Bioimpacts. 2025;15:30030.
doi: 10.34172/bi.30030
Review
STAT signaling pathways in immune cells and their associated mechanisms in cancer pathogenesis
Sepideh Sohrabi Conceptualization, Investigation, Visualization, Writing – original draft, Writing – review & editing, 1, 2 
Shiva Alipour Investigation, Writing – review & editing, 1, 2 
Zahra Ghahramanipour Investigation, Writing – review & editing, 1 
Javad Masoumi Investigation, Writing – review & editing, 1
Behzad Baradaran Project administration, Supervision, Validation, Writing – review & editing, 1, 2, * 
Author information:
1Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
2Department of Immunology, Faculty of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran
Abstract
Introduction:
Signal transducer and activator of transcriptions (STATs) factors as critical proteins in cell signaling regulate diverse biological processes such as differentiation and proliferation of cells. STATs have been shown to play distinct roles in modulating immune responses mediated by innate and adaptive immune cell subsets due to their significant roles in cytokine signaling.
Methods:
In the current study, we review recent studies on the contribution of individual STAT proteins to cytokine signaling, development, and activity of diverse immune cells that constitute the whole immune system and help its performance against endogenous or exogenous agents with a particular focus on meaningful STAT factor in each of innate and adaptive immune cells’ subsets to clarify their function in favor of the tumor or against it.
Results:
Dysregulation of signaling pathways in the immune cells is associated with various immune disorders, such as the inability of immune system cells in the effective destruction of cancerous cells. Increase of knowledge about these pathways’ functions is essential to understand how they can be effectively targeted to eliminate tumors.
Conclusion:
The majority of immune cells use the Jak/STAT signaling pathway, which is one of the most important signaling pathways with a role in induction of proper immune responses. Since each of the STAT factors has a specific role in diverse immune cells’ subsets, appropriate targeting of them can be a promising strategy for patients who suffer from immune system disorders; specifically it can be beneficial as an approach for cancer immunotherapy.
Keywords: Innate immune system, Adaptive immune system, STAT, Cancer
Copyright and License Information
© 2025 The Author(s).
This work is published by BioImpacts as an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (
http://creativecommons.org/licenses/by-nc/4.0/). Non-commercial uses of the work are permitted, provided the original work is properly cited.
Funding Statement
This study was supported by the Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran.
Introduction
The immune system is basically divided into innate and adaptive immune systems, comprising cells that are known to act against endogenous or exogenous pathogens.1 The innate immune system can be considered the fastest and frontline of immune defense against environmental agents. The innate immune sy0stem is known chiefly for neutrophils and macrophages (MQs) as phagocytes.2 Natural killer (NK) cells are innate cytotoxic cells. These cells recognize infected cells by natural cytotoxicity receptors (NCRs).3 Other cells such as cancer-associated fibroblasts (CAFs), myeloid-derived suppressor cells (MDSCs), eosinophils, basophils, mast cells, and dendritic cells (DCs) are included in this category. The distinguishing superiority of the adaptive immune system over the innate immune system is the existence of memory cells and advanced recognition systems. The adaptive immune system consists of the humoral (B cell) and cellular immune (helper T cells (Ths), regulatory T (Treg) cells, and cytotoxic T lymphocytes (CTLs)) systems. The humoral immune system consists of B cells, which get activated by the B cell receptor (BCR) that is necessary for their activation, differentiation, and immunoglobulin (Ig) production.4 Two fates of B cells would be developing into plasma cells or memory B cells. Igs stimulate the complement system to trigger the destruction of bacterial cells. Moreover, antibodies ease the opsonization of foreign particles. The cellular immune system is mainly composed of T cells. T cells interact with antigen-presenting cells (APCs) through major histocompatibility complex (MHC) molecules and receive signals by T cell receptor (TCR) to counteract the pathogen directly (CD8+ T cells kill infected cells) or by other immune cells such as B cells and make them produce antibodies (especially CD4+ T cells).5 T lymphocytes also have regulatory effects on the immune system, which is discussed in more detail below. Immune system components are associated with each other by divergent signaling pathways, which make this complicated plexus to do their function. Immune cells express numerous types of signaling molecules for different goals such as differentiation, recognition, antigen presentation, immunogenic function, nourishment, apoptosis, and interaction with immune or non-immune cells. Learning about these pathways can be a promising way to understand immune cells' cross-talking and,subsequently help us use the information to direct some parts of the immune system for particular therapeutic goals such as treatment of autoimmune disease, cancer, and rejection of transplanted organs, and disease progression/regression foresight.6-8 One of the most significant signaling pathways employed in the majority of immune cells is the Janus kinase (Jak)/signal transducer and activator of transcription (STAT) signaling pathway.
STAT proteins are known as regulators of various biological processes like cellular maintenance, proliferation, immunity, and differentiation that occur after exposure to various agents consisting of growth factors, cytokines, and other stimuli.9,10 Gene ablation investigations of the various STATs discovered their crucial functions, notably in the formation and maintenance of blood and immune cells.11 Every STAT comprises between 750 and 900 residues of amino acids.12 Seven members of this protein exist: STAT1, STAT2, STAT3, STAT4, STAT5A, STAT5B, and STAT6. Although the mentioned STATs have some similarities, each of them is located in a special chromosomal location and responds to specific extracellular stimuli, thus regulating different cellular processes.13,14 Six domains organize STAT proteins, i.e., the N-terminal domain (NTD), coiled-coil domain (CCD), deoxyribonucleic acid (DNA) binding domain (DBD), linker domain (LD), Src-homology 2 (SH2) domain, and transcription activation domain (TAD).15 Entering the nucleus and STAT-constant dimerization are done through the NTD.16 The DBD is used for attaching to particular palindromic sequences in the promoters of objective genes.17 The CCD domain mediates cooperation among several proteins related to nuclear import and export.18 The SH2 domain is needed to recognize tyrosine phosphorylation on receptor subunits.19 Finally, the TAD, located in the C-terminal region and encompassing tyrosine and serine phosphorylation sites, is required for the highest STAT-regulated gene transcriptional activation.20 Mechanistically, cytokine receptors are phosphorylated through the Jak. This process can activate cytokine receptor-associated kinases that phosphorylate the STAT molecules in the tyrosine site. The SH2 domain exerts the function of homo-or hetero-dimerization of two STAT molecules and lets them transmit to the nucleus for attaching to special DNA elements for mediating specific gene expression.21 Targeting the Jak/STAT pathway during type I and type II cytokine signal transduction has been found to exert positive therapeutic benefits in the area of numerous autoimmune disorders, including rheumatoid arthritis (RA), systemic lupus erythematosus(SLE), psoriasis (PsO), and inflammatory bowel disease (IBD) and various cancers.22 There are specific Jak inhibitors for each autoimmune disorder. For instance, Tofacitinib, baricitinib, upadacitinib, and peficitinib are the four oral inhibitors of Jak and subsequent downstream STAT proteins that have been authorized for the treatment of RA. Tofacitinib was the first JAK1/JAK2/JAK3 inhibitor with a greater preference for JAK1 and JAK3 to be authorized by the European Medicines Agency (EMA; March 2017) and US Food and Drug Administration (FDA; November 2012) to treat moderate-to-severe active RA.23 In summary, the interactions between the Jak/STAT pathway and pathways mediated by other immune receptors possess the capability to regulate cytokine signaling in various immune cells. Therefore, considering cytokine signaling and their related Jak/STAT signaling pathway is crucial in immune cells and treating different related disorders.24 A comprehensive search was conducted throughout the Web of Science, PubMed, and Scopus databases to retrieve scholarly papers in the categories of original research, literature reviews, systematic reviews, and meta-analyses. The search was limited to articles published before to the year 2023. In this review, we review new findings about the function of STAT proteins in the battle against cancer, specifically how these proteins influence cytokine signaling, development, and activity in innate and adaptive immune cell subsets.
Overview of the JAK/STAT-3 pathway
STAT3 is located in chromosomal region 17 and exists in eukaryotes with conservation.25 Phosphorylation of the linked Jak's intracellular domain is instantly triggered when cytokines attach to the tyrosine kinase-associated receptor on the cell membrane. The phosphorylated tyrosine site on Jak serves as a binding point to attract STAT3 with the SH2 domain for phosphorylation alteration. Moreover, the phosphorylated STAT3 is transported to the nucleus to interact with the genes and control specific transcriptional activity (Fig. 1).26 It has been revealed that the expression of the STAT3 target gene can be stimulated in the case of recruiting nuclear factor-κB (NF-κB) and Lys-685 acetylation without requiring STAT3 phosphorylation.27 Histone acetyltransferases (like CREB-binding protein (CBP)/histone acetyltransferase p300 (p300)) acetylate lysine 685 (K685) and histone methyltransferases (like histone-lysine N-methyltransferase SET9 (SET9)) methylate lysine 140 (K140), favoring or impairing STAT3 transcriptional function, respectively.28 Two isoforms with diverse functions, including STAT3α and STAT3β, organize STAT3. Phosphorylation of tyrosine 705 (Y705) or serine 727 (S727) results in STAT3 activation.29 However, S727 deficiency in STAT3β causes this isoform activation just in the condition of Y705 phosphorylation. STAT3α can exert transcription activity better than STAT3β. On the other hand, STAT3β is more potent for binding to specific DNA sites in contrast to STAT3α. Some stimulus consisting of interferon (IFN)-Is, interleukin (IL)-21, IL-27, granulocyte colony-stimulating factor (G-CSF), and leptin, the members of IL-10 family (IL-10, IL-19, IL-20, IL-22, IL-24, and IL-26), and the participants of IL-6 family (ciliary neurotrophic factor )CNTF(, leukemia inhibitory factor (LIF), oncostatin M )OSM(,cardiotrophin-1 (CT-1), IL-6, IL-11, IL-31, etc.) possess the capability to induce STAT3.10,30,31 STAT3 plays different roles including controlling Th17 cells’ differentiation, improving tumor progression activity of tumor-associated MQ (TAMs) and MDSCs, hampering cell apoptosis in multiple myeloma patients, resistant to chemotherapy in high-grade breast cancers, and modulating cancer stem cells (CSCs).32-35 Hypoxia-inducible factor-1α-Jak1-STAT3, retinol-binding protein 4-Jak2-STAT3, IL-6-Jak2-STAT3, and IL-6-Jak1-STAT3 have been considered the pathways by which CSCs’ survival increases during breast or colon cancer.35
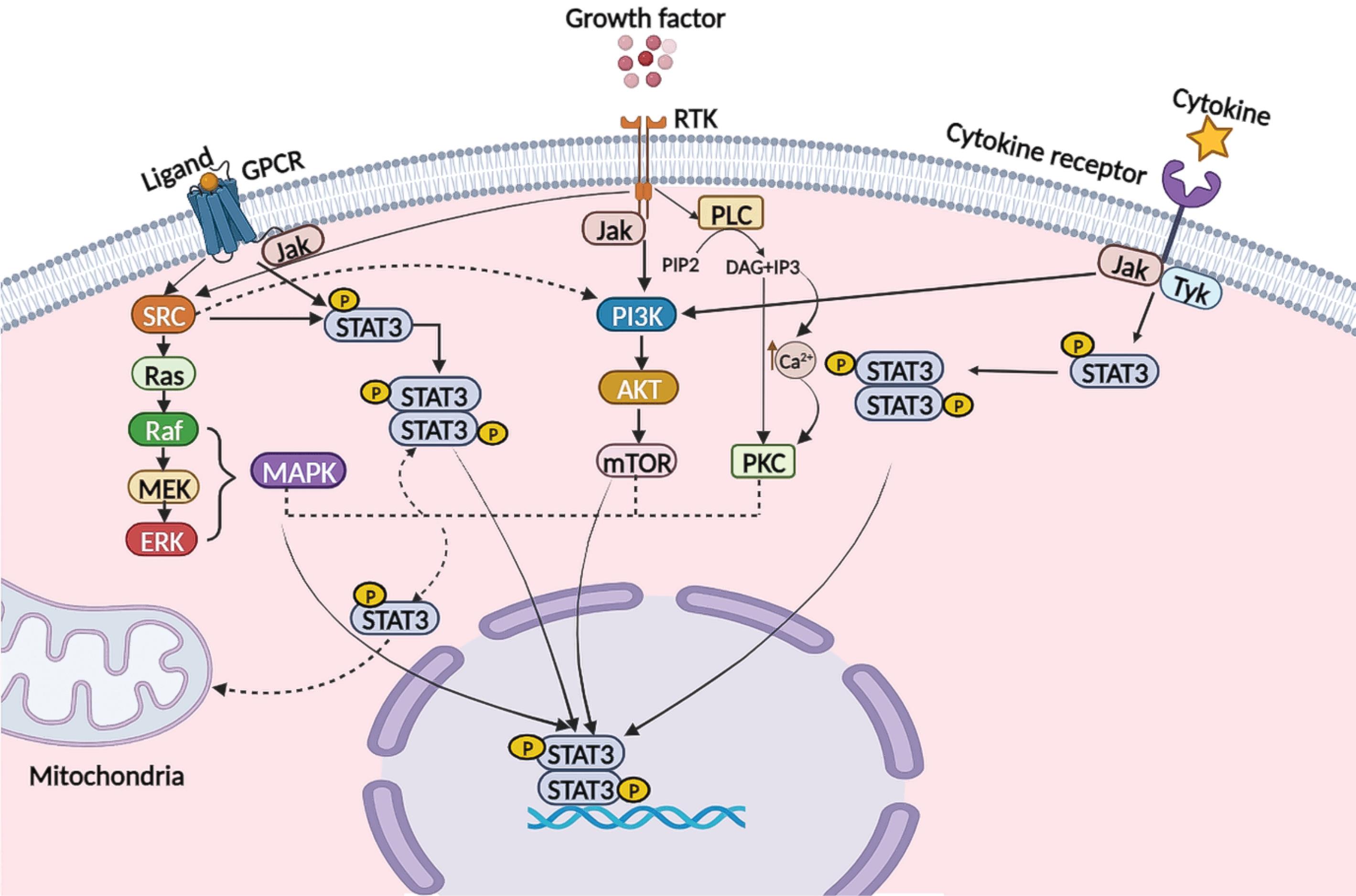

Fig. 1.
STAT3 signaling pathway. After induction of GPCR, RTK, and cytokine receptors by their associated ligands, STAT3 undergoes Y705 phosphorylation, forms homodimers or heterodimers, and moves to the nucleus where it transcriptionally affects several biological activities. These receptors work via engaging adaptor kinases (like Jaks and SRC) to spread downstream signals since, except for receptor tyrosine kinases, they lack intrinsic kinase activity. Furthermore, serine/threonine kinases (such as MAPK, mTOR, and PKC) can phosphorylate STAT3 at S727, enhancing STAT3 transcriptional function in the cell's nucleus or guiding STAT3 to the mitochondria. Abbreviations: AKT: protein kinase B; Ca2+: calcium ion; DAG: diacylglycerol; ERK: extracellular signal-regulated kinase; GPCR: G-protein coupled receptor; IP3: inositol triphosphate; Jak: Janus kinase; MAPK: mitogen-activated protein kinase; MEK: mitogen-activated protein kinase; mTOR: mechanistic target of rapamycin; P: phosphorylation; PI3K: phosphatidylinositol 3-kinase; PIP2: phosphatidylinositol 4,5-bisphosphate; PKC: protein kinase C; PLC: phospholipase C; RAF: rapidly accelerated fibrosarcoma; RAS: rat sarcoma virus; RTK: receptor tyrosine kinase; S727: serine 727; SRC: proto-oncogene tyrosine-protein kinase; STAT: signal transducer and activator of transcription; Tyk: Non-receptor tyrosine-protein kinase; Y705: tyrosine 705.
.
STAT3 signaling pathway. After induction of GPCR, RTK, and cytokine receptors by their associated ligands, STAT3 undergoes Y705 phosphorylation, forms homodimers or heterodimers, and moves to the nucleus where it transcriptionally affects several biological activities. These receptors work via engaging adaptor kinases (like Jaks and SRC) to spread downstream signals since, except for receptor tyrosine kinases, they lack intrinsic kinase activity. Furthermore, serine/threonine kinases (such as MAPK, mTOR, and PKC) can phosphorylate STAT3 at S727, enhancing STAT3 transcriptional function in the cell's nucleus or guiding STAT3 to the mitochondria. Abbreviations: AKT: protein kinase B; Ca2+: calcium ion; DAG: diacylglycerol; ERK: extracellular signal-regulated kinase; GPCR: G-protein coupled receptor; IP3: inositol triphosphate; Jak: Janus kinase; MAPK: mitogen-activated protein kinase; MEK: mitogen-activated protein kinase; mTOR: mechanistic target of rapamycin; P: phosphorylation; PI3K: phosphatidylinositol 3-kinase; PIP2: phosphatidylinositol 4,5-bisphosphate; PKC: protein kinase C; PLC: phospholipase C; RAF: rapidly accelerated fibrosarcoma; RAS: rat sarcoma virus; RTK: receptor tyrosine kinase; S727: serine 727; SRC: proto-oncogene tyrosine-protein kinase; STAT: signal transducer and activator of transcription; Tyk: Non-receptor tyrosine-protein kinase; Y705: tyrosine 705.
