed with ID8-ova-HOCl lysate determined a median survival increase of ~16-20 days and ~23-35 days compared to control untreated mice (Fig. 4B), indicating a beneficial effect of HOCl oxidation of antigens on overall animal survival, as previously reported.
20
Interestingly, in the case of DCs pulsed with ID8-ova-HOCl a slight survival improvement of ~4 days was observed with α-DCs over 4-DCs, although not statistically significant. For DCs pulsed with ID8-ova-Sq a median survival improvement of ~10 days over untreated controls was observed in animals vaccinated with 4-DCs, close to statistical significance (P = 0.059). On the other hand, a larger median survival improvement of ~31 days over untreated controls was observed in the case of α-DCs pulsed with ID8-ova-Sq. Importantly, among the different vaccine formulations tested, this latter condition was the one characterized by the longest median animal survival with an improvement close to significance compared to either 4-DCs pulsed with the same antigen source (P = 0.056) or α-DCs pulsed with ID8-ova-HOCl (P = 0.055). Taken together, in the ID8 model IFNα-differentiated DCs pulsed with SqA treated cell lysates produced a significant improvement in overall survival.
Discussion
Ester derivatives of SqA, like squaric acid dibutylester (SADBE) have been clinically used to treat human papillomas for a long time and were also previously shown to possess immunomodulatory properties.
28
SqA is a known inhibitor of glyoxalase I,
29
which is upregulated in aggressive cancers including ovarian cancer
30
and it is able to convert methylglyoxal, a cytotoxic byproduct of glycolysis, into D-Lactate.
29
From a chemical point of view, SqA is a highly reactive molecule; thanks to both its ring strain and the presence of an α, β-unsaturated carbonyl moiety it can participate in a plethora of reactions, usually favored by ring opening and relaxation. Like HOCl, derivatives of SqA can also promote radical-mediated redox reactions,
31
but contrary to the former, SqA can also act in addition as a direct cross-linking agent between 2 amine groups of 2 distinct proteins or peptides.
32
Interestingly, this property of SqA has been recently used to increase the potency of a MUC1 vaccine by covalently cross-linking the MUC1 epitope to three different T-helper-cell epitopes.
33
Our group
19,20,34,35
and others
27,36-38
have previously reported on the increased immunogenicity of cancer antigen(s) treated with HOCl; however HOCl is a rather unstable molecule characterized by a short half-life. Given the higher stability and longer shelf life of squaric acid, its current use in the clinic (hence its availability in GMP grade and clinically approval) and its high reactivity and additional cross-linking properties, we therefore decided to investigate in this work the immunogenic properties of SqA treatment of cancer antigens in the context of DC cancer vaccination. We also theorized that the addition of IFNα during the differentiation of DCs, could replicate some of the signals associated with the HPV infection and perhaps enhance the effect of SqA.
Our results indicated that SqA is able to induce tumor cell death in cancer cells. Importantly, we were able to show that this treatment does not impact on the oxidation status of HMGB1, preserving this important DAMP signal in its immunogenic reduced form,
26
at least when limited to 1 hour. When tested in vitro, DCs pulsed with tumor cells treated with SqA (ID8-ova-Sq) were more potent inducers of a Th1 type response than untreated cells (ID8-ova), as they led to both higher IFNγ production by antigen-specific CD8+ and CD4+ T-cells. In addition, α-DCs pulsed with ID8-ova-Sq lysate outperformed α-DCs pulsed with ID8-ova-HOCl lysate in terms of CD8 response.
Alongside testing new cell antigen treatments to increase their immunogenicity, we also implemented a different cytokine cocktail used to differentiate DCs from their bone marrow precursors in an effort to obtain a more powerful cell preparation for cancer vaccine therapies. Indeed, we found that “canonical” mouse 4-DCs induced a sustained IL-4 production in DC-T cell co-cultures, at least in vitro, which can be detrimental in the context of cancer vaccination.
39
In addition to this, previous evidence suggested that DCs pulsed with whole tumor cell lysates have lower IL-12p70 production, a feature associated with impaired p38 MAPK signaling.
