Bioimpacts. 2025;15:30129.
doi: 10.34172/bi.30129
Systematic Review
The effectiveness of antioxidant agents in delaying progression of diabetic nephropathy: A systematic review of randomized controlled trials
Ika Rahayu Conceptualization, Data curation, Formal analysis, Writing – original draft, Writing – review & editing, 1, 2 
Nur Arfian Conceptualization, Data curation, Formal analysis, Writing – review & editing, 3, 4, * 
Christina Yeni Kustanti Formal analysis, Visualization, Writing – review & editing, 5, 6 
Mae Sri Hartati Wahyuningsih Funding acquisition, Supervision, Visualization, Writing – review & editing, 4, 7 
Author information:
1Doctoral Program of Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada, Yogyakarta, Indonesia
2Department of Biochemistry, Faculty of Medicine and Health Sciences, Universitas Kristen Krida Wacana, Jakarta, Indonesia
3Department of Anatomy, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada, Yogyakarta, Indonesia
4Center for Herbal Medicine, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada, Yogyakarta, Indonesia
5Sekolah Tinggi Ilmu Kesehatan Bethesda Yakkum, Yogyakarta, Indonesia
6Lotus Care, Private Clinic for Wound and Palliative Care, Homecare, Yogyakarta, Indonesia
7Department of Pharmacology and Therapy, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada, Yogyakarta, Indonesia
Abstract
Introduction:
Oxidative stress plays a central role in the pathophysiology of diabetes mellitus and its complications, including diabetic nephropathy. Excessive production of reactive oxygen species (ROS) alters renal metabolic pathways, leading to inflammation, endothelial dysfunction, and fibrosis, ultimately resulting in end-stage renal disease (ESRD). Studies have shown that exogenous antioxidants can improve the pathophysiological condition of patients with diabetic nephropathy. Objective: This systematic review aims to investigate the types of antioxidant agents that inhibit the development of diabetic nephropathy and the effectiveness of antioxidant agent interventions to repair kidney structure and function.
Methods:
A systematic review of randomized controlled trials that examined the role of antioxidants in improving diabetic nephropathy was conducted. The literature search was performed on PubMed, ScienceDirect, and EBSCO. The inclusion criteria covered articles on the antioxidant activity of herbal extracts and compounds that inhibit the progression of diabetic nephropathy in humans. In addition, the articles were written in English and published between 2012 and 2022. The reporting of the systematic review followed the Preferred Reporting Elements for Systematic Review and Meta-Analysis (PRISMA) guideline. The full texts of all potentially relevant systematic reviews were assessed for quality using the Risk of Bias 2 (RoB 2) tool.
Results:
A total of 2,367 articles were identified in the three databases, of which only 15 articles met the inclusion criteria. Antioxidant agents that inhibit diabetic nephropathy can be classified as single antioxidants (silymarin, baicalin, epigallocatechin gallate, vitamin E, selenium, curcumin, α-lipoic acid, and tocotrienol-rich vitamin E) and combined antioxidants (α-lipoic acid with vitamin B6, and resveratrol with losartan). Antioxidant agents have been shown to reduce oxidative stress and inflammation, but their role in the progression of fibrosis remains unclear. The oxidative stress marker MDA was significantly reduced by silymarin, curcumin, vitamin E, tocotrienol-rich vitamin E, selenium, ALA, vitamin B, resveratrol and losartan. Silymarin was found to be the most effective (-3.43 µmol/L; 6.02 to 0.83). Compared to silymarin and epigallocatechin gallate, vitamin E was more effective (at -35.4 ng/L; P < 0.001) in reducing inflammation by decreasing TNF-α levels. In addition, tocotrienol-rich vitamin E, silymarin, baicalin, and selenium showed a decrease TGF-β levels, but did not show statistically significant differences between the placebo and intervention groups.
Conclusion:
Potential antioxidant agents, such as flavonoids, vitamins, fatty acids, and antioxidant minerals, were examined in this systematic review. These agents contribute to reducing markers of oxidative stress and hyperglycemia-induced inflammation. Although several antioxidants play a role in reducing fibrosis markers, the effect does not appear to be statistically significant.