The role of STATs in the innate immune system
Cancer-associated fibroblasts
CAFs, as the resident cells in the tumor's mass milieu, mostly originate from mesoderm, similar to the other fibroblasts. They have interlinking functions between tumor cells and the immune system, so these cells have been studied to see if they can be utilized in cancer immunotherapy. It is shown that CAFs use various signaling pathways, such as STATs, to produce the mentioned effects.36 Accumulating evidence suggests that a reduction in longevity in patients with colorectal cancer can be associated with STAT3 phosphorylation in CAFs.37 Multiple cytokines, such as leukemia inhibitory factor (LIF), can intensify activation of STAT3 in CAFs, resulting in the production of immunosuppressive factors, including transforming growth factor beta (TGF-β), chemokine (C-C motif) ligand 2 (CCL-2), and vascular endothelial growth factor (VEGF) for the tumor-promoting function of these fibroblasts.38,39 Moreover, IL-1 and IL-6 are secreted from TGF-β-stimulated CAFs, leading to glycoprotein 130 (GP130)/STAT3 signaling in tumor cells and establishing further metastasis.40,41 Elevated STAT3 activity in CAFs also affects other immune cells. For example, secreted CCL-2 through CAFs with STAT3 hyperactivation may trigger MDSC immunosuppressive activation in hepatocellular carcinoma.39 CAFs-secreted IL-6 increases STAT3 activation in DCs, which enables them to expand Treg cells and inhibit effector T-cell proliferation (Fig. 2).42
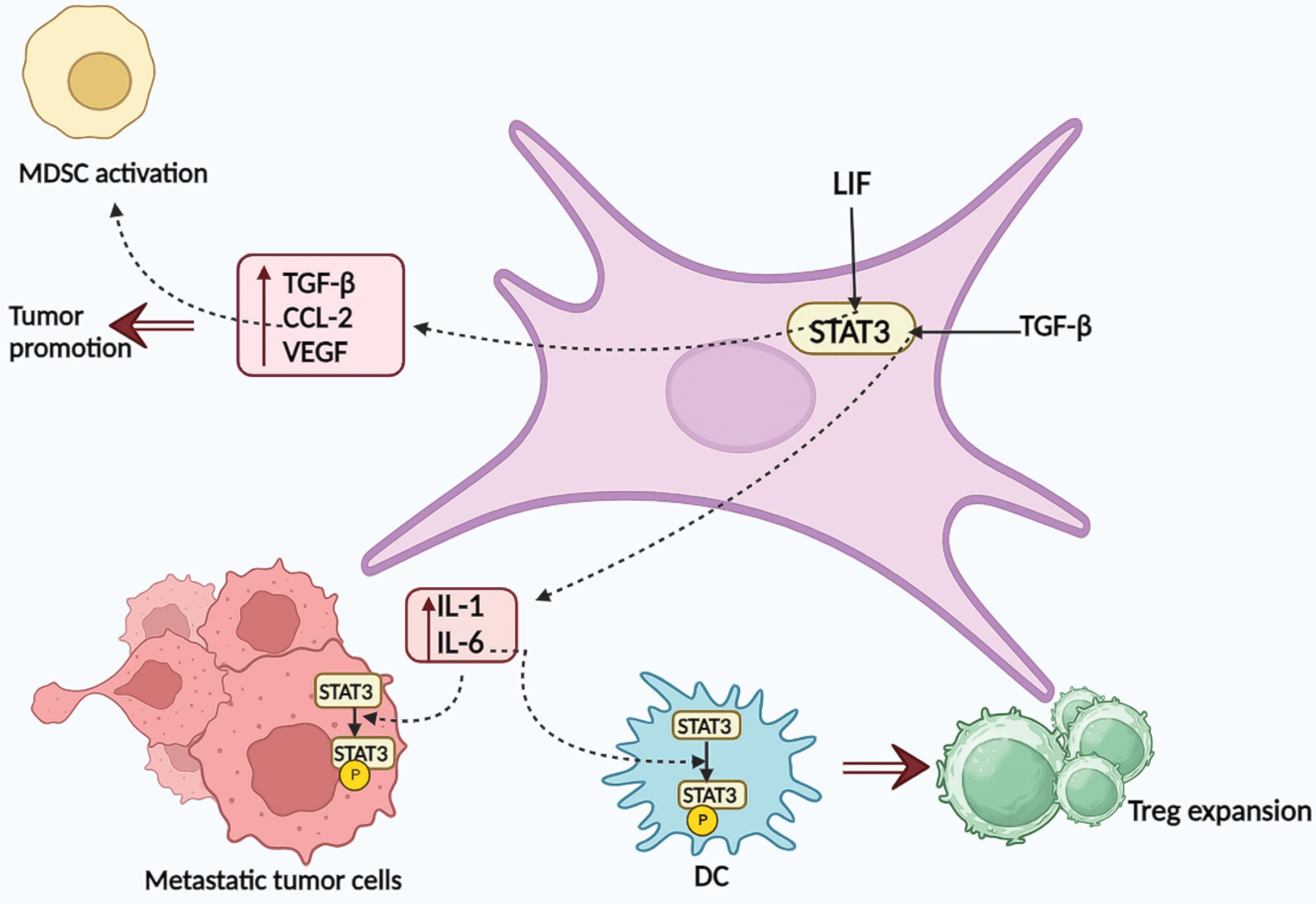
Fig. 2.
Different roles of STAT3 in CAFs. LIF can accelerate STAT3 activation in CAFs, leading to the synthesis of immunosuppressive substances, including TGF-β, CCL-2, and VEGF, for the tumor-promoting activity of these fibroblasts. TGF-stimulated CAFs release IL-1 and IL-6, which activate STAT3 signaling in tumor cells and promote further metastasis. MDSC immunosuppressive response may be brought on by secreted CCL-2 through CAFs with hyperactivated STAT3. Furthermore, CAFs-secreted IL-6 increases STAT3 activation in DCs, which enables them to expand Treg cells. Abbreviations: CAF: cancer-associated fibroblast; CCL: chemokine (C-C motif) ligand; DC: dendritic cell; IL: interleukin; LIF: leukemia inhibitory factor; STAT: signal transducer and activator of transcription; TGF-β: transforming growth factor beta; Treg: regulatory T; VEGF: vascular endothelial growth factor.
.
Different roles of STAT3 in CAFs. LIF can accelerate STAT3 activation in CAFs, leading to the synthesis of immunosuppressive substances, including TGF-β, CCL-2, and VEGF, for the tumor-promoting activity of these fibroblasts. TGF-stimulated CAFs release IL-1 and IL-6, which activate STAT3 signaling in tumor cells and promote further metastasis. MDSC immunosuppressive response may be brought on by secreted CCL-2 through CAFs with hyperactivated STAT3. Furthermore, CAFs-secreted IL-6 increases STAT3 activation in DCs, which enables them to expand Treg cells. Abbreviations: CAF: cancer-associated fibroblast; CCL: chemokine (C-C motif) ligand; DC: dendritic cell; IL: interleukin; LIF: leukemia inhibitory factor; STAT: signal transducer and activator of transcription; TGF-β: transforming growth factor beta; Treg: regulatory T; VEGF: vascular endothelial growth factor.
Myeloid-derived suppressor cells
MDSCs are the activated forms of monocytes and neutrophils, capable of immunosuppressive effects. MDSCs are classified into two subgroups, named monocytic MDSCs (M-MDSCs) and granulocytic/polymorphonuclear MDSCs (PMN-MDSCs), based on the monocytic or granulocytic myeloid cell lineages. They get activated to restrict chronic infection and autoimmune disease, although they deteriorate malignancy-caused conditions in the body. MDSCs, like many other immune cells, use the plexus of interlinking signal pathways such as STAT pathways for their survival and action.43,44 Some studies displayed that STAT1 can play a role in IFN-γ and IL-1β activation, and this protein loss in MDSCs inhibits their suppressive effects on T cells due to lack of nitric oxide synthase (iNOS) and arginase activation.45 The MDSC population can be decreased due to IFN regulatory factor-8 (IRF-8) overexpression. It has been revealed that induction of G-CSF/granulocyte-MQ colony-stimulating factor (GM-CSF) diminishes IRF-8 expression after recruiting STAT3 and STAT5 pathways in these cells.46 Although several mechanisms could regulate the production of reactive oxygen species (ROS) in different cells, in MDSCs, it has been indicated that STAT3, by increasing activation of NADPH oxidase (NOS) 2, has a vital role in the production of augmented ROS. Moreover, STAT3 activation mediates MDSC generation and migration by regulating the S100 calcium-binding protein A8 (S100A8) and S100A9 expression.47,48 Moreover, β2 adrenergic receptors on MDSCs can be mediated through STAT3,49 and the IL-6/STAT3 axis regulates MDSC differentiation in tumor microenvironment (TME).50 Peng Qu et al. and colleagues, in their study about lung bronchioalveolar adenocarcinoma, have demonstrated thatincreased expression of STAT3 in lung alveolar cells induced production of IL-6 and IL-1β, resulting in MDSC enhancement in the lung. All of the above indicated that STAT3 led to inflammation and tumor progression via augmenting MDSC involvement.51 Another study revealed that enhancement of suppressors of cytokine signaling (SOCS) 1 as STAT3 inhibitor occurs after stimulator of IFN genes (STING) induction leading to tumor suppression via regulating MDSC differentiation.52 Furthermore, in the mice model of breast cancer, utilizing both direct suppressor of STAT3 (HJC0152) and STING agonist c-diAM (PS)2 is associated with enhancement of CD8+ T cells and also reduction of Treg cells and MDSCs in the TME.53 Another inhibitory function of STAT in MDSCs can be defined by the STAT6 subtype, which can be activated after IL-4 and IL-13 attachment to CD124 receptors in mentioned cells, leading to overexpression of TGF-β and arginase and further diminishing of T cell and M1 MQ recruitment.45,54
Neutrophils
Neutrophils, also called PMN leukocytes, are the most affluent circulating granulocytes. They are known as the frontline of immune defense cells and play the role of phagocytes. PMNs are derived from bone marrow in a naïve form, which means they need to be differentiated and activated, and the Jak/STAT signaling pathways can play an essential role in this procedure.55 As mentioned before, survival, activation, and differentiation of neutrophils can be controlled by the involvement of type II cytokine receptors and following the Jak/STAT signaling pathway.56,57 Moreover, Jak/STAT, besides phosphoinositide 3-kinases (PI3K) signaling, has the potential of controlling the anti-apoptotic B cell lymphoma-2 (Bcl-2) family protein A1/Bcl-2 related gene expressed in fetal liver (BFL-1) expression by which neutrophils mediate their maintenance and hemostasis.58 Some investigations demonstrate that all-trance retinoic acid (atRA) increases SOCS1 expression for suppressing STAT1, which leads to lower neutrophil gathering, enhances N2 neutrophil phenotype polarization, and declines neutrophil extracellular traps (NET) organization.59 STAT2 besides extracellular signal-regulated kinases (ERK), protein tyrosine kinase 2 (PTK2), rat sarcoma virus (RAS), rapidly accelerated fibrosarcoma (Raf)-1, IkappaB (IκB), and GBY involves in N1 phenotype signaling.60 It is better to note that Oncostatin M (OSM) (neutrophil-derived granule cargo) involves Jak/STAT, PI3K, and mitogen-activated protein kinase (MAPK) signaling pathways by which tumor angiogenesis and neovascularization occur, mainly in skin squamous cell carcinoma.61,62 Additionally, in acute inflammation, infiltration of inflammatory neutrophils is restricted after IL-6-induced STAT3 activation. Thus, the activation of STAT3 after IL-6 induction is essential for ending the innate immune response in such inflammations.58 Another study has displayed that the outcome of cross-talkbetweentumor-associated neutrophils (TANs) and neoplastic cells is GM-CSF secretion from tumor cells, which in turn induces neutrophil synthesis and IL-6 generation. IL-6 can also boost VEGF-A production via the Jak/STAT signaling pathway to tumor progression.63
Eosinophils
Eosinophils are bone marrow-derived granulocytes. An activated eosinophil gets degranulated and releases variant proinflammatory molecules. It is known that these cells are also capable of immunomodulatory effects. Eosinophils are a part of IgE-mediated hypersensitivity reactions. STAT signaling pathways are necessary for the eosinophil's function and survival.64 Based on investigations, IL-31 attachment to the IL-13 receptor of eosinophil activates Jak/STAT, leading to the postponement of eosinophil apoptosis and provoking the release of proinflammatory cytokines and chemokines.65,66 In nasal polyps, phosphorylated-STAT3 (pSTAT3) and VEGF expression and eosinophil infiltration were elevated displaying that VEGF and eosinophil infiltration can be controlled via STAT3.67 In another study, it has been indicated that the majority of malignant T cell patients who had positive staining for phosphor-Y705 of STAT3 (pY-STAT3), contained positive stains for eosinophils, too.68 Surprisingly, IL-5 (which mainly plays a role in the proliferation and differentiation of B cells) activates Jak2-STAT1/STAT5 and MAPK pathways for controlling genes correlated with the survival, proliferation, and effector function of eosinophils.69-71 Studies also demonstrated that in atopic dermatitis (AD), eosinophil skin infiltration needed the activation of STAT6.72
Basophil and Mast cells
Basophils, as a member of granulocytes, can release the granules and produce inflammatory cytokines like IL-4 and IL-13 while activated by endogen or exogenous stimuli such as IgE-mediated pathways.73 Mast cells, which are also called mastocytes, take part in both innate and adaptive immune systems. They are found in tissues such as skin, mucosal surfaces, and many other sites of the body. Mast cells have antibacterial and anti-parasitic functions and contribute to inflammatory disorders as they are a part of immune system cells in tissues under chronic inflammation. STAT proteins are the crucial signal transducers to activate these two types of innate immune cells.74 In associated disease with allergy, mast cell mitochondrial STAT3 is a vital factor in their degranulation. Some experimental evidence indicated that utilizing the mast cell mitochondrial STAT3 inhibitors can alleviate exocytosis and cytokine secretion due to phosphorylation of STAT3 in serine 727 and reduction of oxidative phosphorylation (OXPHOS) function. A diminish in the level of blood histamine exemplifies the importance of such inhibitors.75 It has been indicated that the STAT5 transcription factor (TF) connects to the promoter and an intronic region of the GATA-binding factor 2 (Gata)2 gene, which is essential for maintaining the Fc receptor-chain (Fcer)1a messenger ribonucleic acid(mRNA) and expression of high-affinity Fc receptor for immunoglobulin E (FcεR) Iα protein in mast cells and basophils. In mast cells, GATA2 also has the ability to maintain the expression of KIT proto-oncogene (Kit) mRNA and c-Kit protein. Furthermore, the synthesis of histamine and expression of Il4 mRNA depends on GATA2. Generally, differentiation (from pre-bone morphogenetic proteins (BMPs)) and maintenance of mast cells and basophils require the STAT5-GATA2 pathway.76 Based on relevant studies, IL-3, which is a crucial factor in mast cell and basophil biology besides the FcƐRI signaling pathway, activates phosphorylated STAT5. Furthermore, IgE-mediated mast cell activation resulting in degranulation and production of cytokines requires STAT5 activation, too.77,78
Natural killer cells
NK cells are granular lymphocytes found in most body tissues and are a part of the innate immune system. They are specified to kill microorganisms and infected cells and are capable of tumor-destructive effects. These cells perform their cytotoxic actions through the Jak/STAT pathway.79 Various cytokines including IFNs, IL-2, IL-7, IL-12, IL-15, IL-21, and IL-27 are needed for NK cell maturation and activation. These cytokines exert their functions through the downstream Jak/STAT pathway.80 STAT family members have different functions in NK cells in a way that STAT1, STAT4, and STAT5 positively influence NK cells and promote their cytotoxicity, while STAT3 and STAT6 hamper NK cell activation. IFN-γ production and NK cell cytotoxicity are controlled by STAT1.81 Here, we will discuss the main role of each STAT in NK cells. It has been revealed that MHCI expression is lowered in a STAT1-deficient manner, which affects the responsive potential of NK cells.82 STAT1 exists in immunological synapses between NK cells and target cells, including tumor cells. In this position, STAT1 can bind to different proteins and guide various processes.83 Loss of STAT3 raises CD226 (as an activating receptor), perforin, and granzyme B expression. Moreover, STAT3 is involved in stimulating the expression of NK receptor group 2, member D (NKG2D) after induction of IL-10 and IL-21.84 IL-15-activated STAT3 prohibits IL-15-induced STAT5, which is thought to mediate NK cell cytotoxicity.84 IL-12 and IFN-γ stimulate STAT4-mediated cytotoxicity in NK cells.85 STAT4 can also bind to the perforin promoter of NK cells and up-regulates IL-12 receptor (IL-12R) expression.86,87 STAT5 is the main regulator of NK cells due to its capability of controlling survival and cell growth as well as cell maturation and activation. Furthermore, STAT5 suppresses VEGF-A produced by NK cells, resulting in the inhibition of tumor progression.88 It is attractive to speculate that the STAT5B defect (despite STAT5A) is paralleled by the reduction of NK cell numbers and weakens its cytolytic responses.89 STAT5B possesses the potential of down-regulating VEGF-A expression as the promising tumor-promoting factor.90 Interestingly, activated STAT5 following IL-2 and IL-12 induction has a pro-angiogenic manner besides NK cell cytotoxicity.91,92 In NK cells, STAT6 provokes the production of IL-5 and IL-13 and restricts their associated cytotoxicity.93 Nevertheless, besides its tumor-promoting effect, STAT6 can exert a tumor-suppressive effect by elevating IFN-γ expression after IL-2 and IL-4 co-stimulation with NK cells (Fig. 3).94
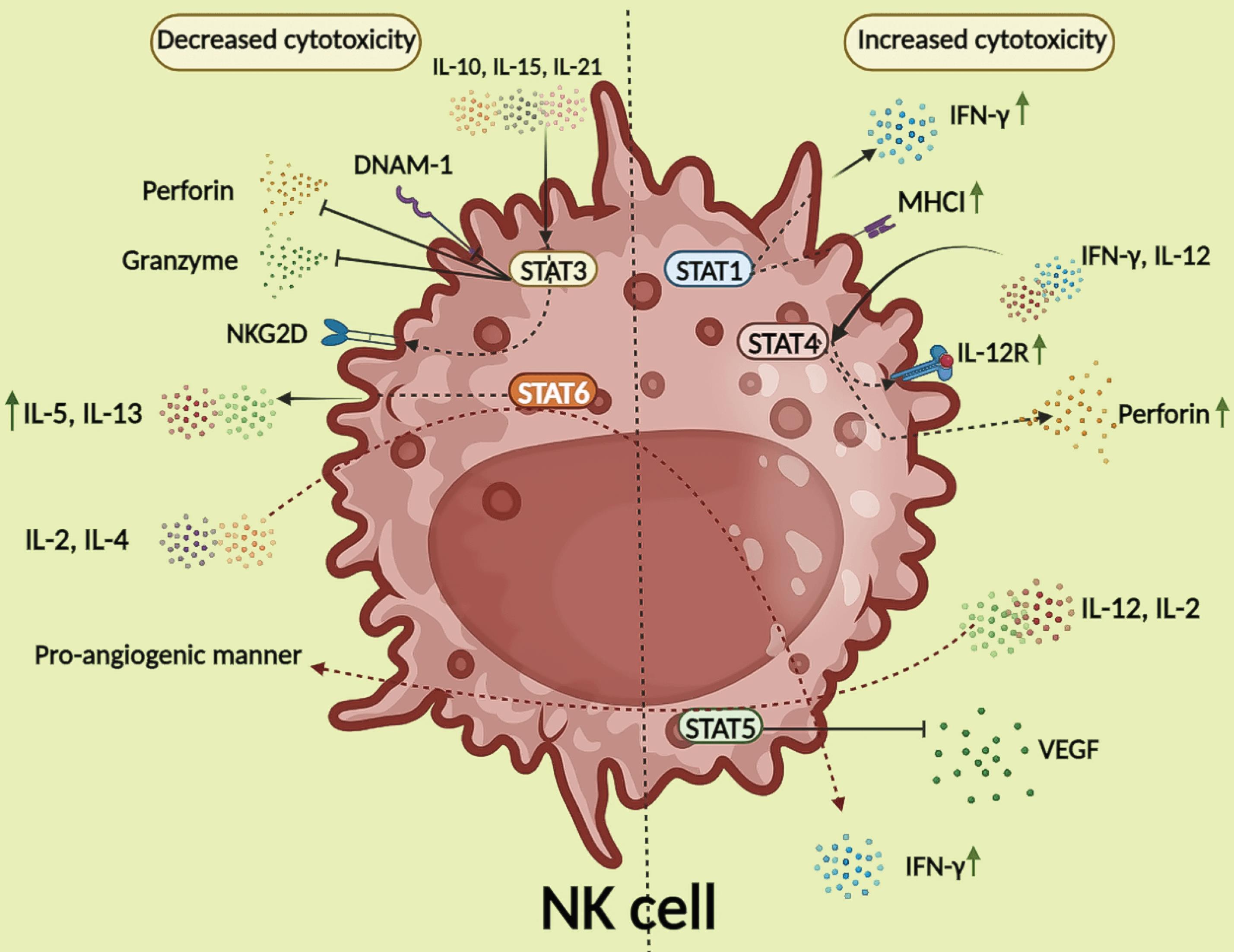

Fig. 3.