40
We, therefore, decided to introduce IFNα in substitution of IL-4 in the system, in an attempt to re-activate this pathway.
41,42
An additional reason to introduce IFNα was to mimic signals from a viral infection, which in the case of papilloma may synergize with SqA to produce a curative immune response. In doing so we observed that, compared to conventional 4-DCs, DCs differentiated in the presence of IFN-α (α-DCs) generally expressed higher levels of MHC-I and CD86 and lower levels of F4/80, indicative of a more mature phenotype and a potential better interaction with CD28
43,44
leading to subsequent CD8+ T-cell increased activation. Moreover, α-DCs stimulated higher production of IFNγ and lower IL-4 production by both CD8+ and CD4+ T-cells in an antigen-specific manner compared to 4-DCs pulsed with the same antigen source, at least in vitro.
Importantly, the modality of lysate preparation did not affect these maturation markers, indicating that exposure of tumor cells to HOCl and even more so to SqA enhances DC performance presumably through enhanced cross-presentation of tumor antigen. In agreement with these observations, animals vaccinated with α-DCs pulsed with ID8-ova-Sq exhibited significantly slower ascites accumulation and the best overall survival among tested conditions.
So far, several methods have been implemented to improve the immunogenicity of whole tumor antigen(s) for DC vaccination, from UV irradiation
20,45
to heat shock
46,47
or high hydrostatic pressure
48
(see
18
for a review). It is generally believed that these different treatments improve immunogenic potential by increasing exposure/release of DAMPs as a consequence of cell stress.
18
Similarly, SqA treatment may induce the expression of proteins with adjuvant properties or neutralize proteins with the reverse function in tumor cells, hence having a positive impact on downstream immune responses. An alternative or complementary explanation is that squaric acid may react with tumor proteins introducing chemical changes (e.g. alteration in the redox status, cross-linking, aggregation etc) that could potentially favor either uptake by antigen presenting cells, proteolysis, antigen processing, cross-presentation or a combination of the above explaining in this way the increased immunogenicity experimentally observed. In particular, this possibility has already been shown in a few proof-of-principle studies showing that administration of a conjugate vaccine obtained by SqA-mediated chemical cross-linking of different peptide epitopes
33
(or a protein carrier with a bacterial carbohydrate)
49,50
increased induced antigen-specific immune responses. A higher stability towards metabolic degradation has been proposed to explain such results.
33
In any case, chemical cross-linking may well just represent only a partial explanation for the overall improved immunogenicity and further tests in this direction (e.g. mass spectrometry-based approaches) will help to prove these hypotheses elucidating the exact mechanisms of action of squaric acid.
Nonetheless, the observations here presented, together with the increased stability and clinically approved use of SqA above mentioned, support further clinical testing of SqA treatment of tumor antigen in future DC vaccine trials.
Conclusion
Improving current dendritic cancer cell therapies will potentially provide for new and more potent personalized approaches to treat cancer. Our study illustrates that squaric acid treatment of tumor cell antigens greatly improves their immunogenicity stimulating a more effective immune response. Further to this, we also showed that when coupled with DCs generated with GM-CSF and IFNα instead of the canonical GM-CSF and IL-4 differentiation cocktail, this vaccine was particularly powerful in prolonging animal survival in an ovarian cancer mouse model. These results support future efforts aimed at translating this approach into the clinic. Such efforts could in fact importantly contribute to improve DC-based immunotherapies which showed so far only moderate success and thus help to exploit their full recognized (but still covered) potential.
51,52
Ethical approval
None to be declared.
Competing interests
Authors declare no conflict of interests.
Supplementary Materials
Supplementary data contains Figs. S1-S2.
(pdf)
Research Highlights
What is current knowledge?
simple
-
√ Dendritic cells (DCs) process and present antigens on their
surface to stimulate T cell responses against pathogens.
-
√ DC vaccines have been used in the clinic for cancer therapy,
although with only moderate success so far.
What is new here?
simple
-
√ Treating tumor cell antigens with squaric acid increases
their immunogenicity.
-
√ DCs differentiated with IFNα from their precursors present
a more immunogenic phenotype.