Keywords: Antioxidant, Diabetes mellitus, Diabetic nephropathy, Diabetic kidney disease, Kidney fibrosis
Copyright and License Information
© 2025 The Author(s).
This work is published by BioImpacts as an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (
http://creativecommons.org/licenses/by-nc/4.0/). Non-commercial uses of the work are permitted, provided the original work is properly cited.
Funding Statement
The authors would like to thank Universitas Gadjah Mada for funding this study through the Rekognisi Tugas Akhir (RTA) program with a grant number 5075/UN1.P.II/Dit-Lit/PT.01.01/2023. This study could not have been done without their support.
Introduction
Diabetes mellitus (DM) is a chronic disease and a global health problem that is increasing in prevalence.1 Approximately 537 million adults between the ages of 20 and 79 years live with diabetes.2 Uncontrolled diabetes mellitus can lead to complications and have a significant impact on the patient’s life. Diabetes is also the leading cause of chronic and end-stage renal disease worldwide, with an average of 50% of individuals with diabetes mellitus suffering from kidney disease.3,4 The current management aims to minimize the risk of micro- and macrovascular complications, including cardiovascular disease and diabetic nephropathy.5 Diabetic nephropathy is a significant long-term complication of diabetes. Patients with kidney disease are treated with renal replacement therapies such as hemodialysis or kidney transplantation. The estimated glomerular filtration rate (eGFR) has been used to predict this outcome due to its strong correlation with the progression of kidney disease and the subsequent need for dialysis to a significant extent.6 Although many alternative solutions are available to prevent kidney disease, current strategies have not provided stable long-term outcomes.7
Oxidative stress plays a crucial role in the pathophysiology of diabetic nephropathy. Hyperglycemic conditions induce excessive production of reactive oxygen species (ROS) in mitochondria, activate nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, and release endothelial nitric oxide synthase (eNOS), all of which are sources of ROS. The increased amount of ROS causes deoxyribonucleic acid (DNA) damage, resulting in the breakage of DNA strand. In response to this, the DNA repair mechanism is activated. In addition, the enzyme poly (ADP ribose) polymerase 1 (PARP1) plays a role in triggering the inhibition of the glycolytic enzyme glyceraldehyde-3-phosphate dehydrogenase (GAPDH) through poly-ADP-ribosylation, causing a bottleneck in glycolysis and leading to the accumulation of early glycolytic intermediates.4 These promote the polyol pathway flux, increase the formation of advanced glycation end products (AGEs) as well as the expression of AGE receptors and their activating ligands, activate protein kinase C (PKC) isoforms, and over-activate the hexosamine pathway.8 Endothelial cells and smooth muscle are targets of oxidative damage caused by modifications to proteins, lipids, and nucleic acids.9 Therefore, it is necessary to increase the levels of antioxidants in the body to combat ROS.
Based on a review of 14 studies, Bolignano et al found that antioxidants can ameliorate early kidney disease caused by diabetes mellitus.10 This study aims to update their previous findings by adding several parameters related to structural kidney disease due to hyperglycemia, including oxidative stress markers, inflammation, and fibrosis.
This study investigates the types of antioxidant agents that inhibit the development of diabetic nephropathy and the effectiveness of antioxidant agent interventions to repair kidney structure and function. In addition, this study observes changes in the levels of urine albumin-to-creatinine ratio (UACR), creatinine, AGE production, inflammatory markers, and antioxidants in patients who received antioxidant therapy and those who did not.
Methods
Registration and reporting guideline
The reporting of the systematic review followed the Preferred Reporting Elements for Systematic Review and Meta-Analysis (PRISMA) guideline.11 The review protocol was registered in the Open Science Framework under the DOI 10.17605/OSF.IO/9EBNQ.