Different functions of STAT family members in NK cells. STAT1 can increase IFN-γ and MHCII production. STAT3 diminishes DNAM-1, perforin, and granzyme B expression. Moreover, STAT3 stimulates the expression of NKG2D after induction of IL-10 and IL-21. STAT4 can also bind to the perforin promoter of NK cells and up-regulate IL-12R expression. IL-12 and IFN-γ stimulate STAT4-mediated cytotoxicity in NK cells via increasing the expression of perforin IL-12R. Furthermore, STAT5 suppresses VEGF-A produced by NK cells, resulting in the inhibition of tumor progression. On the other hand, activated STAT5 following IL-2 and IL-12 induction has a pro-angiogenic manner besides NK cell cytotoxicity. In NK cells, STAT6 provokes the production of IL-5 and IL-13 and restricts their associated cytotoxicity. STAT6 can exert a tumor-suppressive effect by elevating IFN-γ expression after IL-2 and IL-4 co-stimulation with NK cells. Abbreviations: DNAM-1; DNAX accessory molecule-1; IFN-γ; interferon gamma; IL: interleukin; IL-12R: interleukin 12 receptor; MHCII: major histocompatibility complex class II; NK: natural killer cell; NKG2D; STAT: signal transducer and activator of transcription; VEGF: vascular endothelial growth factor.
.
Different functions of STAT family members in NK cells. STAT1 can increase IFN-γ and MHCII production. STAT3 diminishes DNAM-1, perforin, and granzyme B expression. Moreover, STAT3 stimulates the expression of NKG2D after induction of IL-10 and IL-21. STAT4 can also bind to the perforin promoter of NK cells and up-regulate IL-12R expression. IL-12 and IFN-γ stimulate STAT4-mediated cytotoxicity in NK cells via increasing the expression of perforin IL-12R. Furthermore, STAT5 suppresses VEGF-A produced by NK cells, resulting in the inhibition of tumor progression. On the other hand, activated STAT5 following IL-2 and IL-12 induction has a pro-angiogenic manner besides NK cell cytotoxicity. In NK cells, STAT6 provokes the production of IL-5 and IL-13 and restricts their associated cytotoxicity. STAT6 can exert a tumor-suppressive effect by elevating IFN-γ expression after IL-2 and IL-4 co-stimulation with NK cells. Abbreviations: DNAM-1; DNAX accessory molecule-1; IFN-γ; interferon gamma; IL: interleukin; IL-12R: interleukin 12 receptor; MHCII: major histocompatibility complex class II; NK: natural killer cell; NKG2D; STAT: signal transducer and activator of transcription; VEGF: vascular endothelial growth factor.
Macrophages
MQs are the famous phagocytes of the immune system. They are found at extravascular sites and react to exogenous pathogens, injured cells, and cellular debris, so they play a role in tissue injury repair. MQs receive signals from the periphery through STAT signaling pathways, which make them mature and activated.95 STAT1 activation is triggered through IFN-𝛾 leading to boost proinflammatory cytokine production. Thus, polarization to M1 MQs is induced. Moreover, iNOS and IL-12 transcription in M1 MQ is promoted by IFN-𝛾-induced STAT1.96 In MQs, IRF5, activated by proinflammatory cytokines, affects IFN-𝛾/Jak/STAT1 positively. However, IRF4, activated by anti-inflammatory cytokines, inhibits the IRF5 effect on these cells.97,98 Conversely, reprogramming of the M2 MQ phenotype occurs after IL-4, IL-13, and IL-10 stimulation and following Jak/STAT-signaling activation. After IL-4 stimulation, STAT3 and STAT6 play a role in polarization to M2 phenotype. The expression of cellular Myelocytomatosis (c-Myc) is induced to up-regulate the expression of the M2 phenotype genes, including scavengerreceptor class B type 1 (Scarb1), MQ mannose receptor (Mrc1), STAT6, and peroxisome proliferator-activated receptor gamma (PPAR-𝛾).99,100 IL-13 has the potential to activate STAT1, STAT3, and STAT6. The expression of resistin-like molecule alpha 1 (Fizz1), chitinase-like protein (Ym1), mannose receptor, and anti-inflammatory cytokines as M2 phenotype genes is enhanced through STAT3 and STAT6. In contrast, STAT1 is involved in the activation of proinflammatory cytokines. Thus, the prevalence of STAT3 and STAT6 over STAT1 activation possesses the capability to justify the anti-inflammatory response of IL-13.101 It has been revealed that the expression of the M2 phenotype genes including TGF-β and IL-10 is stimulated. Also the genes implicated in the production of M1 cytokines, including tumor necrosis factor-α (TNF-𝛼) are suppressed through IL-10 attachment to its specific receptor and subsequent Jak1 and STAT3 activation.102 SOCS1 and SOCS3 are two significant regulators of the Jak/STAT signaling pathway in M1/M2 MQs.103 M1 phenotype development is suppressed when SOCS1-induced IL-4 hampers STAT1. SOCS3 blocks STAT3 via IFN-𝛾 and toll-like receptor (TLR) 4 ligands, leading to the inhibition of M2 phenotype formation.104 Simultaneously, STAT6 (the M2 phenotype transcription factor) and STAT1 (necessary for M1 phenotype formation) are activated by SOCS1 and SOCS3, respectively.101 Other studies indicate that STAT5 signaling in MQs is activated through tumor-derived GM-CSF. STAT5 deficiency in MQs enhances expression of tumor-promoting factors and increases tumor cell migration and metastasis.105 A previous study revealed that aging leads to diminished MQ response to some signals such as IFN-γ and lipopolysaccharides (LPS). Reduced tyrosine phosphorylation of STAT1 α and β explains the reason for this phenomenon, which in turn restricts STAT1 gene expression. The mentioned mechanism explained why tumor-killing potential and nitrogen and oxygen species generation are lower in the MQ of aged people in contrast to young ones.106
Dendritic cells
DCs are the most potent cells for antigen capture and presentation and are also called APCs. DCs are divided into two major subgroups named plasmacytoid DCs (pDCs) and conventional DCs (cDCs), which have two types: cDC1 and cDC2. cDC1 is specified to activate Th1, and cDC2 activates follicular helper T (Tfh) cells. cDCs interaction with T cells is basically under the control of STAT signaling pathways.107,108 The different roles of each STAT will be further defined. IFN-I-induced STAT1 augments the activation and maturation of DCs via upregulating the expression of MHC and co-stimulatory molecules for presenting antigens to T cells effectively.109 It has been revealed that the cDC activation and their related potential for cross-presenting antigens to CD8+ T cells are enhanced after induction of TLR3, -4, -7, and -9 and subsequent STAT2 activation.110,111 About the STAT3 protein roles in DCs, it should be declared that FMS‐like tyrosine kinase three ligand (FLT3L) despite IL-32 enables STAT3 to up-regulate the expression of Pu.1 which is required for the differentiation of DCs.112 Previous studies demonstrated that STAT3 can regulate the development of pDC but not CD103+ DCs and GM-CSF/IL-4-derived monocyte-derived DC (moDCs) from bone marrow.113,114 Furthermore, the decreased expression of MHC and co-stimulatory molecules and the following inhibited T-cell induction can occur after IL-10-mediated STAT3 stimulation.115 Additionally, IL-6/IL-10-induced STAT3 results in enhanced activation of Treg and the generation of tolerogenic DCs.116,117 Fukao et al, in their study for inducible expression of STAT4 in DCs and MQs, have demonstrated that STAT4 expression can be augmented in the presence of TNF-α and IL-1β leading to moDC maturation.118 It has been suggested that GM-CSF-mediated STAT5 activation is related to enhanced differentiation of cDCs (CD103+ DCs), as well as suppression of FLT3L-dependent pDCs and CD8+ cDCs formation. In addition to suppressing the transcription factors IRF7, IRF8, and Spi-B, as well as certain receptors, including TLR9 and FLT3, necessary for the growth of pDCs, GM-CSF/STAT5 also stimulates IRF4, which is necessary for cDC development.119-121 Due to the induction of cytokine-induced SH2-containing protein (SHP2) and SOCS1, SOCS2, and SOCS3 during DC maturation, it has been shown that the STAT6 signaling pathway remains less active in mature DCs (mDCs) as opposed to immature DCs (iDCs), suggesting that DC maturation depends on STAT6 signaling pathway suppression.109 The main roles of STAT protein in each innate immune cell are indicated in Table 1.
Table 1.
The total function of STATs in the innate immune cells
|
Cell type
|
Main triggered STAT(s)
|
Total role of triggered STAT(s) in related cell
|
References
|
| CAFs |
STAT3 |
Activatory |
38,39
|
| MDSCs |
STAT1 |
Activatory |
45
|
| STAT3 |
Activatory |
47
|
| STAT5 |
Activatory |
46
|
| STAT6 |
Activatory |
54
|
| Neutrophils |
STAT1 |
Activatory |
59
|
| STAT2 |
Activatory |
60
|
| STAT3 |
Inhibitory |
58
|
| Eosinophils |
STAT1 |
Activatory |
71
|
| STAT3 |
Inhibitory |
72
|
| STAT5 |
Activatory |
71
|
| STAT6 |
Activatory |
72
|
| Basophils |
STAT5 |
Activatory |
76
|
| Mast cells |
STAT3 |
Activatory |
75
|
| STAT5 |
Activatory |
76
|
| NK cells |
STAT1 |
Activatory |
82
|
| STAT3 |
Inhibitory |
84
|
| STAT4 |
Activatory |
85
|
| STAT5 |
Activatory or inhibitory (based on conditions) |
88,91,92
|
| STAT6 |
Activatory or inhibitory (based on conditions) |
93,94
|
| M1 MQs |
STAT1 |
Activatory |
96
|
| STAT5 |
Activatory |
105
|
| M2 MQs |
STAT3 |
Activatory |
101
|
| STAT6 |
Activatory |
101
|
| DCs |
STAT1 |
Activatory |
109
|
| STAT2 |
Activatory |
110
|
| STAT3 |
Inhibitory |
112
|
| STAT4 |
Activatory |
118
|
| STAT5 |
Activatory or inhibitory (based on condition) |
119,120
|
Abbreviations: CAFs: cancer-associated fibroblasts; DCs: dendritic cell; MDSCs: myeloid-derived suppressor cells; MQs: macrophages; NKs: natural killer cells; STAT: signal transducer and activator of transcription.
The role of STATs in the adaptive immune system cells
Helper T cells
T cells are a major part of the adaptive immune system. CD4+ T cells, which are commonly known as Th cells, are the lymphocytes divided into many types like Th1, Th2, Th17, and follicular Th cells.122 The vital role of Jak and STAT in the polarization of different Th cells has been proven so that irregularities in these pathways are correlated with multiple immunological disorders.123 Not only are Jak and STAT involved in Th cell function, but we can also see other signaling molecules such as PI3K, MAPK, and ERK.124
Th1 cells
Th1 activation is intended to create a defense against intracellular infections. T-box transcription factor TBX21 (bet) is the primary TF. However, other important TFs for Th1 polarization include STAT1, which is essential for IFN-γ signaling, and STAT4, which is required for IL-12 signaling, are involved.125 In Th1 cells, IL-12 and TCR trigger Map3K8, which STAT4 binds to directly. This Map3K8 then activates downstream ERK, leading to an increase in the production of cytokines.126 Interestingly, STAT5A and STAT5B play roles in Th1 and Treg through the expression of T-bet and forkhead box P3 (FOXP3), respectively.127 Th1-particular pathways are blocked via STAT6, too. It has been recommended that Th1 can be silenced through the attachment of STAT6 to the particular site of the IL4 promoter.128
Th2 cells
Th2 cells stand against parasites and develop allergic responses. IL-4, IL-5, and IL-13 are the main products of these cells. GATA3 is known as a specific TF for Th2 cells.129 STAT6 is a factor for Th2 polarization and also inhibits Th1 polarization. Although some studies have indicated that Th2 polarization can happen even with the loss of STAT6.130 Phosphorylation of STAT6 in CD4+ CD161+ T cells is enhanced in asthmatic patients, resulting in IL-4 and IgE level augmentation, and STAT3 and STAT6 are in turn activated through IL-4 and IL-13, while in monocytes, IL-13 can affect just the activation of STAT1.131 I-Cheng Ho and coworkers in their study about essential functions before and after Th2 differentiation have displayed that differentiation of Th2 cells and B cell class switching leading to IgE production occurred after the effect of the IL-4/STAT6 axis in asthma.132 T cells with impaired STAT6 activity are incapable of decreasing the expression of the cell cycle-dependent kinase inhibitor p27KIP after IL-4 provocation, leading to hampered progression from the G1 to the S phase of the cell cycle.133 It is interesting to clarify that STAT3 can raise Th2 cytokines and survival and extend their inflammatory effects.134 However, Camille A Knosp and colleagues revealed that a lack of STAT3 activation was paralleled with increased Th2 cell responses.135 Other studies have shown that in Th2 cells, expression of T1ST2 (a member of the IL-1R family), as IL-33R, is controlled by GATA3 and STAT5. Furthermore, STAT5 activators, including IL-2, IL-7, and TSLP, stimulate the expression of GATA3. Relative findings have also demonstrated that T1ST2, expressed on Th2 cells, triggers IL-13 generation in a NF-κB and p38-dependent manner.136 Thereby, two factors are required for Th2 cell differentiation. The first is STAT3, which is required to attachment STAT6 to its target gene. Another factor is STAT5A/B, which increases the expression of GATA3 and IL-4Rα.72 Another finding revealed that STAT1 can be activated through histamine recruitment, which affects the Th2 response, and subsequent AD is observed.137
Th17 cells
Th17 cells will be activated against extracellular bacteria. RAR-related orphan receptor gamma (RORγt), as the main TF of these cells, is induced via STAT3, and both of these factors up-regulate the expression of IL-17 (Fig. 4).138 Although Th17 can play a role in inducing inflammatory conditions, it can have a suppressive effect on the immune system during cancer. Chalmin and colleagues have indicated that during Th17 cell differentiation, IL-6/ STAT3 axis and growth factor independence-1 (Gfi-1) transcription, which is down-regulated as a result of TGF-β, increased the expression of CD39 and CD73 ectonucleotidases. CD39 and CD73 ectonucleotidases, via inducing adenosine release, contribute to inhibiting the effector functions of CD4+ and CD8+ T cells in vitro, leading to the inhibitory fate of Th17 cells in the immune system.139 Some findings revealed that suppression of SOCS3 results in improved phosphorylation of STAT3 and following enhancement in IL-17 generation.140 IL-17 expression is limited through STAT5 A/B which may inhibit RORγt in Th17 cells (Fig. 4).141 Furthermore, for Th17 cell lineage differentiation, there is competition between STAT3 and STAT5 for attaching to binding sites on Il17 loci. After STAT5 attachment to the Il17 gene locus, the nuclear receptor co-repressor 2 (NCOR2) is recruited to interact with STAT5.142 In T-bet-deficient cells, the potential of hampering Th17 response via IFN-γ and IL-27 decreases while even in STAT1-deficient T cells, T-bet possesses the capability to down-regulate IL-22, RORγt, IL-17F, and IL-17A expression.143 Two studies displayed that loss of STAT4 causes diminished production of IL-17 in Th17 cells although other investigations indicated that STAT4 might not regulate the expression of IL-17, positively.144,145
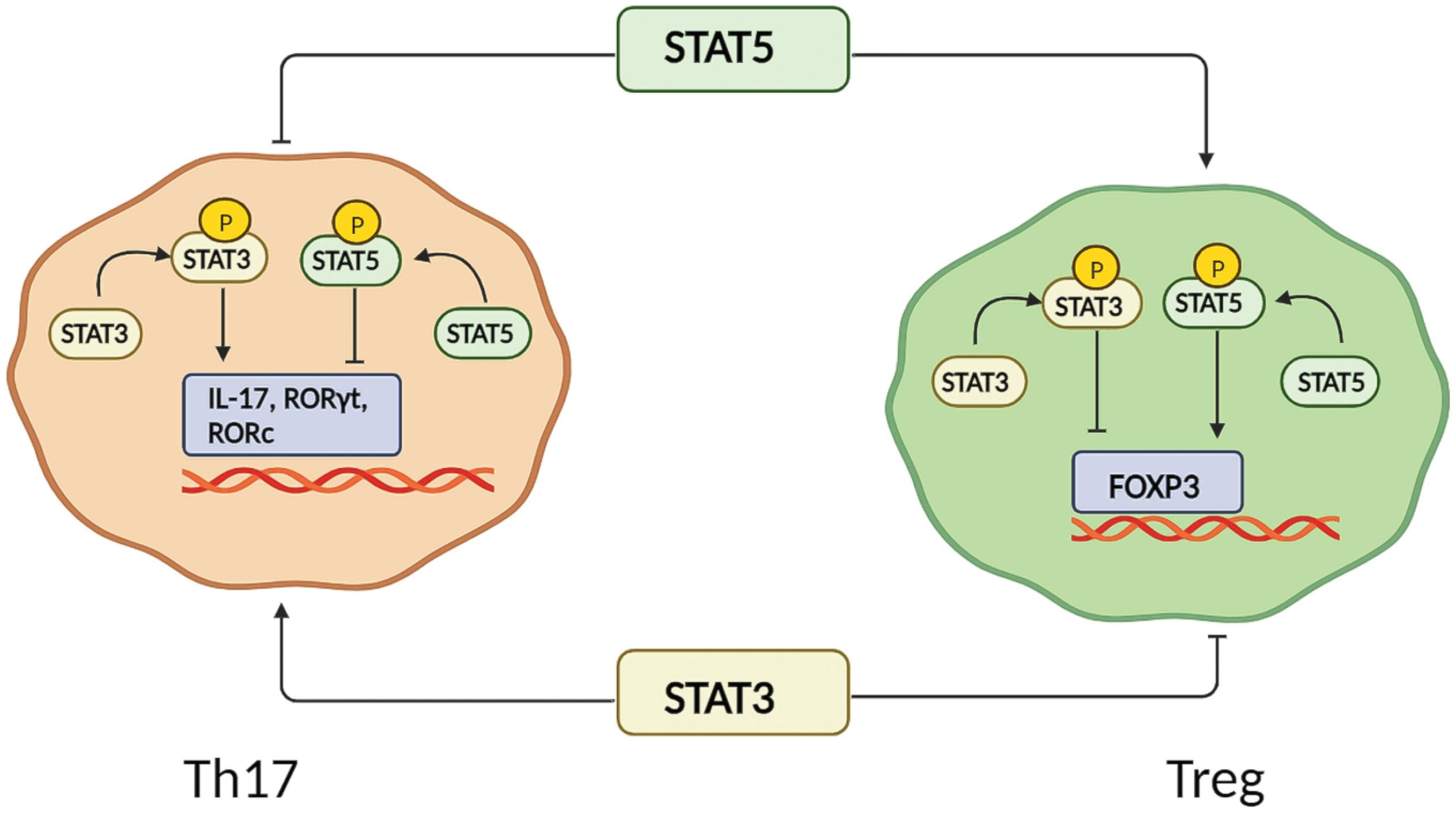
Fig. 4.