-
√ Both treatments increased animal survival in a tumor
mouse model supporting the clinical translation of this
promising novel DC vaccine formulation.
References
- Mills K, Fuh K. Recent Advances in Understanding, Diagnosing, and Treating Ovarian Cancer. F1000Research 2017; 6:84. doi: 10.12688/f1000research.9977.1 [Crossref] [ Google Scholar]
- Ferlay J, Shin H-R, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010; 127:2893-917. doi: 10.1002/ijc.25516 [Crossref] [ Google Scholar]
- Chiang CL-L, Benencia F, Coukos G. Whole tumor antigen vaccines. Semin Immunol 2010; 22:132-43. doi: 10.1016/j.smim.2010.02.004 [Crossref] [ Google Scholar]
- Adams SF, Levine DA, Cadungog MG, Hammond R, Facciabene A, Olvera N. Intraepithelial T cells and tumor proliferation. Cancer 2009; 115:2891-902. doi: 10.1002/cncr.24317 [Crossref] [ Google Scholar]
- Constantino J, Gomes C, Falcão A, Neves BM, Cruz MT. Dendritic cell-based immunotherapy: a basic review and recent advances. Immunol Res 2017; 65:798-810. doi: 10.1007/s12026-017-8931-1 [Crossref] [ Google Scholar]
- Garg AD, Vara Perez M, Schaaf M, Agostinis P, Zitvogel L, Kroemer G. Trial watch: Dendritic cell-based anticancer immunotherapy. Oncoimmunology 2017; 6:e1328341. doi: 10.1080/2162402X.2017.1328341 [Crossref] [ Google Scholar]
- Anguille S, Lion E, Tel J, de Vries IJM, Couderé K, Fromm PD. Interleukin-15-Induced CD56+ Myeloid Dendritic Cells Combine Potent Tumor Antigen Presentation with Direct Tumoricidal Potential. PLoS One 2012; 7:e51851. doi: 10.1371/journal.pone.0051851 [Crossref] [ Google Scholar]
- Anguille S, Smits EL, Cools N, Goossens H, Berneman ZN, Van Tendeloo VF. Short-term cultured, interleukin-15 differentiated dendritic cells have potent immunostimulatory properties. J Transl Med 2009; 7:109. doi: 10.1186/1479-5876-7-109 [Crossref] [ Google Scholar]
- Farkas Á, Kemény L. Interferon-α in the generation of monocyte-derived dendritic cells: recent advances and implications for dermatology. Br J Dermatol 2011; 165:247-54. doi: 10.1111/j.1365-2133.2011.10301.x [Crossref] [ Google Scholar]
- Gigante M, Mandic M, Wesa AK, Cavalcanti E, Dambrosio M, Mancini V. Interferon-alpha (IFN-α)–conditioned DC Preferentially Stimulate Type-1 and Limit Treg-type In Vitro T-cell Responses From RCC Patients. J Immunother 2008; 31:254-62. doi: 10.1097/CJI.0b013e318167b023 [Crossref] [ Google Scholar]
- Harris KM. Monocytes differentiated with GM-CSF and IL-15 initiate Th17 and Th1 responses that are contact-dependent and mediated by IL-15. J Leukoc Biol 2011; 90:727-34. doi: 10.1189/jlb.0311132 [Crossref] [ Google Scholar]
- Leplina OY, Tyrinova T V, Tikhonova MA, Ostanin AA, Chernykh ER. Interferon alpha induces generation of semi-mature dendritic cells with high pro-inflammatory and cytotoxic potential. Cytokine 2015; 71:1-7. doi: 10.1016/j.cyto.2014.07.258 [Crossref] [ Google Scholar]
- McGranahan N, Furness AJS, Rosenthal R, Ramskov S, Lyngaa R, Saini SK. Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science 2016; 351:1463-9. doi: 10.1126/science.aaf1490 [Crossref] [ Google Scholar]
- Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 2015; 348:124-8. doi: 10.1126/science.aaa1348 [Crossref] [ Google Scholar]