Search strategy and eligibility criteria
A thorough search was conducted for articles published between 2012 and 2022 in electronic databases, including PubMed, Science Direct, and EBSCO. The search strategy used a combination of Medical Subject Headings (MeSH), Emtree, keywords, and Boolean operators, following the Population, Intervention, Comparison, and Outcomes (PICO) framework. The population consisted of adults with diabetic nephropathy (type 1 and type 2 diabetes) aged 18 years and older. The interventions included antioxidant agents such as silymarin, baicalin, tocotrienol-rich vitamin E, epigallocatechin gallate, vitamin E, and selenium. For comparison, standard therapy was included. The outcomes were changes in the levels of ultra-glomerular structure markers (UACR, proteinuria, creatinine, and eGFR), changes in the levels of oxidative stress markers involved in the development of diabetic nephropathy (AGEs and malondialdehyde/MDA), changes in the levels of pro-inflammatory markers (tumor necrosis factor-α/TNF-α), changes in the levels of fibrosis markers (transforming growth factor-β/TGF-β, matrix-metalloproteinase-2/MMP-2, and matrix-metalloproteinase-9/MMP-9), and changes in the levels of total antioxidant capacity and antioxidant enzymes. The main search terms were “antioxidant" AND "diabetic nephropathy" OR "antioxidant" AND "diabetic kidney disease." The database searches were performed by the first author (IR), validated by CYK, and reviewed by the other authors during research meetings. Fig. 1 shows the detailed search strategy. In addition, forward citation searches were conducted to identify citations to specific articles, while backward citation searches were conducted to examine the reference list of relevant systematic reviews and other studies (see Supplementary file 1).

Fig. 1.
Database search strategy.
.
Database search strategy.
Study screening and selection
Articles included in this review met the following criteria: (1) quantitative studies that used a randomized controlled trial (RCT) design; (2) examining the effect of single or combined antioxidant activity or antioxidant compounds from medicinal plant extracts on inhibiting diabetic nephropathy in humans; (3) clearly stating the name of the single or combined active compounds along with the dose of administration; (4) identifying the names of the active compounds contained in the medicinal plants and their dosage; and (5) written in English.
On the other hand, articles excluded from this review met the following criteria: (1) studies in the forms of case studies, reviews, editorials, letters, and short communication; (2) studies that involved individuals under the age of 18 years; 3) studies focusing on diabetic patients without kidney disease or chronic kidney disease patients without diabetes; 4) studies focusing on patients with chronic diabetic nephropathy or acute renal replacement therapy (e.g., hemodialysis or peritoneal dialysis); and 5) studies testing the effects of drugs, synthetic antioxidants, or mixtures of antioxidants without precisely determined compositions.
Screening of articles based on titles and abstracts was conducted independently by two reviewers (IR and NA). After duplicates were removed, titles and abstracts from the literature search were examined to identify potentially relevant studies. Following this process, full texts were reviewed to determine which studies met the inclusion criteria. Any discrepancies in the screening process were discussed and resolved with the other reviewers (CYK and MSH).
Data extraction and study quality assessment
The data were extracted by the first author (IR) and verified by the second author (NA). After the data were entered into an Excel file, a table was created to present the characteristics of the included studies. The table includes information about the author, study location, diagnosis of the participants, diagnostic standards, sample size, mean age, sex, type of the interventions, and duration. The study quality was assessed using the Risk of Bias 2 (RoB 2) tool developed by the Cochrane Collaboration.12 This tool was designed to assess study quality based on five domains and the approach used (intention-to-treat or per-protocol). The domains assessed include: (1) bias due to the randomization procedure, (2) bias due to deviations from the intended interventions, (3) bias due to missing data on the outcomes, (4) bias in the measurement of the outcomes, and (5) bias in the selection of the reported outcomes. Each item was rated as “yes”, “probably yes”, “no”, “probably no”, and “no information”. Meanwhile, specific questions related to each domain were rated as "low risk of bias", "high risk of bias", or "some concerns”.
Data synthesis
The final review presented a narrative synthesis of the results of the selected studies, including the types of antioxidant agents used in the intervention and the changes in the levels of ultra-glomerular structure markers, oxidative stress markers, proinflammatory markers, and total antioxidant capacity.
This study included several RCTs that used single or combined antioxidants in patients with diabetic nephropathy. Moreover, this study analyzed the dosage and duration of antioxidant administration to the patients, as well as its comparison. However, only articles that met the quality criteria as determined by the ROB 2 were included in the subsequent analysis.