Opposed roles of STAT3 and STAT5 in Th17 and Treg cells. STAT5 induces FOXP3 expression in Treg cells while down-regulate the expression of IL-17, RORc, and RORγt in Th17 cells. STAT3 acts totally in an opposed manner in contrast to STAT5 in the mentioned cells. Abbreviations: FOXP3: forkhead box P3; IL: interleukin; STAT: signal transducer and activator of transcription; Th: T helper; ROR: RAR-related orphan receptor.
.
Opposed roles of STAT3 and STAT5 in Th17 and Treg cells. STAT5 induces FOXP3 expression in Treg cells while down-regulate the expression of IL-17, RORc, and RORγt in Th17 cells. STAT3 acts totally in an opposed manner in contrast to STAT5 in the mentioned cells. Abbreviations: FOXP3: forkhead box P3; IL: interleukin; STAT: signal transducer and activator of transcription; Th: T helper; ROR: RAR-related orphan receptor.
Follicular helper T cells
Tfh cells are a subset of Th cells. They adjust and coordinate B cells maturation and activity and, subsequently, the humoral immune responses. They are resident in germinal centers (GCs) of secondary lymphoid tissues. There are different subsets of Tfh consisting of Tfh1, Tfh2, Tfh3, and T follicular regulatory (Tfr). Activation of STAT signaling pathways induces Tfh cell differentiation and activation that are further defined with more details in each subset.146,147 In acute viral infections, IFNs and IL-6-activated STAT1 cause induction of the Bcl-6 expression and trigger Tfh1 cell differentiation at an early stage.148 T-bet is also a critical factor in Tfh1 expansion and maintenance.149 STAT4 activated by IL-12 may contribute to the differentiation of Tfh1 cells, so a lack of STAT4 can lead to diminished production of IL-21 and IFN-γ from Tfh1 cells.150 Generally, some extra signals besides STAT1/4 are required for Tfh1 differentiation.146 Further investigations demonstrate that IL-2-STAT5 and IL-2R expression is hampered by IL-4 in Tfh2 differentiation unlike the Th2 differentiation process.151 Furthermore, Tfh2 differentiation is improved by IL-4-STAT6. Other findings revealed thatin Tfh2 cells, cooperation of basic leucine zipper ATF-like transcription factor (BATF), STAT3, and STAT6 results in enhanced production of IL-4.152 Whereas, STAT3 can play a suppressive role at the early stage of differentiation of Tfh2 cells.153 It has been indicated thatin the process of TFH formation, STAT3 might not be effective alone. TGF-β besides cytokines that activate STAT3/STAT4 (IL23/IL12), can affect Tfh17 differentiation positively through increasing the expression of IL-21, C-X-C chemokine receptor type 5 (CXCR5), and Bcl-6.154 Bcl-6 expression is suppressed through the IL-2/STAT5 axis which can impair Tfh differentiation.155 Based on investigations, the concentration-dependent manner of IL-2 is considered to guide Tfh, Treg, and Tfr differentiation. Tfh cells can be converted to Tfr-like cells during exposure to IL-2. Furthermore, IL-2-induced STAT3 develops the expression of FOXP3 and maintains the expression of Bcl-6 as well.155,156 Thus, rather than being entirely distinct inhibitory agents, IL-2-STAT5 should be viewed as a controller of Tfr differentiation.156
Regulatory T (Treg) cells
Another subset of T lymphocytes consists of the regulatory T cells called Treg cells. Treg has immunomodulatory function and tolerance to self-antigens by their special cytokine production and signaling, which results in the suppressive impact of these cells on autoimmunity. There are complicated signaling pathways, such as STATs for Treg cell differentiation and activation.157 FOXP3 is the main TF in Treg cells.129 The maintenance of Foxp3 expression relies on the conserved non-coding sequence 2 (CNS2) region which contains binding sites for STAT5. Demethylation of this region occurs via STAT5, leading to the conversion of naïve CD4+ FOXP3 - T cells into CD4+ FOXP3+ induced Treg (iTreg)s (Fig. 4).158 The promoter of the FOXP3 gene is a location to which STAT5 can bind. Thus, this STAT5 can help Treg differentiation by regulating FOXP3 expression.159 STAT5 probably inhibits RORc thus STAT5A and STAT5B are known as factors by which Th17 differentiation is regulated, negatively (Fig. 4).141 Any impairment in members of the IL-2/CD122/Jak3/STAT-5 signaling pathway causes thymic and peripheral Treg reduction.141 Moreover, on iTregs, the expression of the IL-6 receptor is diminished by IL-2 to inhibit iTregs from differentiating to Th17 cells.160 It has been indicated that for controlling iTreg development, STAT5 and STAT3 signaling should be balanced manner.142 IL-4-dependent STAT6 and IL-12-dependent STAT4 block Treg development through decreasing STAT5 attachment to the promoter and/or CNS2 region of the Foxp3 gene.161 Furthermore, IL-6/STAT3 suppresses the expression of FOXP3.162 Based on investigations, homeostasis and inhibitory function of Tregs are controlled by the IFN-α signaling pathway, and further phosphorylation of STAT5 and STAT1.163
Cytotoxic T lymphocytes
CTLs are the differentiated form of CD8+ T cells and capable of recognizing virus-infected cells and cancer cells to induce cytotoxicity against them by releasing the contents of the granules, which encompass lytic agents, also called lytic granules (LG).164 It has been shown that induction of CTL is restricted after STAT1 deficiency, and this event has been decreased more in the case of mutation in the S727 phospho-receptor site.165 Furthermore, multiple findings explained that S727-phosphorylated STAT1 is an indispensable factor in DC activation. This information illustrates that S727-phosphorylated-STAT1 is required for DC maturation, which in turn stimulates CTL activation and is also used for direct control of T cells.166 Yu et al have demonstrated that loss of STAT3 in CD8+ T cells can increase their cell division.167 Improved proliferation in CD8+ T cells with STAT3 deficiency manner results from p21waf1 (cyclin-dependent kinase (CDK) inhibitor), p27KIP1 (cyclin-dependent kinase (CDK) inhibitor), forkhead box protein O1 (FoxO1), and FoxO3A downregulation. In contrast to CD4+ T cells, CD8+ T cells are more impressible to STAT3 growth repressive effects. Interestingly, some studies indicated that IFN-𝛾 production is improved through STAT3 pathways, but exact triggered mechanisms have not been considered yet. Regulatory CD8+ T cells, which produce IL-10, are hampered by STAT3, too.167,168 An investigation revealed that STAT3 induced by IL-10 and IL-21 can lead to memory CD8+ T cell differentiation and do not let them convert to effector ones. Activated CD8+ T cell initial expansion can be trigger by STAT3 recruitment.168 According to J Z Quin and coworkers' findings, for maintaining effector CD8+ T cells, the expression of Bcl-2 is stimulated via STAT5 after IL-7 and IL-15 induction.169 Although some other reports have demonstrated that STAT5 is not required for induction of Bcl-2 expression.170,171
B cells
B cells are the bone marrow-derived lymphocytes and the arm of the humoral immune system with the ability of Ig production, which can release proinflammatory cytokines and present antigens to T cells. Overall, they have a key role in the activation of T cells and substantially, initiation of inflammation in the immune system. B and T lymphocytes are interlinked in various aspects.172 These complex functions by B cells need to be worked out by unique signaling pathways, and STATs are the important ones. It has been shown that Src family kinases such as Lyn start BCR engagement, and multiple following signaling cascades are begun for proliferation, differentiation, and even restriction of B cell growth.173 Correlation between CD19 and BCR could enhance STAT1 tyrosine phosphorylation compared to BCR lonely cross-linking. Preceding events help us to consider CD19 as a positive modulator for STAT activation after BCR engagement.174 Signaling guided by STAT1 and IFN-γ receptor (IFN-γR) is a pivotal factor in spontaneously developed germinal centers (GCs [Spt-GCs]) and the formation of Tfh cells. Moreover, IFN-γ-R and STAT1 signaling regulate Spt-GC and Tfh cell development by inducing B cells to express T-bet and produce IFN-γ.175 Some findings have recommended that gain of function mutation in STAT1 leads to deficiency in B-cell differentiation.176 Moreover, in the bone marrow and marginal zone, STAT1 can improve B-cell differentiation in mice.177,178 For regulation of B-cell response and differentiation, STAT3 is activated through IL-21.179 Moreover, the number of antigen-specific memory B cells is decreased and the formation of plasma cells from naive B cells is inhibited after IL-21 induction due to STAT3 mutations. A defect in the molecular mechanism, which is needed for plasma cell (PC) production, is what led to the previous incident. Whereas IL-21-stimulated Ig secretion and memory B cell formation are unaffected by STAT1 impairment.180 Differently from mutated-STAT3, mutated-STAT1 after IL-21 induction decreases the expression of the paired box (PAX5) and enhances the expression of PR domain zinc finger protein 1 (PRDM1) and X-box binding protein 1 (XBP)in contrast to normal naive B cells, indicating the unessential role of STAT1 for this process. IL-6-induced STAT3 can mediate the function of B cells by improving antibody secretion from human PCs through augmenting IL-6R expression.181,182 Uri Rozovski and colleagues in their study revealed that in chronic lymphocytic leukemia cells, B-cell receptor stimulation has the potential of activating the Jak2/STAT3 signaling pathway.183 The prominent roles of STAT protein in each adaptive immune cell are summarized in Table2.
Table 2.
The total roles of main STATs in the adaptive immune cells
|
Cell type
|
Main triggered STAT(s)
|
Total roles of triggered STAT(s) in related cell
|
References
|
| Th1 |
STAT1 |
Activatory |
125
|
| STAT4 |
Activatory |
125
|
| STAT5 |
Activatory |
127
|
| STAT6 |
Inhibitory |
128
|
| Th2 |
STAT1 |
Activatory |
137
|
| STAT3 |
Activatory or inhibitory (based on condition) |
134,135
|
| STAT5 |
Activatory |
136
|
| STAT6 |
Activatory |
130
|
| Th17 |
STAT3 |
Activatory |
138
|
| STAT4 |
Activatory or inhibitory (based on condition) |
144,145
|
| STAT5 |
Inhibitory |
141
|
| TFHs |
Tfh1 |
STAT1 |
Activatory |
146
|
| STAT4 |
Activatory |
146
|
| Tfh2 |
STAT3 |
Activatory |
152
|
| STAT5 |
Inhibitory |
151
|
| STAT6 |
Activatory |
152
|
| Tfh17 |
STAT3 |
Activatory |
154
|
| STAT4 |
Activatory |
154
|
| Tfr |
STAT5 |
Activatory |
156
|
| Tregs |
STAT1 |
Activatory |
163
|
| STAT3 |
Inhibitory |
162
|
| STAT4 |
Inhibitory |
161
|
| STAT5 |
Activatory |
159
|
| STAT6 |
Inhibitory |
161
|
| CTLs |
STAT1 |
Activatory |
165
|
| STAT3 |
Activatory or inhibitory (based on condition) |
167
|
| STAT5 |
Not clear |
169-171
|
| B cells |
STAT1 |
Activatory |
175
|
| STAT3 |
Inhibitory |
183
|
Abbreviations: CTL: cytotoxic T lymphocytes; Th: helper T cells; STAT: signal transducer and activator of transcription; TFH: follicular helper T; Tfr: T follicular regulatory cells; Treg: regulatory T.
The inhibitory and activatory roles of main triggered STATs in different innate and adaptive immune cells have been demonstrated (Fig. 5).
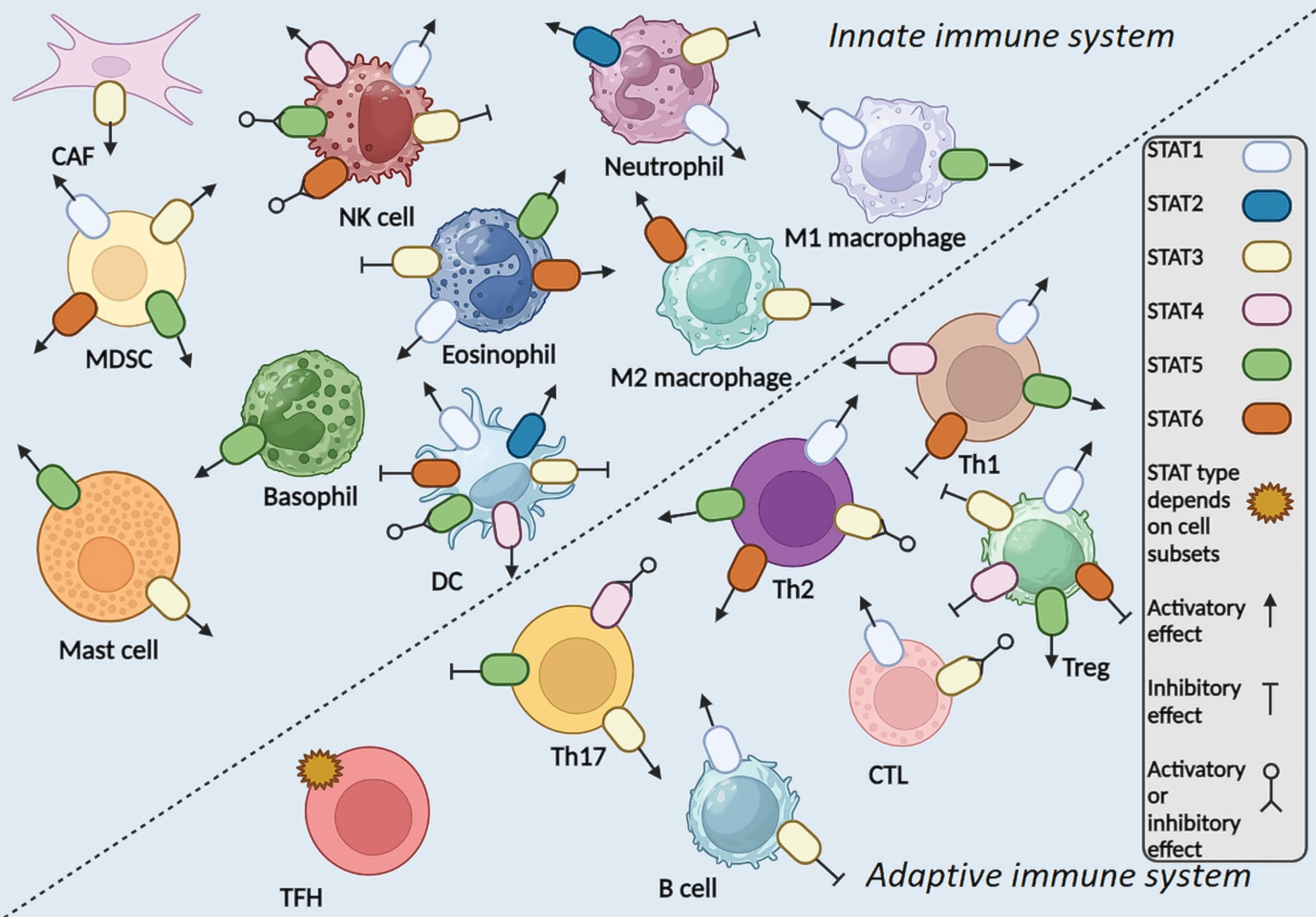
Fig. 5.
Inhibitory or activatory roles of different STATs in innate and adaptive immune cells. Abbreviations: CAF: cancer-associated fibroblast; CTL: cytotoxic T lymphocyte; DC: dendritic cell; MDSC: myeloid-derived suppressor cells; NK: natural killer cell; STAT: signal transducer and activator of transcription; TFH: follicular helper T cell; Th: helper T cell; Treg: regulatory T.
.
Inhibitory or activatory roles of different STATs in innate and adaptive immune cells. Abbreviations: CAF: cancer-associated fibroblast; CTL: cytotoxic T lymphocyte; DC: dendritic cell; MDSC: myeloid-derived suppressor cells; NK: natural killer cell; STAT: signal transducer and activator of transcription; TFH: follicular helper T cell; Th: helper T cell; Treg: regulatory T.