- Snyder A, Makarov V, Merghoub T, Yuan J, Zaretsky JM, Desrichard A. Genetic Basis for Clinical Response to CTLA-4 Blockade in Melanoma. N Engl J Med 2014; 371:2189-99. doi: 10.1056/NEJMoa1406498 [Crossref] [ Google Scholar]
- Van Allen EM, Miao D, Schilling B, Shukla SA, Blank C, Zimmer L. Genomic correlates of response to CTLA-4 blockade in metastatic melanoma. Science 2015; 350:207-11. doi: 10.1126/science.aad0095 [Crossref] [ Google Scholar]
- Ophir E, Bobisse S, Coukos G, Harari A, Kandalaft LE. Personalized approaches to active immunotherapy in cancer. Biochim Biophys Acta - Rev Cancer 2016; 1865:72-82. doi: 10.1016/j.bbcan.2015.07.004 [Crossref] [ Google Scholar]
- Vandenberk L, Belmans J, Van Woensel M, Riva M, Van Gool SW. Exploiting the Immunogenic Potential of Cancer Cells for Improved Dendritic Cell Vaccines. Front Immunol 2015; 6:663. doi: 10.3389/fimmu.2015.00663 [Crossref] [ Google Scholar]
- Kandalaft LE, Chiang CL, Tanyi J, Motz G, Balint K, Mick R. A Phase I vaccine trial using dendritic cells pulsed with autologous oxidized lysate for recurrent ovarian cancer. J Transl Med 2013; 11:149. doi: 10.1186/1479-5876-11-149 [Crossref] [ Google Scholar]
- Chiang CLL, Kandalaft LE, Tanyi J, Hagemann AR, Motz GT, Svoronos N. A dendritic cell vaccine pulsed with autologous hypochlorous acid-oxidized ovarian cancer lysate primes effective broad antitumor immunity: From bench to bedside. Clin Cancer Res 2013; 19:4801-15. doi: 10.1158/1078-0432.CCR-13-1185 [Crossref] [ Google Scholar]
- Word AP, Nezafati KA, Cruz PD. Treatment of warts with contact allergens. Contact Dermatitis 2015; 26:32-7. doi: 10.1097/DER.0000000000000092 [Crossref] [ Google Scholar]
- Makmura L, Hamann M, Areopagita A, Furuta S, Muñoz A, Momand J. Development of a Sensitive Assay to Detect Reversibly Oxidized Protein Cysteine Sulfhydryl Groups. Antioxid Redox Signal 2001; 3:1105-18. doi: 10.1089/152308601317203611 [Crossref] [ Google Scholar]
- Garrigan K, Moroni-Rawson P, McMurray C, Hermans I, Abernethy N, Watson J. Functional comparison of spleen dendritic cells and dendritic cells cultured in vitro from bone marrow precursors. Blood 1996; 88:3508-12. [ Google Scholar]
- Krysko D V, Garg AD, Kaczmarek A, Krysko O, Agostinis P, Vandenabeele P. Immunogenic cell death and DAMPs in cancer therapy. Nat Rev Cancer 2012; 12:860-75. doi: 10.1038/nrc3380 [Crossref] [ Google Scholar]
- Kepp O, Senovilla L, Vitale I, Vacchelli E, Adjemian S, Agostinis P. Consensus guidelines for the detection of immunogenic cell death. Oncoimmunology 2014; 3:e955691. doi: 10.4161/21624011.2014.955691 [Crossref] [ Google Scholar]
- Kazama H, Ricci J-E, Herndon JM, Hoppe G, Green DR, Ferguson TA. Induction of Immunological Tolerance by Apoptotic Cells Requires Caspase-Dependent Oxidation of High-Mobility Group Box-1 Protein. Immunity 2008; 29:21-32. doi: 10.1016/j.immuni.2008.05.013 [Crossref] [ Google Scholar]
- Prokopowicz ZM, Arce F, Biedroń R, Chiang CL-L, Ciszek M, Katz DR. Hypochlorous acid: a natural adjuvant that facilitates antigen processing, cross-priming, and the induction of adaptive immunity. J Immunol 2010; 184:824-35. doi: 10.4049/jimmunol.0902606 [Crossref] [ Google Scholar]