Results
Study selection
A total of 2367 articles were found in the three databases combined. After duplicates were removed, titles and abstracts of the remaining articles were screened. This resulted in the exclusion of 1378 articles from further examination, leaving only 348 articles to be assessed for eligibility. Furthermore, 289 articles with irrelevant abstracts were excluded, leaving 59 articles for full-text review. Finally, 15 articles met all inclusion criteria (see Fig. 2).
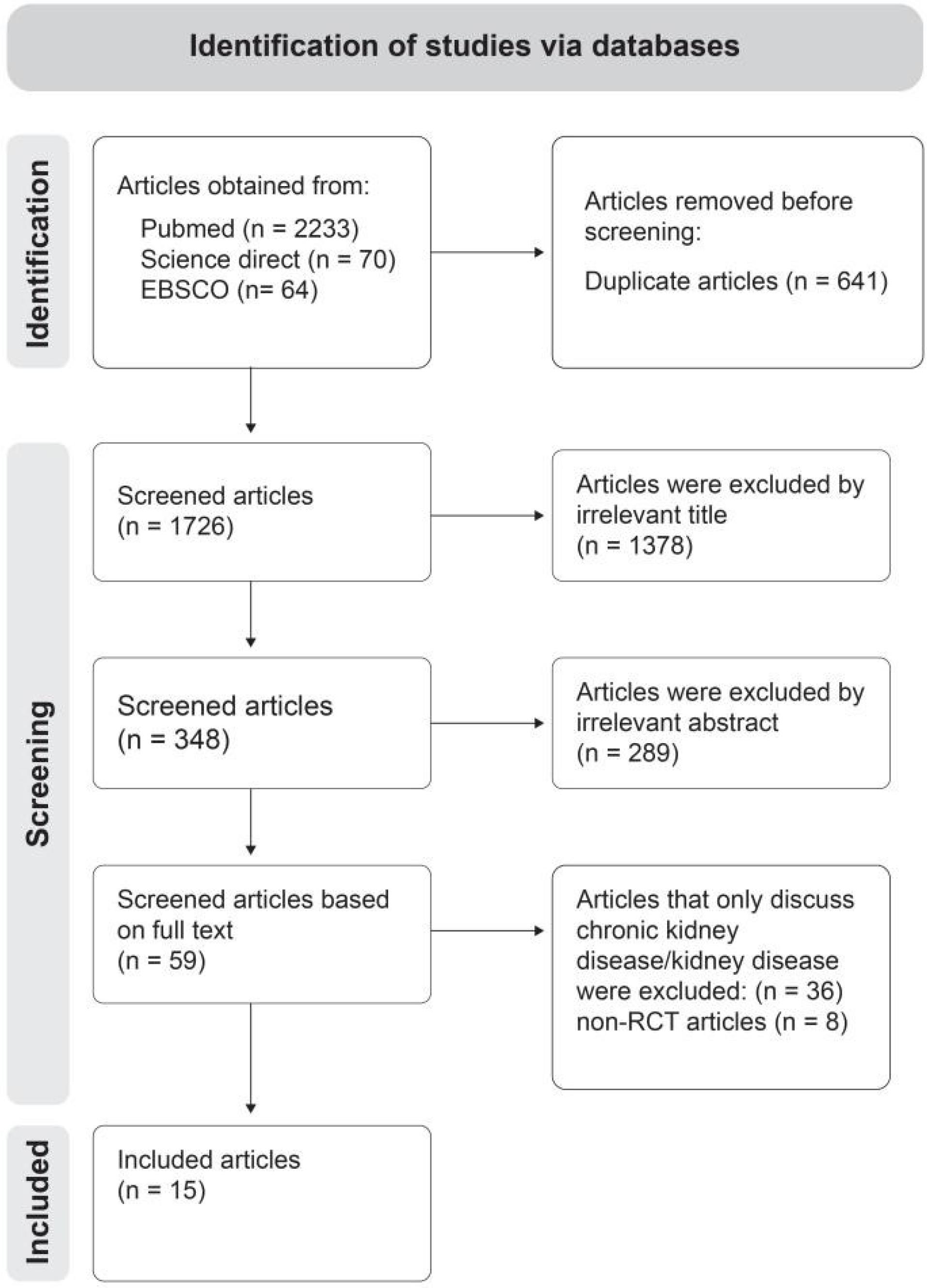
Fig. 2.