A perspective on the different roles of STATs in cancer
STAT role in the innate immune system in cancer
CAFs and MDSCs, as members of the TME, provide a place for tumor proliferation and metastasis. As discussed before, CAF and MDSCs have a main signaling pathway as the regulator of the cells' function, which is called STAT3. Targeting this TF may cause the favorite results for cancer immunotherapy by adjustment of the mentioned cells' function.172,184 According to investigations, IL-6 binds to its receptor IL-6Ra on the cell surface of neutrophils, hepatocytes, monocytes, and B and T cells, activating Jak1, which stimulates the STAT3.185 This signaling promotes growth and prevents apoptosis, including the activation of genes encoding Bcl-xl and Bcl-2.186 STAT3 is thought to be a negative modulator of inflammatory reactions. Moreover, mice lacking the STAT3 gene in neutrophils show increased inflammatory activity.187 It is shown that eosinophils may have double-edged effects on cancer. Some studies resulted in the association of eosinophils with good prognosis in breast cancer cases.188 However, some studies pointed out that Jak3/STAT3 and eosinophils had cooperative attitudes in T cell-induced malignancies. These data show that this signaling pathway can be considered the appropriate target for immunotherapy planning.188 Treg-produced IL-13 has been proven to induce basophil activation through STAT5, which in turn contributes to Th2 response. Previous events cause tumor promotion, thereby either hampering the basophil activation or the IL-3/IL-3R axis, resulting in induced anti-tumor effects.189,190 The tumor microenvironment's cytokines influence NK-cell development and function. Many cytokines communicate through the Jak/STAT pathway, which is constitutively active in a wide range of tumor cells.191 STAT1 plays a vital role in IFN-γ expression and NK-cell cytotoxicity.192 Conversely, STAT3 activity is thought to impede tumor immune surveillance, allowing the cancer to evade the immune system.193 Even STAT3 activation in NK cells decreases cytotoxicity, according to animal studies. So, STAT3 deletion in NK cells increased cytotoxicity in leukemia and melanoma models and resulted in longer survival.194 STAT5 inhibits NK-cell tumor-promoting activity. NK cells, like myeloid cells, have the ability to promote tumor growth by secreting VEGF-A. STAT5 inhibits VEGF-A expression and tumor promotion, with STAT5B being the critical isoform.195 It has been demonstrated that the tumor-inducing factor cyclooxygenase-2 (COX-2) is expressed more often in MQs following STAT3 ablation and Jak/STAT suppression and that blocking COX-2 improves tumors' sensitivity to ruxolitinib.196 Another study has revealed that arachidonate 5-lipoxygenase (ALOX5), which encodes 5-lipoxygenase stimulates, metastases and invasion in pancreatic cancer via controlling TAM polarization via the Jak/STAT pathway. Zileuton blocks ALOX5 to prevent these outcomes.197 Studies have demonstrated that the accumulation of STAT3 precludes the differentiation of immature DCs to mature ones and increases the impacts of tumor-derived factors on the differentiation of myeloid cells in cancer. Moreover, being able to recognize CD28 receptors on T cells, the level of expression of intracellular IFN- γ, and the suppression of the generation of the cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) in breast cancer lymph nodes are all improved as a result of enhanced CD86 expression on DCs after STAT3 blocking.198
STAT role in the adaptive immune system in cancer
Th cells are capable of the anti-cancer immune response, as seen in CD8+ T cells. It is observed that Th1 has the potential to limit angiogenesis and metastasis and to activate the apoptotic processes in cancer cells by IFN-γ production, which is induced by the expression of T-bet TF, which in turn becomes activated through STAT1 and STAT4 activatory signaling pathways.199 Chimeric antigen receptor (CAR) T cells can get Th1 phenotype and function. Subsequently, they can be used in T cell therapy for several types of malignancies.200 Th2 cell action through STAT5 and STAT6 is more related to malignant cells' progression in comparison to anti-cancer function. Thus, the application of Th2 in T cell therapy has some limitations, whereas Th2 anti-tumor roles through innate immune cells are seen in pancreatic and colon cancer.201,202 Recent research mentioned STAT3's role in stem cells and cancer stem cell control. One study intended to determine the position of activated STAT3 in gastric cancer stem cells (GCSCs) and the Th17/Treg cell paradigm. The results show that gastro-spheroids, as a model of GCSCs, had higher levels of STAT3 activity, increased levels of VEGF, TGF-β, and lower levels of IL-6. Treatment with stattic small molecules prevents STAT3 activation in cancer cells, which lowers stemness properties. For instance, stemness gene expression, in vivo tumorigenicity potential, and TGF-β expression are all down-regulated in the cancer cells. Additionally, co-culture of STAT3-inhibited cancer cells with normal peripheral blood mononuclear cells (PBMCs) results in a decrease in the proportion of Treg cells and an increase in Th17 cells.203 It is well-recognized that IL-17 is an angiogenic agent and may enhance metastasis and tumor growth via STAT-3 signaling and other mechanisms.204 Research about the STAT role in Tfh in cancer has displayed that the contribution of STAT3, STAT6, and BATF results in augmented production of IL-4 that can exert a tumor-suppressing effect. Furthermore, in Tfh2 cells, it has been reported that the anti-tumor effect of IL-21 resulting from STAT4 induction can be increased in combination with anti-CTLA-4 or anti- programmed death-1 (PD-1) immunotherapy.205 STAT is a crucial signaling pathway that Tregs activate to support angiogenesis, metastasis, immunosuppression, and apoptosis resistance in cancer cells. The TME's immunosuppression is facilitated by STAT3 and STAT5.206 Tregs interact with a variety of cells, including M2 cells, through STAT signaling. For example, the development of monocytes into M2 cells is mediated by Tregs through STAT3 activation, secretion of IL-10, and VEGF generation. In turn, M2 cells release IL‐6207 and IL‐10208 for increasing Treg activity within the TME.209 It has been revealed that Ruxolitinib, a Jak/STAT inhibitor, promotes CTL infiltration in the tumor microenvironment in the pancreatic cancer mice model by selectively inhibiting STAT1 and STAT3 activation. The suppression of STAT3 increases T cell activation and effector function by decreasing the generation of immune suppressive cytokines by tumor cells. Ruxolitinib afterward dramatically enhances the effectiveness of anti-PD-1 immunotherapy to circumvent pancreatic cancer resistance.210 According to Viganò and colleagues’ investigations about primary mediastinal large B-cell lymphoma (PMBCL), the gain of function mutation in IL-4R results in constitutive activation of the Jak/STAT pathway and further increase in the expression of downstream cytokine and B cell-specific antigens, leading to the development of lymphomagenesis. STAT6-induced IL-4 and IL-13 lead to differentiation and activation of B cells, resulting in establishing tumor suppression.211
STAT3 therapeutic potentials
As previously stated, STAT3 is a transcription factor in the cell cytoplasm that regulates multiple signaling pathways in the immune system, especially those related to malignancies.26 This member of the STAT protein family causes a reduction in anti-tumor responses, which are usually made by NK cells, Treg cells, CD8+ T cells, and DCs.212 STAT3 is known to suppress MHCΙΙ and expression of CD86, IL-12, CXC motif chemokine ligand (CXCL)10, and CCL5, which overall causes the impedance of maturation and antigen-presentation in DCs that is followed by the debilitation of Th1 and CTL aggregation and anti-cancer immune responses.213,214 STAT3 activation in MQs has a similar effect to DC activation and its further impacts on T cells.215 In addition, STAT3 activators in bone marrow progenitor cells lead to neutrophil inhibition. The immunosuppression that happens through STAT3 has a notable impact on protecting tumor cells by providing a perfect TME and promoting their growth.214 In parallel, STAT3 induces the direct expression of cancer-promoting genes. Recent research has indicated that STAT3 activation is associated with a poor prognosis in patients with solid tumors.216 These results suggest that targeting the STAT3 signaling pathway in different directions can be a promising therapeutic option for various malignancies. STAT3 is found to play a role in the growth, invasion, promotion, and metastasis of cancerous cells, such as breast cancer, hepatocellular carcinoma (HCC), non-small cell lung cancer (NSCLC), glioblastoma, and colorectal cancers.217,218 Hampering STAT3 by the special agents is utilized in the control of diseases in the context of inflammation or infection, diabetes, osteoporosis, and even psychotic disorders next to malignancies.26,219,220 Some drugs are STAT3 inhibitors with completed clinical trials and benefits in terms of safety, tolerability, and sufficient anti-tumor effects. STAT3 inhibitor agents can have a negative influence on tumor growth, suppress angiogenesis, and reduce the probability of tumor metastasis.221,222 Small molecules, peptides, oligonucleotides (with direct impact), phytochemical agents, and Chimeric antigen receptor redirected T cells (CAR T-cells) are examples of a class of drugs able to inhibit STAT3 activation.220 AZD9150 works as an inhibitor of the STAT3-related gene and has been shown to impact numerous solid malignancies, such as HCC. BIOMOL, as a small-molecule inhibitor, works directly on STAT3.223 Napabucasin is an example with the small molecule and possesses the capability to suppress the STAT3 mRNA formation from the related gene, which has been involved in clinical trials for treating malignancies such as pancreatic adenocarcinoma, colorectal carcinoma, and malignant pleural mesothelioma.224 Curcumin and even some antibiotics such as nifuroxazide and atovaquone are drugs with the purpose of preventing STAT3 from becoming phosphorylated.225,226 Furthermore, it is shown that restriction of STAT3 function elevates the sensitivity of tumor cells to radiotherapy and chemotherapy.227,228
Concluding remarks
Undoubtedly, STAT transcription factors influence diverse immune cell subsets’ differentiation, proliferation, and function. Even numerous studies have demonstrated the therapeutic potential of their targeting in immune system disorders. Controlled and balanced activation of these cells requires correlation between various signaling pathways, and one of the most significant pathways employed in the majority of immune cells is the Jak/STAT signaling pathway. As we reviewed, different STAT factors play meaningful roles in diverse innate and adaptive immune cell subsets, and subsequently, they show their impacts to overcome immune-related disorders or act in favor of disease progress like tumor growth. For instance, a reduction in longevity in patients with colorectal cancer can be associated with STAT3 phosphorylation in CAFs, and even STAT3 can lead to tumor progression via augmenting MDSC involvement. Also, in allergy, STAT signaling pathways are necessary for the eosinophil's function and survival because Jak/STAT leads to the postponement of eosinophil apoptosis. About mast cells, mitochondrial STAT3 is a vital factor in their degranulation. NK cells that perform cytotoxic actions, act through Jak/ STAT: STAT1, STAT4, and STAT5 positively influence NK cells and promote their cytotoxicity, while STAT3 and STAT6 hamper NK cell activation. About MQs, STAT1 activation is triggered through IFN-𝛾, leading to boost proinflammatory cytokine production. Thus, polarization to M1 MQs occurs, but on the other hand, after IL-4 stimulation, STAT3 and STAT6 play a role in polarization to M2 phenotype. DCs that are very important for antigen presentation require STAT1 to up-regulate the expression of MHC and co-stimulatory molecules for their effective performance, but STAT3 decreases the expression of these mentioned molecules. Irregularities in Jak/STAT pathways in T cells are correlated with multiple immunological disorders. For example, Th1 activation, which is needed to create a defense against intracellular infections, requires STAT1 for IFN-γ signaling and STAT4 for IL-12 signaling. For protection against parasites, Th2 cells are required, so STAT6 should perform Th2 polarization. Against extracellular bacteria, Th17 cells will be activated, and these cells are induced via STAT3, which even up-regulates the expression of IL-17. For immunomodulatory function and tolerance to self-antigens, Tregs’ appropriate functions are required, but any impairment in the STAT-5 signaling pathway causes a reduction in these Treg numbers. Finally, for regulation of B-cell response and differentiation, STAT3 is activated through IL-21. In conclusion, targeting each of these STAT factors with paying precise attention to its specific role in each immune cell subset may cause the favorite results for immune system disorders such as cancer immunotherapy by adjusting the mentioned cells' function.
Review Highlights
What is the current knowledge?
-
One of the significant mechanisms involved in the immune cells’ activation is the Jack/STAT signaling pathway.
-
Jack/STAT signaling pathway alteration causes various illnesses, including cancer.
What is new here?
-
Determining the inhibitory, activatory, or both roles of STATs in modulating innate and adaptive immune cells.
-
Investigation of alteration of specific STAT in immune cells leading to tumor progression or inhibition.
Acknowledgments
This study was supported by the Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. Figures were created using the BioRender online tool (Free version).
Competing Interests
The authors declare that there is no conflict of interest.
Ethical Statement
Not applicable.
References
- Meier-Schellersheim M, Varma R, Angermann BR. Mechanistic models of cellular signaling, cytokine crosstalk, and cell-cell communication in immunology. Front immunl 2019; 10:2268. [ Google Scholar]
- Nicholson LB. The immune system. Essays Biochem 2016; 60:275-301. doi: 10.1042/ebc20160017 [Crossref] [ Google Scholar]
- Lanier LL. NK cell recognition. Annu Rev Immunol 2005; 23:225-74. doi: 10.1146/annurev.immunol.23.021704.115526 [Crossref] [ Google Scholar]
- Kurosaki T, Shinohara H, Baba Y. B cell signaling and fate decision. Annu Rev Immunol 2010; 28:21-55. doi: 10.1146/annurev.immunol.021908.132541 [Crossref] [ Google Scholar]
- Smith-Garvin JE, Koretzky GA, Jordan MS. T cell activation. Annu Rev Immunol 2009; 27:591-619. doi: 10.1146/annurev.immunol.021908.132706 [Crossref] [ Google Scholar]
- Delgoffe GM, Murray PJ, Vignali DA. Interpreting mixed signals: the cell's cytokine conundrum. Curr Opin Immunol 2011; 23:632-8. doi: 10.1016/j.coi.2011.07.013 [Crossref] [ Google Scholar]
- Morel PA, Lee REC, Faeder JR. Demystifying the cytokine network: Mathematical models point the way. Cytokine 2017; 98:115-23. doi: 10.1016/j.cyto.2016.11.013 [Crossref] [ Google Scholar]
- Leppkes M, Siegmund B, Becker C. Editorial: Immune-Epithelial Crosstalk in Inflammatory Bowel Diseases and Mucosal Wound Healing. Front Immunol 2018; 9:1171. doi: 10.3389/fimmu.2018.01171 [Crossref] [ Google Scholar]
- Shuai K, Stark GR, Kerr IM, Darnell JE, Jr Jr. A single phosphotyrosine residue of Stat91 required for gene activation by interferon-gamma. Science 1993; 261:1744-6. doi: 10.1126/science.7690989 [Crossref] [ Google Scholar]
- Darnell JE, Jr Jr. Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 1994; 264:1415-21. doi: 10.1126/science.8197455 [Crossref] [ Google Scholar]
- Awasthi N, Liongue C, Ward AC. STAT proteins: a kaleidoscope of canonical and non-canonical functions in immunity and cancer. J Hematol Oncol 2021; 14:198. doi: 10.1186/s13045-021-01214-y [Crossref] [ Google Scholar]
- Kisseleva T, Bhattacharya S, Braunstein J, Schindler CW. Signaling through the JAK/STAT pathway, recent advances and future challenges. Gene 2002; 285:1-24. doi: 10.1016/s0378-1119(02)00398-0 [Crossref] [ Google Scholar]
- Copeland NG, Gilbert DJ, Schindler C, Zhong Z, Wen Z, Darnell JE, Jr Jr. Distribution of the mammalian Stat gene family in mouse chromosomes. Genomics 1995; 29:225-8. doi: 10.1006/geno.1995.1235 [Crossref] [ Google Scholar]
- Heim MH. The STAT protein family. In: Signal Transducers and Activators of Transcription (STATs) Activation and Biology. Springer; 2003. p. 11-26.