- Holzer AM, Kaplan LL, Levis WR. Haptens as drugs: contact allergens are powerful topical immunomodulators. J Drugs Dermatol 2006; 5:410-6. [ Google Scholar]
- Douglas KT, Nadvi IN. Inhibition of glyoxalase I: a possible transition-state analogue inhibitor approach to potential antineoplastic agents?. FEBS Lett 1979; 106:393-6. [ Google Scholar]
- Taniguchi H, Horinaka M, Yoshida T, Yano K, Goda AE, Yasuda S. Targeting the Glyoxalase Pathway Enhances TRAIL Efficacy in Cancer Cells by Downregulating the Expression of Antiapoptotic Molecules. Mol Cancer Ther 2012; 11:2294-300. doi: 10.1158/1535-7163.MCT-12-0031 [Crossref] [ Google Scholar]
- Yamamoto Y, Ohno M, Eguchi S. Oxidative Rearrangement of 4-Hydroxy-2-cyclobutenone A New Route to Highly Substituted Furanones from Squaric Acid. J Org Chem 1994; 59:4707-9. doi: 10.1021/jo00096a001 [Crossref] [ Google Scholar]
- Skopinska-Wisniewska J, Kuderko J, Bajek A, Maj M, Sionkowska A, Ziegler-Borowska M. Collagen/elastin hydrogels cross-linked by squaric acid. Mater Sci Eng C 2016; 60:100-8. doi: 10.1016/j.msec.2015.11.015 [Crossref] [ Google Scholar]
- Palitzsch B, Hartmann S, Stergiou N, Glaffig M, Schmitt E, Kunz H. A fully synthetic four-component antitumor vaccine consisting of a mucin glycopeptide antigen combined with three different T-helper-cell epitopes. Angew Chemie - Int Ed 2014; 53:14245-9. doi: 10.1002/anie.201406843 [Crossref] [ Google Scholar]
- Kandalaft LE, Powell DJ, Chiang CL, Tanyi J, Kim S, Bosch M. Autologous lysate-pulsed dendritic cell vaccination followed by adoptive transfer of vaccine-primed ex vivo co-stimulated T cells in recurrent ovarian cancer. Oncoimmunology 2013; 2:e22664. doi: 10.4161/onci.22664 [Crossref] [ Google Scholar]
- Chiang CLL, Hagemann AR, Leskowitz R, Mick R, Garrabrant T, Czerniecki BJ. Day-4 myeloid dendritic cells pulsed with whole tumor lysate are highly immunogenic and elicit potent anti-tumor responses. PLoS One 2011:6. doi: 10.1371/journal.pone.0028732 [Crossref]
- Chiang CL-L, Ledermann JA, Rad AN, Katz DR, Chain BM. Hypochlorous acid enhances immunogenicity and uptake of allogeneic ovarian tumor cells by dendritic cells to cross-prime tumor-specific T cells. Cancer Immunol Immunother 2006; 55:1384-95. doi: 10.1007/s00262-006-0127-9 [Crossref] [ Google Scholar]
- Chiang CL-L, Ledermann JA, Aitkens E, Benjamin E, Katz DR, Chain BM. Oxidation of ovarian epithelial cancer cells by hypochlorous acid enhances immunogenicity and stimulates T cells that recognize autologous primary tumor. Clin Cancer Res 2008; 14:4898-907. doi: 10.1158/1078-0432.CCR-07-4899 [Crossref] [ Google Scholar]
- Biedroń R, Konopiński MK, Marcinkiewicz J, Józefowski S. Oxidation by neutrophils-derived HOCl increases immunogenicity of proteins by converting them into ligands of several endocytic receptors involved in antigen uptake by dendritic cells and macrophages. PLoS One 2015; 10:e0123293. doi: 10.1371/journal.pone.0123293 [Crossref] [ Google Scholar]
- Gocheva V, Wang H-W, Gadea BB, Shree T, Hunter KE, Garfall AL. IL-4 induces cathepsin protease activity in tumor-associated macrophages to promote cancer growth and invasion. Genes Dev 2010; 24:241-55. doi: 10.1101/gad.1874010 [Crossref] [ Google Scholar]