PRISMA flow diagram.
.
PRISMA flow diagram.
Characteristics of the studies
Patients included in the studies were between the ages of 55 and 70 years and had been diagnosed with diabetic nephropathy (see Table 1). The 15 studies were published between 2012 and 2022, with 80% of them conducted in Asia, 13,3% in America, and 6,7% in Europe. The patients received either single or combined antioxidant therapy. The single antioxidants included silymarin, baicalin, epigallocatechin gallate, vitamin E, selenium, curcumin, α-lipoic acid, and tocotrienol-rich vitamin E. In addition, the combined antioxidants included a combination of α-lipoic acid and vitamin B6 as well as a combination of resveratrol and losartan. The daily doses were silymarin 420 mg, baicalin 2400 mg, epigallocatechin gallate 800 mg, vitamin E 800-1200 IU, selenium 200µg, curcumin 320 mg, α-lipoic acid 600 mg, and tocotrienol-rich vitamin E 400 mg. The combination of lipoic acid and vitamin B6 was given at 800 mg/day and 80 mg/day, respectively. Meanwhile, the combination of resveratrol and losartan was given at 500 mg/day and 12.5 mg/day, respectively. All studies compared the group that received placebo with the group that received antioxidants while receiving standard therapy.
Table 1.
Characteristics of studies on diabetic nephropathy interventions
|
Author, year
|
Setting
|
Characteristics of population
|
|
Study population
|
Diagnosis
|
Sample size (n)
|
Mean age
|
Sex (%)
|
Design
|
Duration
(weeks)
|
| Aghadavod et al. (2018) |
Iran |
Diabetic nephropathy |
Proteinuria > 0.3 g/24h |
54 |
I: 62.2 ± 9.8
C: 64.5 ± 9.2 |
Male: 29.6
Female: 70.4 |
I: Vitamin E 800 IU/day
C: Placebo |
12 |
Bahmani et al
(2015) |
Iran |
Diabetic nephropathy |
eGFR > 30 ml/min/1.73 m2
Proteinuria > 0.3 g/24h |
60 |
I: 63.1 ± 12.6
C: 61.4 ± 9.3 |
Male: 50
Female: 50 |
I: Selenium 200 µg/day
C: Placebo |
12 |
Bahmani et al
(2016) |
Iran |
Diabetic nephropathy |
eGFR > 30 ml/min/1.73 m2
Proteinuria > 0.3 g/24h |
60 |
I: 63.1 ± 12.6
C: 61.4 ± 9.3 |
Male: 50
Female: 50 |
I: Selenium 200 µg/day
C: Placebo |
12 |
Borges et al
(2016) |
Brazil |
Diabetic nephropathy |
UACR > 30 mg/gcreatinine |
45 |
I: 63
C: 59 |
Male: 60
Female: 40 |
I: Epigallocatechin gallate 800mg/day
C: Placebo |
12 |
| Fallahzadeh et al (2012) |
Iran |
Diabetic nephropathy |
Albuminuria > 300 mg/24h
eGFR > 30 ml/min/1.73 m2 |
60 |
I: 55.9 ± 8.3
C: 57.6 ± 7.5 |
Male: -
Female: 100 |
I: Silymarin 140 mg (3x/day)
C: Placebo |
12 |
| Jimenez-Osorio et al/ (2016) |
Mexico |
Diabetic nephropathy |
Proteinuria ≥ 1 g/24h |
101 |
I: 55.0 ± 6 1.6
C: 56.2 ± 6 1.5 |
Male: 59
Female: 15 |
I: Curcumin 320 mg/day
C: Placebo |
12 |
Khatami et al
(2016) |
Iran |
Diabetic nephropathy |
Proteinuria > 0.3 g/24h |
60 |
I: 61.2 ± 10.0
C: 62.2 ± 13.8 |
Male: 35
Female: 65 |
I: Vitamin E 1200 IU/day
C: Placebo |
12 |
| Koay et al (2021) |
Malaysia |
Diabetic nephropathy |
eGFR 30-60 ml/min/1.73 m2
UACR > 20-200 mg/mmol |
59 |
I: 66
C: 70 |