- Miklossy G, Hilliard TS, Turkson J. Therapeutic modulators of STAT signalling for human diseases. Nat Rev Drug Discov 2013; 12:611-29. doi: 10.1038/nrd4088 [Crossref] [ Google Scholar]
- Strehlow I, Schindler C. Amino-terminal signal transducer and activator of transcription (STAT) domains regulate nuclear translocation and STAT deactivation. J Biol Chem 1998; 273:28049-56. doi: 10.1074/jbc.273.43.28049 [Crossref] [ Google Scholar]
- Lim CP, Cao X. Structure, function, and regulation of STAT proteins. Mol Biosyst 2006; 2:536-50. doi: 10.1039/b606246f [Crossref] [ Google Scholar]
- Zhu M, John S, Berg M, Leonard WJ. Functional association of Nmi with Stat5 and Stat1 in IL-2- and IFNgamma-mediated signaling. Cell 1999; 96:121-30. doi: 10.1016/s0092-8674(00)80965-4 [Crossref] [ Google Scholar]
- Liu KD, Gaffen SL, Goldsmith MA. JAK/STAT signaling by cytokine receptors. Curr Opin Immunol 1998; 10:271-8. doi: 10.1016/s0952-7915(98)80165-9 [Crossref] [ Google Scholar]
- Paulson M, Press C, Smith E, Tanese N, Levy DE. IFN-Stimulated transcription through a TBP-free acetyltransferase complex escapes viral shutoff. Nat Cell Biol 2002; 4:140-7. doi: 10.1038/ncb747 [Crossref] [ Google Scholar]
- Guanizo AC, Fernando CD, Garama DJ, Gough DJ. STAT3: a multifaceted oncoprotein. Growth Factors 2018; 36:1-14. doi: 10.1080/08977194.2018.1473393 [Crossref] [ Google Scholar]
- Tzeng HT, Chyuan IT, Lai JH. Targeting the JAK-STAT pathway in autoimmune diseases and cancers: A focus on molecular mechanisms and therapeutic potential. Biochem Pharmacol 2021; 193:114760. doi: 10.1016/j.bcp.2021.114760 [Crossref] [ Google Scholar]
- Winthrop KL. The emerging safety profile of JAK inhibitors in rheumatic disease. Nat Rev Rheumatol 2017; 13:234-43. doi: 10.1038/nrrheum.2017.23 [Crossref] [ Google Scholar]
- Ivashkiv LB. Jak-STAT signaling pathways in cells of the immune system. Rev Immunogenet 2000; 2:220-30. [ Google Scholar]
- Leonard WJ, O'Shea JJ. Jaks and STATs: biological implications. Annu Rev Immunol 1998; 16:293-322. doi: 10.1146/annurev.immunol.16.1.293 [Crossref] [ Google Scholar]
- Johnson DE, O'Keefe RA, Grandis JR. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol 2018; 15:234-48. doi: 10.1038/nrclinonc.2018.8 [Crossref] [ Google Scholar]
- Bharadwaj U, Kasembeli MM, Robinson P, Tweardy DJ. Targeting Janus Kinases and Signal Transducer and Activator of Transcription 3 to Treat Inflammation, Fibrosis, and Cancer: Rationale, Progress, and Caution. Pharmacol Rev 2020; 72:486-526. doi: 10.1124/pr.119.018440 [Crossref] [ Google Scholar]
- Roca Suarez AA, Van Renne N, Baumert TF, Lupberger J. Viral manipulation of STAT3: Evade, exploit, and injure. PLoS Pathog 2018; 14:e1006839. doi: 10.1371/journal.ppat.1006839 [Crossref] [ Google Scholar]
- Maritano D, Sugrue ML, Tininini S, Dewilde S, Strobl B, Fu X. The STAT3 isoforms alpha and beta have unique and specific functions. Nat Immunol 2004; 5:401-9. doi: 10.1038/ni1052 [Crossref] [ Google Scholar]
- Zhong Z, Wen Z, Darnell JE, Jr Jr. Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science 1994; 264:95-8. doi: 10.1126/science.8140422 [Crossref] [ Google Scholar]
- Heinrich PC, Behrmann I, Müller-Newen G, Schaper F, Graeve L. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem J 1998; 334:297-314. doi: 10.1042/bj3340297 [Crossref] [ Google Scholar]
- Zhang M, Zhou L, Xu Y, Yang M, Xu Y, Komaniecki GP. A STAT3 palmitoylation cycle promotes T(H)17 differentiation and colitis. Nature 2020; 586:434-9. doi: 10.1038/s41586-020-2799-2 [Crossref] [ Google Scholar]
- Ostrand-Rosenberg S, Sinha P. Myeloid-derived suppressor cells: linking inflammation and cancer. J Immunol 2009; 182:4499-506. doi: 10.4049/jimmunol.0802740 [Crossref] [ Google Scholar]
- Catlett-Falcone R, Landowski TH, Oshiro MM, Turkson J, Levitzki A, Savino R. Constitutive activation of Stat3 signaling confers resistance to apoptosis in human U266 myeloma cells. Immunity 1999; 10:105-15. doi: 10.1016/s1074-7613(00)80011-4 [Crossref] [ Google Scholar]
- Yang L, Shi P, Zhao G, Xu J, Peng W, Zhang J. Targeting cancer stem cell pathways for cancer therapy. Signal Transduct Target Ther 2020; 5:8. doi: 10.1038/s41392-020-0110-5 [Crossref] [ Google Scholar]
- Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer 2020; 20:174-86. doi: 10.1038/s41568-019-0238-1 [Crossref] [ Google Scholar]
- Heichler C, Scheibe K, Schmied A, Geppert CI, Schmid B, Wirtz S. STAT3 activation through IL-6/IL-11 in cancer-associated fibroblasts promotes colorectal tumour development and correlates with poor prognosis. Gut 2020; 69:1269-82. doi: 10.1136/gutjnl-2019-319200 [Crossref] [ Google Scholar]
- Albrengues J, Bertero T, Grasset E, Bonan S, Maiel M, Bourget I. Epigenetic switch drives the conversion of fibroblasts into proinvasive cancer-associated fibroblasts. Nat Commun 2015; 6:10204. doi: 10.1038/ncomms10204 [Crossref] [ Google Scholar]
- Yang X, Lin Y, Shi Y, Li B, Liu W, Yin W. FAP Promotes Immunosuppression by Cancer-Associated Fibroblasts in the Tumor Microenvironment via STAT3-CCL2 Signaling. Cancer Res 2016; 76:4124-35. doi: 10.1158/0008-5472.can-15-2973 [Crossref] [ Google Scholar]
- Huang H, Zhang Y, Gallegos V, Sorrelle N, Zaid MM, Toombs J. Targeting TGFβR2-mutant tumors exposes vulnerabilities to stromal TGFβ blockade in pancreatic cancer. EMBO Mol Med 2019; 11:e10515. doi: 10.15252/emmm.201910515 [Crossref] [ Google Scholar]
- Ham IH, Oh HJ, Jin H, Bae CA, Jeon SM, Choi KS. Targeting interleukin-6 as a strategy to overcome stroma-induced resistance to chemotherapy in gastric cancer. Mol Cancer 2019; 18:68. doi: 10.1186/s12943-019-0972-8 [Crossref] [ Google Scholar]
- Cheng JT, Deng YN, Yi HM, Wang GY, Fu BS, Chen WJ. Hepatic carcinoma-associated fibroblasts induce IDO-producing regulatory dendritic cells through IL-6-mediated STAT3 activation. Oncogenesis 2016; 5:e198. doi: 10.1038/oncsis.2016.7 [Crossref] [ Google Scholar]
- Gabrilovich DI, Bronte V, Chen SH, Colombo MP, Ochoa A, Ostrand-Rosenberg S, Schreiber H. The terminology issue for myeloid-derived suppressor cells. Cancer Res 2007; 67:425; author reply 6. doi: 10.1158/0008-5472.can-06-3037 [Crossref] [ Google Scholar]
- Dorhoi A, Kotzé LA, Berzofsky JA, Sui Y, Gabrilovich DI, Garg A. Therapies for tuberculosis and AIDS: myeloid-derived suppressor cells in focus. J Clin Invest 2020; 130:2789-99. doi: 10.1172/jci136288 [Crossref] [ Google Scholar]
- Bronte V, Serafini P, De Santo C, Marigo I, Tosello V, Mazzoni A. IL-4-induced arginase 1 suppresses alloreactive T cells in tumor-bearing mice. J Immunol 2003; 170:270-8. doi: 10.4049/jimmunol.170.1.270 [Crossref] [ Google Scholar]
- Waight JD, Netherby C, Hensen ML, Miller A, Hu Q, Liu S. Myeloid-derived suppressor cell development is regulated by a STAT/IRF-8 axis. J Clin Invest 2013; 123:4464-78. doi: 10.1172/jci68189 [Crossref] [ Google Scholar]
- Sinha P, Okoro C, Foell D, Freeze HH, Ostrand-Rosenberg S, Srikrishna G. Proinflammatory S100 proteins regulate the accumulation of myeloid-derived suppressor cells. J Immunol 2008; 181:4666-75. doi: 10.4049/jimmunol.181.7.4666 [Crossref] [ Google Scholar]
- Cheng P, Corzo CA, Luetteke N, Yu B, Nagaraj S, Bui MM. Inhibition of dendritic cell differentiation and accumulation of myeloid-derived suppressor cells in cancer is regulated by S100A9 protein. J Exp Med 2008; 205:2235-49. doi: 10.1084/jem.20080132 [Crossref] [ Google Scholar]
- Mohammadpour H, MacDonald CR, Qiao G, Chen M, Dong B, Hylander BL. β2 adrenergic receptor-mediated signaling regulates the immunosuppressive potential of myeloid-derived suppressor cells. J Clin Invest 2019; 129:5537-52. doi: 10.1172/jci129502 [Crossref] [ Google Scholar]
- Lin Q, Ren L, Jian M, Xu P, Li J, Zheng P. The mechanism of the premetastatic niche facilitating colorectal cancer liver metastasis generated from myeloid-derived suppressor cells induced by the S1PR1-STAT3 signaling pathway. Cell Death Dis 2019; 10:693. doi: 10.1038/s41419-019-1922-5 [Crossref] [ Google Scholar]
- Qu P, Du H, Wang X, Yan C. Matrix metalloproteinase 12 overexpression in lung epithelial cells plays a key role in emphysema to lung bronchioalveolar adenocarcinoma transition. Cancer Res 2009; 69:7252-61. doi: 10.1158/0008-5472.can-09-0577 [Crossref] [ Google Scholar]
- Zhang CX, Ye SB, Ni JJ, Cai TT, Liu YN, Huang DJ. STING signaling remodels the tumor microenvironment by antagonizing myeloid-derived suppressor cell expansion. Cell Death Differ 2019; 26:2314-28. doi: 10.1038/s41418-019-0302-0 [Crossref] [ Google Scholar]
- Pei J, Zhang Y, Luo Q, Zheng W, Li W, Zeng X. STAT3 inhibition enhances CDN-induced STING signaling and antitumor immunity. Cancer Lett 2019; 450:110-22. doi: 10.1016/j.canlet.2019.02.029 [Crossref] [ Google Scholar]
- Sinha P, Clements VK, Ostrand-Rosenberg S. Reduction of myeloid-derived suppressor cells and induction of M1 macrophages facilitate the rejection of established metastatic disease. J Immunol 2005; 174:636-45. doi: 10.4049/jimmunol.174.2.636 [Crossref] [ Google Scholar]
- Rosales C. Neutrophil: A Cell with Many Roles in Inflammation or Several Cell Types?. Front Physiol 2018; 9:13. doi: 10.3389/fphys.2018.00113 [Crossref] [ Google Scholar]
- Scheel-Toellner D, Wang K, Henriquez NV, Webb PR, Craddock R, Pilling D. Cytokine-mediated inhibition of apoptosis in non-transformed T cells and neutrophils can be dissociated from protein kinase B activation. Eur J Immunol 2002; 32:486-93. doi: 10.1002/1521-4141(200202)32:2<486::aidimmu486>3.0.co;2-u [Crossref] [ Google Scholar]
- Ellis TN, Beaman BL. Interferon-gamma activation of polymorphonuclear neutrophil function. Immunology 2004; 112:2-12. doi: 10.1111/j.1365-2567.2004.01849.x [Crossref] [ Google Scholar]
- Fielding CA, McLoughlin RM, McLeod L, Colmont CS, Najdovska M, Grail D. IL-6 regulates neutrophil trafficking during acute inflammation via STAT3. J Immunol 2008; 181:2189-95. doi: 10.4049/jimmunol.181.3.2189 [Crossref] [ Google Scholar]
- Cai W, Wang J, Hu M, Chen X, Lu Z, Bellanti JA, Zheng SG. All trans-retinoic acid protects against acute ischemic stroke by modulating neutrophil functions through STAT1 signaling. J Neuroinflammation 2019; 16:175. doi: 10.1186/s12974-019-1557-6 [Crossref] [ Google Scholar]
- Shaul ME, Levy L, Sun J, Mishalian I, Singhal S, Kapoor V. Tumor-associated neutrophils display a distinct N1 profile following TGFβ modulation: A transcriptomics analysis of pro- vsantitumor TANs. Oncoimmunology 2016; 5:e1232221. doi: 10.1080/2162402x.2016.1232221 [Crossref] [ Google Scholar]
- Korzus E, Nagase H, Rydell R, Travis J. The mitogen-activated protein kinase and JAK-STAT signaling pathways are required for an oncostatin M-responsive element-mediated activation of matrix metalloproteinase 1 gene expression. J Biol Chem 1997; 272:1188-96. doi: 10.1074/jbc.272.2.1188 [Crossref] [ Google Scholar]
- Simonneau M, Frouin E, Huguier V, Jermidi C, Jégou JF, Godet J. Oncostatin M is overexpressed in skin squamous-cell carcinoma and promotes tumor progression. Oncotarget 2018; 9:36457-73. doi: 10.18632/oncotarget.26355 [Crossref] [ Google Scholar]
- Queen MM, Ryan RE, Holzer RG, Keller-Peck CR, Jorcyk CL. Breast cancer cells stimulate neutrophils to produce oncostatin M: potential implications for tumor progression. Cancer Res 2005; 65:8896-904. doi: 10.1158/0008-5472.can-05-1734 [Crossref] [ Google Scholar]
- Gigon L, Fettrelet T, Yousefi S, Simon D, Simon HU. Eosinophils from A to Z. Allergy 2023; 78:1810-46. doi: 10.1111/all.15751 [Crossref] [ Google Scholar]
- Cheung PF, Wong CK, Ho AW, Hu S, Chen DP, Lam CW. Activation of human eosinophils and epidermal keratinocytes by Th2 cytokine IL-31: implication for the immunopathogenesis of atopic dermatitis. Int Immunol 2010; 22:453-67. doi: 10.1093/intimm/dxq027 [Crossref] [ Google Scholar]
- Cornelissen C, Lüscher-Firzlaff J, Baron JM, Lüscher B. Signaling by IL-31 and functional consequences. Eur J Cell Biol 2012; 91:552-66. doi: 10.1016/j.ejcb.2011.07.006 [Crossref] [ Google Scholar]
- Cao Q, Zhang T, Zhang J. Correlation analysis of STAT3 and VEGF expression and eosinophil infiltration in nasal polyps. Eur Arch Otorhinolaryngol 2015; 272:1955-60. doi: 10.1007/s00405-014-3290-1 [Crossref] [ Google Scholar]
- Nakashima C, Ishida Y, Kitoh A, Otsuka A, Kabashima K. Interaction of peripheral nerves and mast cells, eosinophils, and basophils in the development of pruritus. Exp Dermatol 2019; 28:1405-11. doi: 10.1111/exd.14014 [Crossref] [ Google Scholar]
- Takatsu K, Nakajima H. IL-5 and eosinophilia. Curr Opin Immunol 2008; 20:288-94. doi: 10.1016/j.coi.2008.04.001 [Crossref] [ Google Scholar]
- Kotsimbos AT, Hamid Q. IL-5 and IL-5 receptor in asthma. Mem Inst Oswaldo Cruz 1997; 92 Suppl 2:75-91. doi: 10.1590/s0074-02761997000800012 [Crossref] [ Google Scholar]
- Ochiai K, Tanabe E, Ishihara C, Kagami M, Sugiyama T, Sueishi M. Role of JAK2 signal transductional pathway in activation and survival of human peripheral eosinophils by interferon-gamma (IFN-gamma). Clin Exp Immunol 1999; 118:340-3. doi: 10.1046/j.1365-2249.1999.01068.x [Crossref] [ Google Scholar]
- Bao L, Zhang H, Chan LS. The involvement of the JAK-STAT signaling pathway in chronic inflammatory skin disease atopic dermatitis. Jakstat 2013; 2:e24137. doi: 10.4161/jkst.24137 [Crossref] [ Google Scholar]
- Pellefigues C, Mehta P, Chappell S, Yumnam B, Old S, Camberis M, Le Gros G. Diverse innate stimuli activate basophils through pathways involving Syk and IκB kinases. Proc Natl Acad Sci U S A 2021; 118. 10.1073/pnas.2019524118.
- Wang JX, Kaieda S, Ameri S, Fishgal N, Dwyer D, Dellinger A. IL-33/ST2 axis promotes mast cell survival via BCLXL. Proc Natl Acad Sci U S A 2014; 111:10281-6. doi: 10.1073/pnas.1404182111 [Crossref] [ Google Scholar]
- Erlich TH, Sharkia I, Landolina N, Assayag M, Goldberger O, Berkman N. Modulation of allergic responses by mitochondrial STAT3 inhibitors. Allergy 2018; 73:2160-71. doi: 10.1111/all.13467 [Crossref] [ Google Scholar]
- Li Y, Qi X, Liu B, Huang H. The STAT5-GATA2 pathway is critical in basophil and mast cell differentiation and maintenance. J Immunol 2015; 194:4328-38. doi: 10.4049/jimmunol.1500018 [Crossref] [ Google Scholar]
- Rodrigues NP, Boyd AS, Fugazza C, May GE, Guo Y, Tipping AJ. GATA-2 regulates granulocyte-macrophage progenitor cell function. Blood 2008; 112:4862-73. doi: 10.1182/blood-2008-01-136564 [Crossref] [ Google Scholar]
- Tsai FY, Orkin SH. Transcription factor GATA-2 is required for proliferation/survival of early hematopoietic cells and mast cell formation, but not for erythroid and myeloid terminal differentiation. Blood 1997; 89:3636-43. [ Google Scholar]
- Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat Immunol 2008; 9:503-10. doi: 10.1038/ni1582 [Crossref] [ Google Scholar]
- Gotthardt D, Trifinopoulos J, Sexl V, Putz EM. JAK/STAT Cytokine Signaling at the Crossroad of NK Cell Development and Maturation. Front Immunol 2019; 10:2590. doi: 10.3389/fimmu.2019.02590 [Crossref] [ Google Scholar]
- Au-Yeung N, Mandhana R, Horvath CM. Transcriptional regulation by STAT1 and STAT2 in the interferon JAK-STAT pathway. Jakstat 2013; 2:e23931. doi: 10.4161/jkst.23931 [Crossref] [ Google Scholar]
- Robbins SH, Tessmer MS, Van Kaer L, Brossay L. Direct effects of T-bet and MHC class I expression, but not STAT1, on peripheral NK cell maturation. Eur J Immunol 2005; 35:757-65. doi: 10.1002/eji.200425797 [Crossref] [ Google Scholar]
- Putz EM, Majoros A, Gotthardt D, Prchal-Murphy M, Zebedin-Brandl EM, Fux DA. Novel non-canonical role of STAT1 in Natural Killer cell cytotoxicity. Oncoimmunology 2016; 5:e1186314. doi: 10.1080/2162402x.2016.1186314 [Crossref] [ Google Scholar]
- Gotthardt D, Putz EM, Straka E, Kudweis P, Biaggio M, Poli V. Loss of STAT3 in murine NK cells enhances NK cell-dependent tumor surveillance. Blood 2014; 124:2370-9. doi: 10.1182/blood-2014-03-564450 [Crossref] [ Google Scholar]
- Townsend MJ, Weinmann AS, Matsuda JL, Salomon R, Farnham PJ, Biron CA. T-bet regulates the terminal maturation and homeostasis of NK and Valpha14i NKT cells. Immunity 2004; 20:477-94. doi: 10.1016/s1074-7613(04)00076-7 [Crossref] [ Google Scholar]
- Wang KS, Ritz J, Frank DA. IL-2 induces STAT4 activation in primary NK cells and NK cell lines, but not in T cells. J Immunol 1999; 162:299-304. [ Google Scholar]
- Wang KS, Frank DA, Ritz J. Interleukin-2 enhances the response of natural killer cells to interleukin-12 through up-regulation of the interleukin-12 receptor and STAT4. Blood 2000; 95:3183-90. [ Google Scholar]
- Gotthardt D, Sexl V. STATs in NK-Cells: The Good, the Bad, and the Ugly. Front Immunol 2016; 7:694. doi: 10.3389/fimmu.2016.00694 [Crossref] [ Google Scholar]