- Hatfield P, Merrick AE, West E, O'Donnell D, Selby P, Vile R. Optimization of Dendritic Cell Loading With Tumor Cell Lysates for Cancer Immunotherapy. J Immunother 2008; 31:620-32. doi: 10.1097/CJI.0b013e31818213df [Crossref] [ Google Scholar]
- Uddin S, Majchrzak B, Woodson J, Arunkumar P, Alsayed Y, Pine R. Activation of the p38 mitogen-activated protein kinase by type I interferons. J Biol Chem 1999; 274:30127-31. doi: 10.1074/jbc.274.42.30127 [Crossref] [ Google Scholar]
- elger JC, Alagbe O, Pace TWW, Woolwine BJ, Hu F, Raison CL. Early activation of p38 mitogen activated protein kinase is associated with interferon-alpha-induced depression and fatigue. Brain Behav Immun 2011; 25:1094-8. doi: 10.1016/j.bbi.2011.02.015 [Crossref] [ Google Scholar]
- Lapenta C, Santini SM, Spada M, Donati S, Urbani F, Accapezzato D. IFN-α-conditioned dendritic cells are highly efficient in inducing cross-priming CD8+ T cells against exogenous viral antigens. Eur J Immunol 2006; 36:2046-60. doi: 10.1002/eji.200535579 [Crossref] [ Google Scholar]
- Slavik JM, Hutchcroft JE, Bierer BE. CD28/CTLA-4 and CD80/CD86 families. Immunol Res 1999; 19:1-24. doi: 10.1007/BF02786473 [Crossref] [ Google Scholar]
- Kotera Y, Shimizu K, Mulé JJ. Comparative analysis of necrotic and apoptotic tumor cells as a source of antigen(s) in dendritic cell-based immunization. Cancer Res 2001; 61:8105-9. [ Google Scholar]
- Kim HS, Choo YS, Koo T, Bang S, Oh TY, Wen J. Enhancement of antitumor immunity of dendritic cells pulsed with heat-treated tumor lysate in murine pancreatic cancer. Immunol Lett 2006; 103:142-8. doi: 10.1016/j.imlet.2005.10.021 [Crossref] [ Google Scholar]
- Shi H, Cao T, Connolly JE, Monnet L, Bennett L, Chapel S. Hyperthermia enhances CTL cross-priming. J Immunol 2006; 176:2134-41. [ Google Scholar]
- Fucikova J, Moserova I, Truxova I, Hermanova I, Vancurova I, Partlova S. High hydrostatic pressure induces immunogenic cell death in human tumor cells. Int J Cancer 2014; 135:1165-77. doi: 10.1002/ijc.28766 [Crossref] [ Google Scholar]
- Xu P, Kelly M, Vann WF, Qadri F, Ryan ET, Kováč P. Conjugate Vaccines from Bacterial Antigens by Squaric Acid Chemistry: A Closer Look. ChemBioChem 2017; 18:799-815. doi: 10.1002/cbic.201600699 [Crossref] [ Google Scholar]
- Sayeed MA, Bufano MK, Xu P, Eckhoff G, Charles RC, Alam MM. A cholera conjugate vaccine containing ospecific polysaccharide (OSP) of V cholera o1 inaba and recombinant fragment of tetanus toxin heavy chain (OSP:rTTHC) induces serum, memory and lamina proprial responses against OSP and is protective in mice. PLoS Negl Trop Dis 2015; 9:e0003881. doi: 10.1371/journal.pntd.0003881 [Crossref] [ Google Scholar]
- Garg AD, Coulie PG, Van den Eynde BJ, Agostinis P. Integrating Next-Generation Dendritic Cell Vaccines into the Current Cancer Immunotherapy Landscape. Trends Immunol 2017; 38:577-93. doi: 10.1016/j.it.2017.05.006 [Crossref] [ Google Scholar]
- Bol KF, Schreibelt G, Gerritsen WR, De Vries IJM, Figdor CG. Dendritic cell-based immunotherapy: State of the art and beyond. Clin Cancer Res 2016; 22:1897-906. doi: 10.1158/1078-0432.CCR-15-1399 [Crossref] [ Google Scholar]