- Imada K, Bloom ET, Nakajima H, Horvath-Arcidiacono JA, Udy GB, Davey HW, Leonard WJ. Stat5b is essential for natural killer cell-mediated proliferation and cytolytic activity. J Exp Med 1998; 188:2067-74. doi: 10.1084/jem.188.11.2067 [Crossref] [ Google Scholar]
- Gotthardt D, Putz EM, Grundschober E, Prchal-Murphy M, Straka E, Kudweis P. STAT5 Is a Key Regulator in NK Cells and Acts as a Molecular Switch from Tumor Surveillance to Tumor Promotion. Cancer Discov 2016; 6:414-29. doi: 10.1158/2159-8290.cd-15-0732 [Crossref] [ Google Scholar]
- Hanna J, Goldman-Wohl D, Hamani Y, Avraham I, Greenfield C, Natanson-Yaron S. Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat Med 2006; 12:1065-74. doi: 10.1038/nm1452 [Crossref] [ Google Scholar]
- Fraser R, Whitley GS, Johnstone AP, Host AJ, Sebire NJ, Thilaganathan B, Cartwright JE. Impaired decidual natural killer cell regulation of vascular remodelling in early human pregnancies with high uterine artery resistance. J Pathol 2012; 228:322-32. doi: 10.1002/path.4057 [Crossref] [ Google Scholar]
- Katsumoto T, Kimura M, Yamashita M, Hosokawa H, Hashimoto K, Hasegawa A. STAT6-dependent differentiation and production of IL-5 and IL-13 in murine NK2 cells. J Immunol 2004; 173:4967-75. doi: 10.4049/jimmunol.173.8.4967 [Crossref] [ Google Scholar]
- Bream JH, Curiel RE, Yu CR, Egwuagu CE, Grusby MJ, Aune TM, Young HA. IL-4 synergistically enhances both IL-2- and IL-12-induced IFN-gamma expression in murine NK cells. Blood 2003; 102:207-14. doi: 10.1182/blood-2002-08-2602 [Crossref] [ Google Scholar]
- Watanabe S, Alexander M, Misharin AV, Budinger GRS. The role of macrophages in the resolution of inflammation. J Clin Invest 2019; 129:2619-28. doi: 10.1172/jci124615 [Crossref] [ Google Scholar]
- Yeh CH, Shih HC, Hong HM, Lee SS, Yang ML, Chen CJ, Kuan YH. Protective effect of wogonin on proinflammatory cytokine generation via Jak1/3-STAT1/3 pathway in lipopolysaccharide stimulated BV2 microglial cells. Toxicol Ind Health 2015; 31:960-6. doi: 10.1177/0748233713485886 [Crossref] [ Google Scholar]
- Satoh T, Kidoya H, Naito H, Yamamoto M, Takemura N, Nakagawa K. Critical role of Trib1 in differentiation of tissue-resident M2-like macrophages. Nature 2013; 495:524-8. doi: 10.1038/nature11930 [Crossref] [ Google Scholar]
- Eguchi J, Kong X, Tenta M, Wang X, Kang S, Rosen ED. Interferon regulatory factor 4 regulates obesity-induced inflammation through regulation of adipose tissue macrophage polarization. Diabetes 2013; 62:3394-403. doi: 10.2337/db12-1327 [Crossref] [ Google Scholar]
- Bhattacharjee A, Shukla M, Yakubenko VP, Mulya A, Kundu S, Cathcart MK. IL-4 and IL-13 employ discrete signaling pathways for target gene expression in alternatively activated monocytes/macrophages. Free Radic Biol Med 2013; 54:1-16. doi: 10.1016/j.freeradbiomed.2012.10.553 [Crossref] [ Google Scholar]
- Pello OM, De Pizzol M, Mirolo M, Soucek L, Zammataro L, Amabile A. Role of c-MYC in alternative activation of human macrophages and tumor-associated macrophage biology. Blood 2012; 119:411-21. doi: 10.1182/blood-2011-02-339911 [Crossref] [ Google Scholar]
- Malyshev I, Malyshev Y. Current Concept and Update of the Macrophage Plasticity Concept: Intracellular Mechanisms of Reprogramming and M3 Macrophage "Switch" Phenotype. Biomed Res Int 2015; 2015:341308. doi: 10.1155/2015/341308 [Crossref] [ Google Scholar]
- Luwor RB, Baradaran B, Taylor LE, Iaria J, Nheu TV, Amiry N. Targeting Stat3 and Smad7 to restore TGF-β cytostatic regulation of tumor cells in vitro and in vivo. Oncogene 2013; 32:2433-41. doi: 10.1038/onc.2012.260 [Crossref] [ Google Scholar]
- Croker BA, Kiu H, Nicholson SE. SOCS regulation of the JAK/STAT signalling pathway. Semin Cell Dev Biol 2008; 19:414-22. doi: 10.1016/j.semcdb.2008.07.010 [Crossref] [ Google Scholar]
- Qin H, Holdbrooks AT, Liu Y, Reynolds SL, Yanagisawa LL, Benveniste EN. SOCS3 deficiency promotes M1 macrophage polarization and inflammation. J Immunol 2012; 189:3439-48. doi: 10.4049/jimmunol.1201168 [Crossref] [ Google Scholar]
- Jesser EA, Brady NJ, Huggins DN, Witschen PM, O'Connor CH, Schwertfeger KL. STAT5 is activated in macrophages by breast cancer cell-derived factors and regulates macrophage function in the tumor microenvironment. Breast Cancer Res 2021; 23:104. doi: 10.1186/s13058-021-01481-0 [Crossref] [ Google Scholar]
- Yoon P, Keylock KT, Hartman ME, Freund GG, Woods JA. Macrophage hypo-responsiveness to interferon-gamma in aged mice is associated with impaired signaling through Jak-STAT. Mech Ageing Dev 2004; 125:137-43. doi: 10.1016/j.mad.2003.11.010 [Crossref] [ Google Scholar]
- Steinman RM, Hemmi H. Dendritic cells: translating innate to adaptive immunity. Curr Top Microbiol Immunol 2006; 311:17-58. doi: 10.1007/3-540-32636-7_2 [Crossref] [ Google Scholar]
- Guilliams M, Ginhoux F, Jakubzick C, Naik SH, Onai N, Schraml BU. Dendritic cells, monocytes and macrophages: a unified nomenclature based on ontogeny. Nat Rev Immunol 2014; 14:571-8. doi: 10.1038/nri3712 [Crossref] [ Google Scholar]
- Jackson SH, Yu CR, Mahdi RM, Ebong S, Egwuagu CE. Dendritic cell maturation requires STAT1 and is under feedback regulation by suppressors of cytokine signaling. J Immunol 2004; 172:2307-15. doi: 10.4049/jimmunol.172.4.2307 [Crossref] [ Google Scholar]
- Qiu CC, Kotredes KP, Cremers T, Patel S, Afanassiev A, Slifker M. Targeted Stat2 deletion in conventional dendritic cells impairs CTL responses but does not affect antibody production. Oncoimmunology 2020; 10:1860477. doi: 10.1080/2162402x.2020.1860477 [Crossref] [ Google Scholar]
- Xu J, Lee MH, Chakhtoura M, Green BL, Kotredes KP, Chain RW. STAT2 Is Required for TLR-Induced Murine Dendritic Cell Activation and Cross-Presentation. J Immunol 2016; 197:326-36. doi: 10.4049/jimmunol.1500152 [Crossref] [ Google Scholar]
- Hahm B, Trifilo MJ, Zuniga EI, Oldstone MB. Viruses evade the immune system through type I interferon-mediated STAT2-dependent, but STAT1-independent, signaling. Immunity 2005; 22:247-57. doi: 10.1016/j.immuni.2005.01.005 [Crossref] [ Google Scholar]
- Park SJ, Nakagawa T, Kitamura H, Atsumi T, Kamon H, Sawa S. IL-6 regulates in vivo dendritic cell differentiation through STAT3 activation. J Immunol 2004; 173:3844-54. doi: 10.4049/jimmunol.173.6.3844 [Crossref] [ Google Scholar]
- Laouar Y, Welte T, Fu XY, Flavell RA. STAT3 is required for Flt3L-dependent dendritic cell differentiation. Immunity 2003; 19:903-12. doi: 10.1016/s1074-7613(03)00332-7 [Crossref] [ Google Scholar]
- Gharibi T, Barpour N, Hosseini A, Mohammadzadeh A, Marofi F, Ebrahimi-Kalan A. STA-21, a small molecule STAT3 inhibitor, ameliorates experimental autoimmune encephalomyelitis by altering Th-17/Treg balance. Int Immunopharmacol 2023; 119:110160. doi: 10.1016/j.intimp.2023.110160 [Crossref] [ Google Scholar]
- Masjedi A, Ahmadi A, Atyabi F, Farhadi S, Irandoust M, Khazaei-Poul Y. Silencing of IL-6 and STAT3 by siRNA loaded hyaluronate-N,N,N-trimethyl chitosan nanoparticles potently reduces cancer cell progression. Int J Biol Macromol 2020; 149:487-500. doi: 10.1016/j.ijbiomac.2020.01.273 [Crossref] [ Google Scholar]
- Safarzadeh E, Mohammadi A, Mansoori B, Duijf PHG, Hashemzadeh S, Khaze V. STAT3 Silencing and TLR7/8 Pathway Activation Repolarize and Suppress Myeloid-Derived Suppressor Cells From Breast Cancer Patients. Front Immunol 2020; 11:613215. doi: 10.3389/fimmu.2020.613215 [Crossref] [ Google Scholar]
- Fukao T, Frucht DM, Yap G, Gadina M, O'Shea JJ, Koyasu S. Inducible expression of Stat4 in dendritic cells and macrophages and its critical role in innate and adaptive immune responses. J Immunol 2001; 166:4446-55. doi: 10.4049/jimmunol.166.7.4446 [Crossref] [ Google Scholar]
- Tamura T, Tailor P, Yamaoka K, Kong HJ, Tsujimura H, O'Shea JJ. IFN regulatory factor-4 and -8 govern dendritic cell subset development and their functional diversity. J Immunol 2005; 174:2573-81. doi: 10.4049/jimmunol.174.5.2573 [Crossref] [ Google Scholar]
- Shortman K, Naik SH. Steady-state and inflammatory dendritic-cell development. Nat Rev Immunol 2007; 7:19-30. doi: 10.1038/nri1996 [Crossref] [ Google Scholar]
- Zenke M, Hieronymus T. Towards an understanding of the transcription factor network of dendritic cell development. Trends Immunol 2006; 27:140-5. doi: 10.1016/j.it.2005.12.007 [Crossref] [ Google Scholar]
- Wan YY. Multi-tasking of helper T cells. Immunology 2010; 130:166-71. doi: 10.1111/j.1365-2567.2010.03289.x [Crossref] [ Google Scholar]
- Seif F, Khoshmirsafa M, Aazami H, Mohsenzadegan M, Sedighi G, Bahar M. The role of JAK-STAT signaling pathway and its regulators in the fate of T helper cells. Cell Commun Signal 2017; 15:23. doi: 10.1186/s12964-017-0177-y [Crossref] [ Google Scholar]
- Li WX. Canonical and non-canonical JAK-STAT signaling. Trends Cell Biol 2008; 18:545-51. doi: 10.1016/j.tcb.2008.08.008 [Crossref] [ Google Scholar]
- Mullen AC, High FA, Hutchins AS, Lee HW, Villarino AV, Livingston DM. Role of T-bet in commitment of TH1 cells before IL-12-dependent selection. Science 2001; 292:1907-10. doi: 10.1126/science.1059835 [Crossref] [ Google Scholar]
- Dagur PK, Sharma B, Kumar G, Khan NA, Katoch VM, Sengupta U, Joshi B. Mycobacterial antigen(s) induce anergy by altering TCR- and TCR/CD28-induced signalling events: insights into T-cell unresponsiveness in leprosy. Mol Immunol 2010; 47:943-52. doi: 10.1016/j.molimm.2009.11.009 [Crossref] [ Google Scholar]
- Zhang L, Li YG, Li YH, Qi L, Liu XG, Yuan CZ. Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis. PLoS One 2012; 7:e31000. doi: 10.1371/journal.pone.0031000 [Crossref] [ Google Scholar]
- Curiel RE, Lahesmaa R, Subleski J, Cippitelli M, Kirken RA, Young HA, Ghosh P. Identification of a Stat-6-responsive element in the promoter of the human interleukin-4 gene. Eur J Immunol 1997; 27:1982-7. doi: 10.1002/eji.1830270823 [Crossref] [ Google Scholar]
- Zhou L, Chong MM, Littman DR. Plasticity of CD4 + T cell lineage differentiation. Immunity 2009; 30:646-55. doi: 10.1016/j.immuni.2009.05.001 [Crossref] [ Google Scholar]
- van Panhuys N, Tang SC, Prout M, Camberis M, Scarlett D, Roberts J. In vivo studies fail to reveal a role for IL-4 or STAT6 signaling in Th2 lymphocyte differentiation. Proc Natl Acad Sci U S A 2008; 105:12423-8. doi: 10.1073/pnas.0806372105 [Crossref] [ Google Scholar]
- Gernez Y, Tirouvanziam R, Nguyen KD, Herzenberg LA, Krensky AM, Nadeau KC. Altered phosphorylated signal transducer and activator of transcription profile of CD4 + CD161 + T cells in asthma: modulation by allergic status and oral corticosteroids. J Allergy Clin Immunol 2007; 120:1441-8. doi: 10.1016/j.jaci.2007.08.012 [Crossref] [ Google Scholar]
- Ho IC, Tai TS, Pai SY. GATA3 and the T-cell lineage: essential functions before and after T-helper-2-cell differentiation. Nat Rev Immunol 2009; 9:125-35. doi: 10.1038/nri2476 [Crossref] [ Google Scholar]
- Zhu J, Guo L, Watson CJ, Hu-Li J, Paul WE. Stat6 is necessary and sufficient for IL-4's role in Th2 differentiation and cell expansion. J Immunol 2001; 166:7276-81. doi: 10.4049/jimmunol.166.12.7276 [Crossref] [ Google Scholar]
- Alexander WS. Suppressors of cytokine signalling (SOCS) in the immune system. Nat Rev Immunol 2002; 2:410-6. doi: 10.1038/nri818 [Crossref] [ Google Scholar]
- Knosp CA, Carroll HP, Elliott J, Saunders SP, Nel HJ, Amu S. SOCS2 regulates T helper type 2 differentiation and the generation of type 2 allergic responses. J Exp Med 2011; 208:1523-31. doi: 10.1084/jem.20101167 [Crossref] [ Google Scholar]
- Guo L, Wei G, Zhu J, Liao W, Leonard WJ, Zhao K, Paul W. IL-1 family members and STAT activators induce cytokine production by Th2, Th17, and Th1 cells. Proc Natl Acad Sci U S A 2009; 106:13463-8. doi: 10.1073/pnas.0906988106 [Crossref] [ Google Scholar]
- Horr B, Borck H, Thurmond R, Grösch S, Diel F. STAT1 phosphorylation and cleavage is regulated by the histamine (H4) receptor in human atopic and non-atopic lymphocytes. Int Immunopharmacol 2006; 6:1577-85. doi: 10.1016/j.intimp.2006.06.005 [Crossref] [ Google Scholar]
- Chen Z, Laurence A, O'Shea JJ. Signal transduction pathways and transcriptional regulation in the control of Th17 differentiation. Semin Immunol 2007; 19:400-8. doi: 10.1016/j.smim.2007.10.015 [Crossref] [ Google Scholar]
- Chalmin F, Mignot G, Bruchard M, Chevriaux A, Végran F, Hichami A. Stat3 and Gfi-1 transcription factors control Th17 cell immunosuppressive activity via the regulation of ectonucleotidase expression. Immunity 2012; 36:362-73. doi: 10.1016/j.immuni.2011.12.019 [Crossref] [ Google Scholar]
- Wong PK, Egan PJ, Croker BA, O'Donnell K, Sims NA, Drake S. SOCS-3 negatively regulates innate and adaptive immune mechanisms in acute IL-1-dependent inflammatory arthritis. J Clin Invest 2006; 116:1571-81. doi: 10.1172/jci25660 [Crossref] [ Google Scholar]
- Laurence A, Tato CM, Davidson TS, Kanno Y, Chen Z, Yao Z. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 2007; 26:371-81. doi: 10.1016/j.immuni.2007.02.009 [Crossref] [ Google Scholar]
- Yang XP, Ghoreschi K, Steward-Tharp SM, Rodriguez-Canales J, Zhu J, Grainger JR. Opposing regulation of the locus encoding IL-17 through direct, reciprocal actions of STAT3 and STAT5. Nat Immunol 2011; 12:247-54. doi: 10.1038/ni.1995 [Crossref] [ Google Scholar]
- Villarino AV, Gallo E, Abbas AK. STAT1-activating cytokines limit Th17 responses through both T-bet-dependent and -independent mechanisms. J Immunol 2010; 185:6461-71. doi: 10.4049/jimmunol.1001343 [Crossref] [ Google Scholar]
- Mathur AN, Chang HC, Zisoulis DG, Stritesky GL, Yu Q, O'Malley JT. Stat3 and Stat4 direct development of IL-17-secreting Th cells. J Immunol 2007; 178:4901-7. doi: 10.4049/jimmunol.178.8.4901 [Crossref] [ Google Scholar]
- Hildner KM, Schirmacher P, Atreya I, Dittmayer M, Bartsch B, Galle PR. Targeting of the transcription factor STAT4 by antisense phosphorothioate oligonucleotides suppresses collagen-induced arthritis. J Immunol 2007; 178:3427-36. doi: 10.4049/jimmunol.178.6.3427 [Crossref] [ Google Scholar]
- Liu X, Nurieva RI, Dong C. Transcriptional regulation of follicular T-helper (Tfh) cells. Immunol Rev 2013; 252:139-45. doi: 10.1111/imr.12040 [Crossref] [ Google Scholar]
- Jacobson EB, Caporale LH, Thorbecke GJ. Effect of thymus cell injections on germinal center formation in lymphoid tissues of nude (thymusless) mice. Cell Immunol 1974; 13:416-30. doi: 10.1016/0008-8749(74)90261-5 [Crossref] [ Google Scholar]
- Glasmacher E, Agrawal S, Chang AB, Murphy TL, Zeng W, Vander Lugt B. A genomic regulatory element that directs assembly and function of immune-specific AP-1-IRF complexes. Science 2012; 338:975-80. doi: 10.1126/science.1228309 [Crossref] [ Google Scholar]
- Li P, Spolski R, Liao W, Wang L, Murphy TL, Murphy KM, Leonard WJ. BATF-JUN is critical for IRF4-mediated transcription in T cells. Nature 2012; 490:543-6. doi: 10.1038/nature11530 [Crossref] [ Google Scholar]
- Ellyard JI, Vinuesa CG. A BATF-ling connection between B cells and follicular helper T cells. Nat Immunol 2011; 12:519-20. doi: 10.1038/ni.2042 [Crossref] [ Google Scholar]
- Castro A, Sengupta TK, Ruiz DC, Yang E, Ivashkiv LB. IL-4 selectively inhibits IL-2-triggered Stat5 activation, but not proliferation, in human T cells. J Immunol 1999; 162:1261-9. [ Google Scholar]
- Sahoo A, Alekseev A, Tanaka K, Obertas L, Lerman B, Haymaker C. Batf is important for IL-4 expression in T follicular helper cells. Nat Commun 2015; 6:7997. doi: 10.1038/ncomms8997 [Crossref] [ Google Scholar]
- Hercor M, Anciaux M, Denanglaire S, Debuisson D, Leo O, Andris F. Antigen-presenting cell-derived IL-6 restricts the expression of GATA3 and IL-4 by follicular helper T cells. J Leukoc Biol 2017; 101:5-14. doi: 10.1189/jlb.1HI1115-511R [Crossref] [ Google Scholar]
- Schmitt N, Liu Y, Bentebibel SE, Munagala I, Bourdery L, Venuprasad K. The cytokine TGF-β co-opts signaling via STAT3-STAT4 to promote the differentiation of human TFH cells. Nat Immunol 2014; 15:856-65. doi: 10.1038/ni.2947 [Crossref] [ Google Scholar]
- Johnston RJ, Choi YS, Diamond JA, Yang JA, Crotty S. STAT5 is a potent negative regulator of TFH cell differentiation. J Exp Med 2012; 209:243-50. doi: 10.1084/jem.20111174 [Crossref] [ Google Scholar]
- Hao H, Nakayamada S, Yamagata K, Ohkubo N, Iwata S, Inoue Y. Conversion of T Follicular Helper Cells to T Follicular Regulatory Cells by Interleukin-2 Through Transcriptional Regulation in Systemic Lupus Erythematosus. Arthritis Rheumatol 2021; 73:132-42. doi: 10.1002/art.41457 [Crossref] [ Google Scholar]
- Rocamora-Reverte L, Melzer FL, Würzner R, Weinberger B. The Complex Role of Regulatory T Cells in Immunity and Aging. Front Immunol 2020; 11:616949. doi: 10.3389/fimmu.2020.616949 [Crossref] [ Google Scholar]
- Lal G, Zhang N, van der Touw W, Ding Y, Ju W, Bottinger EP. Epigenetic regulation of Foxp3 expression in regulatory T cells by DNA methylation. J Immunol 2009; 182:259-73. doi: 10.4049/jimmunol.182.1.259 [Crossref] [ Google Scholar]
- Burchill MA, Yang J, Vogtenhuber C, Blazar BR, Farrar MA. IL-2 receptor beta-dependent STAT5 activation is required for the development of Foxp3 + regulatory T cells. J Immunol 2007; 178:280-90. doi: 10.4049/jimmunol.178.1.280 [Crossref] [ Google Scholar]
- Zheng SG, Wang J, Horwitz DA. Cutting edge: Foxp3 + CD4 + CD25 + regulatory T cells induced by IL-2 and TGF-beta are resistant to Th17 conversion by IL-6. J Immunol 2008; 180:7112-6. doi: 10.4049/jimmunol.180.11.7112 [Crossref] [ Google Scholar]
- O'Malley JT, Sehra S, Thieu VT, Yu Q, Chang HC, Stritesky GL. Signal transducer and activator of transcription 4 limits the development of adaptive regulatory T cells. Immunology 2009; 127:587-95. doi: 10.1111/j.1365-2567.2008.03037.x [Crossref] [ Google Scholar]
- Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 2006; 441:235-8. doi: 10.1038/nature04753 [Crossref] [ Google Scholar]
- Lee SE, Li X, Kim JC, Lee J, González-Navajas JM, Hong SH. Type I interferons maintain Foxp3 expression and T-regulatory cell functions under inflammatory conditions in mice. Gastroenterology 2012; 143:145-54. doi: 10.1053/j.gastro.2012.03.042 [Crossref] [ Google Scholar]
- Xu T, Pereira RM, Martinez GJ. An Updated Model for the Epigenetic Regulation of Effector and Memory CD8( + ) T Cell Differentiation. J Immunol 2021; 207:1497-505. doi: 10.4049/jimmunol.2100633 [Crossref] [ Google Scholar]
- Kovarik P, Stoiber D, Eyers PA, Menghini R, Neininger A, Gaestel M. Stress-induced phosphorylation of STAT1 at Ser727 requires p38 mitogen-activated protein kinase whereas IFN-gamma uses a different signaling pathway. Proc Natl Acad Sci U S A 1999; 96:13956-61. doi: 10.1073/pnas.96.24.13956 [Crossref] [ Google Scholar]
- Pilz A, Kratky W, Stockinger S, Simma O, Kalinke U, Lingnau K. Dendritic cells require STAT-1 phosphorylated at its transactivating domain for the induction of peptide-specific CTL. J Immunol 2009; 183:2286-93. doi: 10.4049/jimmunol.0901383 [Crossref] [ Google Scholar]
- Yu CR, Dambuza IM, Lee YJ, Frank GM, Egwuagu CE. STAT3 regulates proliferation and survival of CD8 + T cells: enhances effector responses to HSV-1 infection, and inhibits IL-10 + regulatory CD8 + T cells in autoimmune uveitis. Mediators Inflamm 2013; 2013:359674. doi: 10.1155/2013/359674 [Crossref] [ Google Scholar]
- Frederiksen KS, Lundsgaard D, Freeman JA, Hughes SD, Holm TL, Skrumsager BK. IL-21 induces in vivo immune activation of NK cells and CD8( + ) T cells in patients with metastatic melanoma and renal cell carcinoma. Cancer Immunol Immunother 2008; 57:1439-49. doi: 10.1007/s00262-008-0479-4 [Crossref] [ Google Scholar]
- Qin JZ, Zhang CL, Kamarashev J, Dummer R, Burg G, Döbbeling U. Interleukin-7 and interleukin-15 regulate the expression of the bcl-2 and c-myb genes in cutaneous T-cell lymphoma cells. Blood 2001; 98:2778-83. doi: 10.1182/blood.v98.9.2778 [Crossref] [ Google Scholar]
- Osborne LC, Dhanji S, Snow JW, Priatel JJ, Ma MC, Miners MJ. Impaired CD8 T cell memory and CD4 T cell primary responses in IL-7R alpha mutant mice. J Exp Med 2007; 204:619-31. doi: 10.1084/jem.20061871 [Crossref] [ Google Scholar]
- Lindemann MJ, Benczik M, Gaffen SL. Anti-apoptotic signaling by the interleukin-2 receptor reveals a function for cytoplasmic tyrosine residues within the common gamma (gamma c) receptor subunit. J Biol Chem 2003; 278:10239-49. doi: 10.1074/jbc.M209471200 [Crossref] [ Google Scholar]
- Häusser-Kinzel S, Weber MS. The Role of B Cells and Antibodies in Multiple Sclerosis, Neuromyelitis Optica, and Related Disorders. Front Immunol 2019; 10:201. doi: 10.3389/fimmu.2019.00201 [Crossref] [ Google Scholar]
- Papin JA, Palsson BO. The JAK-STAT signaling network in the human B-cell: an extreme signaling pathway analysis. Biophysical Journal 2004; 87:37-46. [ Google Scholar]
- Su L, Rickert RC, David M. Rapid STAT phosphorylation via the B cell receptorModulatory role of CD19. J Biol Chem 1999; 274:31770-4. doi: 10.1074/jbc.274.45.31770 [Crossref] [ Google Scholar]
- Domeier PP, Chodisetti SB, Soni C, Schell SL, Elias MJ, Wong EB. IFN-γ receptor and STAT1 signaling in B cells are central to spontaneous germinal center formation and autoimmunity. J Exp Med 2016; 213:715-32. doi: 10.1084/jem.20151722 [Crossref] [ Google Scholar]
- Nemoto K, Kawanami T, Hoshina T, Ishimura M, Yamasaki K, Okada S. Impaired B-Cell Differentiation in a Patient With STAT1 Gain-of-Function Mutation. Front Immunol 2020; 11:557521. doi: 10.3389/fimmu.2020.557521 [Crossref] [ Google Scholar]
- Chen TT, Tsai MH, Kung JT, Lin KI, Decker T, Lee CK. STAT1 regulates marginal zone B cell differentiation in response to inflammation and infection with blood-borne bacteria. J Exp Med 2016; 213:3025-39. doi: 10.1084/jem.20151620 [Crossref] [ Google Scholar]
- Hsu CC, Meeker SM, Escobar S, Brabb TL, Paik J, Park H. Murine norovirus inhibits B cell development in the bone marrow of STAT1-deficient mice. Virology 2018; 515:123-33. doi: 10.1016/j.virol.2017.12.013 [Crossref] [ Google Scholar]
- Diehl SA, Schmidlin H, Nagasawa M, van Haren SD, Kwakkenbos MJ, Yasuda E. STAT3-mediated up-regulation of BLIMP1 Is coordinated with BCL6 down-regulation to control human plasma cell differentiation. J Immunol 2008; 180:4805-15. doi: 10.4049/jimmunol.180.7.4805 [Crossref] [ Google Scholar]
- Avery DT, Deenick EK, Ma CS, Suryani S, Simpson N, Chew GY. B cell-intrinsic signaling through IL-21 receptor and STAT3 is required for establishing long-lived antibody responses in humans. J Exp Med 2010; 207:155-71. doi: 10.1084/jem.20091706 [Crossref] [ Google Scholar]
- Ettinger R, Kuchen S, Lipsky PE. The role of IL-21 in regulating B-cell function in health and disease. Immunol Rev 2008; 223:60-86. doi: 10.1111/j.1600-065X.2008.00631.x [Crossref] [ Google Scholar]
- Ellyard JI, Avery DT, Phan TG, Hare NJ, Hodgkin PD, Tangye SG. Antigen-selected, immunoglobulin-secreting cells persist in human spleen and bone marrow. Blood 2004; 103:3805-12. doi: 10.1182/blood-2003-09-3109 [Crossref] [ Google Scholar]
- Rozovski U, Wu JY, Harris DM, Liu Z, Li P, Hazan-Halevy I. Stimulation of the B-cell receptor activates the JAK2/STAT3 signaling pathway in chronic lymphocytic leukemia cells. Blood 2014; 123:3797-802. doi: 10.1182/blood-2013-10-534073 [Crossref] [ Google Scholar]
- Papaioannou I, Xu S, Denton CP, Abraham DJ, Ponticos M. STAT3 controls COL1A2 enhancer activation cooperatively with JunB, regulates type I collagen synthesis posttranscriptionally, and is essential for lung myofibroblast differentiation. Mol Biol Cell 2018; 29:84-95. doi: 10.1091/mbc.E17-06-0342 [Crossref] [ Google Scholar]
- Kuraishy A, Karin M, Grivennikov SI. Tumor promotion via injury-and death-induced inflammation. Immunity 2011; 35:467-77. [ Google Scholar]
- Naugler WE, Karin M. The wolf in sheep's clothing: the role of interleukin-6 in immunity, inflammation and cancer. Trends in molecular medicine 2008; 14:109-19. [ Google Scholar]
- Takeda K, Clausen BE, Kaisho T, Tsujimura T, Terada N, Förster I, Akira S. Enhanced Th1 activity and development of chronic enterocolitis in mice devoid of Stat3 in macrophages and neutrophils. Immunity 1999; 10:39-49. [ Google Scholar]
- Fredholm S, Gjerdrum LM, Willerslev-Olsen A, Petersen DL, Nielsen I, Kauczok CS. STAT3 activation and infiltration of eosinophil granulocytes in mycosis fungoides. Anticancer Res 2014; 34:5277-86. [ Google Scholar]
- Das M, Stephen-Victor E, Bayry J. Regulatory T cells do not suppress rather activate human basophils by IL-3 and STAT5-dependent mechanisms. Oncoimmunology 2020; 9:1773193. doi: 10.1080/2162402x.2020.1773193 [Crossref] [ Google Scholar]
- Li F, Sutherland MK, Yu C, Walter RB, Westendorf L, Valliere-Douglass J. Characterization of SGN-CD123A, A Potent CD123-Directed Antibody-Drug Conjugate for Acute Myeloid Leukemia. Mol Cancer Ther 2018; 17:554-64. doi: 10.1158/1535-7163.mct-17-0742 [Crossref] [ Google Scholar]
- Gotthardt D, Sexl V. STATs in NK-cells: the good, the bad, and the ugly. Front immunl 2017; 7:694. [ Google Scholar]
- Au-Yeung N, Mandhana R, Horvath CM. Transcriptional regulation by STAT1 and STAT2 in the interferon JAK-STAT pathway. Jak-stat 2013; 2:e23931. [ Google Scholar]
- Hashemi V, Ahmadi A, Malakotikhah F, Chaleshtari MG, Baghi Moornani M, Masjedi A. Silencing of p68 and STAT3 synergistically diminishes cancer progression. Life Sci 2020; 249:117499. doi: 10.1016/j.lfs.2020.117499 [Crossref] [ Google Scholar]
- Gotthardt D, Putz EM, Straka E, Kudweis P, Biaggio M, Poli V. Loss of STAT3 in murine NK cells enhances NK cell–dependent tumor surveillance. Blood 2014; 124:2370-9. [ Google Scholar]
- Klempner SJ, Gershenhorn B, Tran P, Lee TK, Erlander MG, Gowen K. BRAF V600E mutations in high-grade colorectal neuroendocrine tumors may predict responsiveness to BRAF–MEK combination therapy. Cancer Discovery 2016; 6:594-600. [ Google Scholar]
- Irey EA, Lassiter CM, Brady NJ, Chuntova P, Wang Y, Knutson TP. JAK/STAT inhibition in macrophages promotes therapeutic resistance by inducing expression of protumorigenic factors. Proc Natl Acad Sci U S A 2019; 116:12442-51. doi: 10.1073/pnas.1816410116 [Crossref] [ Google Scholar]
- Hu WM, Liu SQ, Zhu KF, Li W, Yang ZJ, Yang Q. The ALOX5 inhibitor Zileuton regulates tumor-associated macrophage M2 polarization by JAK/STAT and inhibits pancreatic cancer invasion and metastasis. Int Immunopharmacol 2023; 121:110505. doi: 10.1016/j.intimp.2023.110505 [Crossref] [ Google Scholar]
- Nefedova Y, Huang M, Kusmartsev S, Bhattacharya R, Cheng P, Salup R. Hyperactivation of STAT3 is involved in abnormal differentiation of dendritic cells in cancer. J Immunol 2004; 172:464-74. doi: 10.4049/jimmunol.172.1.464 [Crossref] [ Google Scholar]
- Andreu-Sanz D, Kobold S. Role and Potential of Different T Helper Cell Subsets in Adoptive Cell Therapy. Cancers (Basel) 2023; 15. 10.3390/cancers15061650.
- Gacerez AT, Sentman CL. T-bet promotes potent antitumor activity of CD4( + ) CAR T cells. Cancer Gene Ther 2018; 25:117-28. doi: 10.1038/s41417-018-0012-7 [Crossref] [ Google Scholar]
- Jacenik D, Karagiannidis I, Beswick EJ. Th2 cells inhibit growth of colon and pancreas cancers by promoting anti-tumorigenic responses from macrophages and eosinophils. Br J Cancer 2023; 128:387-97. doi: 10.1038/s41416-022-02056-2 [Crossref] [ Google Scholar]
- De Monte L, Reni M, Tassi E, Clavenna D, Papa I, Recalde H. Intratumor T helper type 2 cell infiltrate correlates with cancer-associated fibroblast thymic stromal lymphopoietin production and reduced survival in pancreatic cancer. J Exp Med 2011; 208:469-78. doi: 10.1084/jem.20101876 [Crossref] [ Google Scholar]
- Hajimoradi M, Rezalotfi A, Esmaeilnejad-Ahranjani P, Hassan ZM, Ebrahimi M. STAT3 inactivation suppresses stemness properties in gastric cancer stem cells and promotes Th17 in Treg/Th17 balance. International Immunopharmacology 2022; 111:109048. [ Google Scholar]
- Numasaki M, Fukushi J-i, Ono M, Narula SK, Zavodny PJ, Kudo T. Interleukin-17 promotes angiogenesis and tumor growth. Blood 2003; 101:2620-7. [ Google Scholar]
- Mayberry CL, Logan NA, Wilson JJ, Chang CH. Providing a Helping Hand: Metabolic Regulation of T Follicular Helper Cells and Their Association With Disease. Front Immunol 2022; 13:864949. doi: 10.3389/fimmu.2022.864949 [Crossref] [ Google Scholar]
- Ou W, Thapa RK, Jiang L, Soe ZC, Gautam M, Chang J-H. Regulatory T cell-targeted hybrid nanoparticles combined with immuno-checkpoint blockage for cancer immunotherapy. Journal of Controlled Release 2018; 281:84-96. [ Google Scholar]
- Chen L, Wang S, Wang Y, Zhang W, Ma K, Hu C. IL-6 influences the polarization of macrophages and the formation and growth of colorectal tumor. Oncotarget 2018; 9:17443. [ Google Scholar]
- Vera-Lozada G, Minnicelli C, Segges P, Stefanoff G, Kristcevic F, Ezpeleta J. Interleukin 10 (IL10) proximal promoter polymorphisms beyond clinical response in classical Hodgkin lymphoma: Exploring the basis for the genetic control of the tumor microenvironment. Oncoimmunology 2018; 7:e1389821. [ Google Scholar]
- Najafi M, Farhood B, Mortezaee K. Contribution of regulatory T cells to cancer: A review. Journal of cellular physiology 2019; 234:7983-93. [ Google Scholar]
- Lu C, Talukder A, Savage NM, Singh N, Liu K. JAK-STAT-mediated chronic inflammation impairs cytotoxic T lymphocyte activation to decrease anti-PD-1 immunotherapy efficacy in pancreatic cancer. Oncoimmunology 2017; 6:e1291106. [ Google Scholar]
- Viganò E, Gunawardana J, Mottok A, Van Tol T, Mak K, Chan FC. Somatic IL4R mutations in primary mediastinal large B-cell lymphoma lead to constitutive JAK-STAT signaling activation. Blood 2018; 131:2036-46. doi: 10.1182/blood-2017-09-808907 [Crossref] [ Google Scholar]
- Kortylewski M, Kujawski M, Wang T, Wei S, Zhang S, Pilon-Thomas S. Inhibiting Stat3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nat Med 2005; 11:1314-21. doi: 10.1038/nm1325 [Crossref] [ Google Scholar]
- Kitamura H, Ohno Y, Toyoshima Y, Ohtake J, Homma S, Kawamura H. Interleukin-6/STAT3 signaling as a promising target to improve the efficacy of cancer immunotherapy. Cancer Sci 2017; 108:1947-52. doi: 10.1111/cas.13332 [Crossref] [ Google Scholar]
- Wang T, Niu G, Kortylewski M, Burdelya L, Shain K, Zhang S. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat Med 2004; 10:48-54. doi: 10.1038/nm976 [Crossref] [ Google Scholar]
- Zou S, Tong Q, Liu B, Huang W, Tian Y, Fu X. Targeting STAT3 in Cancer Immunotherapy. Mol Cancer 2020; 19:145. doi: 10.1186/s12943-020-01258-7 [Crossref] [ Google Scholar]
- Wu P, Wu D, Zhao L, Huang L, Shen G, Huang J, Chai Y. Prognostic role of STAT3 in solid tumors: a systematic review and meta-analysis. Oncotarget 2016; 7:19863-83. doi: 10.18632/oncotarget.7887 [Crossref] [ Google Scholar]
- Wang YD, Sun XJ, Yin JJ, Yin M, Wang W, Nie ZQ, Xu J. Long non-coding RNA FEZF1-AS1 promotes cell invasion and epithelial-mesenchymal transition through JAK2/STAT3 signaling pathway in human hepatocellular carcinoma. Biomed Pharmacother 2018; 106:134-41. doi: 10.1016/j.biopha.2018.05.116 [Crossref] [ Google Scholar]
- Jiao J, Zhang R, Li Z, Yin Y, Fang X, Ding X. Nuclear Smad6 promotes gliomagenesis by negatively regulating PIAS3-mediated STAT3 inhibition. Nat Commun 2018; 9:2504. doi: 10.1038/s41467-018-04936-9 [Crossref] [ Google Scholar]
- Wake MS, Watson CJ. STAT3 the oncogene - still eluding therapy?. Febs j 2015; 282:2600-11. doi: 10.1111/febs.13285 [Crossref] [ Google Scholar]
- Zhao C, Li H, Lin HJ, Yang S, Lin J, Liang G. Feedback Activation of STAT3 as a Cancer Drug-Resistance Mechanism. Trends Pharmacol Sci 2016; 37:47-61. doi: 10.1016/j.tips.2015.10.001 [Crossref] [ Google Scholar]
- Berdeja J, Palandri F, Baer MR, Quick D, Kiladjian JJ, Martinelli G. Phase 2 study of gandotinib (LY2784544) in patients with myeloproliferative neoplasms. Leuk Res 2018; 71:82-8. doi: 10.1016/j.leukres.2018.06.014 [Crossref] [ Google Scholar]
- Mascarenhas J, Hoffman R, Talpaz M, Gerds AT, Stein B, Gupta V. Pacritinib vs Best Available Therapy, Including Ruxolitinib, in Patients With Myelofibrosis: A Randomized Clinical Trial. JAMA Oncol 2018; 4:652-9. doi: 10.1001/jamaoncol.2017.5818 [Crossref] [ Google Scholar]
- Li X, Jiang W, Dong S, Li W, Zhu W, Zhou W. STAT3 Inhibitors: A Novel Insight for Anticancer Therapy of Pancreatic Cancer. Biomolecules 2022; 12. 10.3390/biom12101450.
- Yang L, Lin S, Xu L, Lin J, Zhao C, Huang X. Novel activators and small-molecule inhibitors of STAT3 in cancer. Cytokine Growth Factor Rev 2019; 49:10-22. doi: 10.1016/j.cytogfr.2019.10.005 [Crossref] [ Google Scholar]
- Liu Y, Wang X, Zeng S, Zhang X, Zhao J, Zhang X. The natural polyphenol curcumin induces apoptosis by suppressing STAT3 signaling in esophageal squamous cell carcinoma. J Exp Clin Cancer Res 2018; 37:303. doi: 10.1186/s13046-018-0959-0 [Crossref] [ Google Scholar]
- Lv Z, Yan X, Lu L, Su C, He Y. Atovaquone enhances doxorubicin's efficacy via inhibiting mitochondrial respiration and STAT3 in aggressive thyroid cancer. J Bioenerg Biomembr 2018; 50:263-70. doi: 10.1007/s10863-018-9755-y [Crossref] [ Google Scholar]
- Lo HW, Cao X, Zhu H, Ali-Osman F. Constitutively activated STAT3 frequently coexpresses with epidermal growth factor receptor in high-grade gliomas and targeting STAT3 sensitizes them to Iressa and alkylators. Clin Cancer Res 2008; 14:6042-54. doi: 10.1158/1078-0432.Ccr-07-4923 [Crossref] [ Google Scholar]
- Real PJ, Sierra A, De Juan A, Segovia JC, Lopez-Vega JM, Fernandez-Luna JL. Resistance to chemotherapy via Stat3-dependent overexpression of Bcl-2 in metastatic breast cancer cells. Oncogene 2002; 21:7611-8. doi: 10.1038/sj.onc.1206004 [Crossref] [ Google Scholar]