Male: 64.4
Female: 35.6 |
I: Tocotrienol-rich vitamin E 200 mg (2x/day)
C: Placebo |
48 |
Noori et al
(2013) |
Iran |
Diabetic nephropathy |
UACR 30-1000 mg/gcreatinine |
40 |
I: 60.0 ± 2.0
C: 61.0 ± 3.0 |
Male: 32.5
Female: 52.5 |
I: Lipoic acid 800mg/day + vit B6 (pyridoxin) 80 mg (2x/day)
C: Placebo |
12 |
| Sun et al (2016) |
China |
Diabetic nephropathy |
Albuminuria 30-300 mg/24h |
62 |
I: 57.2 ± 8.2
C: 55.6 ± 6.4 |
Male: 45.2
Female: 54.8 |
I: α-lipoic acid IV 600mg/day
C: Placebo |
8 |
| Sattarinezhad et al (2019) |
Iran |
Diabetic nephropathy |
eGFR > 30 ml/min/1.73 m2
UACR > 30 mg/gcreatinine |
60 |
I: 56.8 ± 9.7
C: 55.7 ± 10.8 |
Male: 45
Female: 55 |
I: Resveratrol 500 mg/day + losartan 12,5 mg/day
C: Placebo |
12 |
| Tan et al (2018) |
Malaysia |
Diabetic nephropathy |
UACR > 10 mg/mmol |
66 |
I: 61.6 ± 9.5 |
Male: 72.7
Female: 27.3 |
I: Tocotrienol-rich vitamin E 200 mg (2x/day)
C: Placebo |
8 |
| Tan et al (2019) |
Malaysia |
Diabetic nephropathy |
eGFR 30-60 ml/min/1.73 m2
UACR > 10 mg/mmol |
54 |
I: 59 ± 10
C: 62.8 ± 11.6 |
Male: 64.8
Female: 35.2 |
I: Tocotrienol-rich vitamin E 200 mg (2x/day)
C: Placebo |
8 |
| Voroneanu et al (2017) |
Romania |
DMT2 patients with persistent proteinuria |
eGFR > 30 ml/min/1.73 m2
Proteinuria > 0.5 g/24h |
102 |
I: 63.8 ± 9.6
C: 64.8 ± 9.7 |
Male: 68.6
Female: 31.4 |
I: Silymarin 150 mg (3x/day)
C: Placebo |
96 |
| Yang et al (2019) |
China |
Diabetic nephropathy |
eGFR 30-60 ml/min/1.73 m2
UACR > 10 mg/mmol |
95 |
I: 55.7 ± 9.2
C: 54.8 ± 8.4 |
Male: 49.5
Female: 50.5 |
I: Common treatment + baicalin 800 mg (3x/day)
C: Common treatment |
24 |
Quality assessment
Two researchers independently assessed the study quality using the RoB 2. Any discrepancies among the assessors were discussed and resolved with the help of the other researchers. Table 2 shows the final results of the study quality assessment. Out of the 15 studies, eight studies (53.3%) had a low risk of bias, while seven studies (46.7%) had some concerns about bias. The main areas of concern were the randomization process and the allocation concealment in the included studies. For example, Borges et al. did not mention the randomization of participants into the experiment and control groups. Regarding allocation concealment, authors should clearly state the method used to prevent allocation disclosure, such as opaque sealed envelopes or closed containers. However, several authors, among whom were Voroneanu et al, did not provide sufficient information about this.
Table 2.
Risk of bias assessment
|
Study
|
Domain
|
|
Randomization
|
Deviations from intended interventions
|
Missing data on the outcomes
|
Measurement of the outcomes
|
Selection of the reported outcomes
|
Overall bias
|
| Aghadavod et al, 2018 |
S |
L |
L |
L |
L |
S |
| Bahmani et al, 2015 |
S |
L |
L |
L |
L |
S |
| Bahmani et al, 2016 |
L |
L |
L |
L |
L |
L |
| Borges et al, 2016 |
S |
L |
L |
L |
L |
S |
| Fallahzadeh et al, 2012 |
L |
L |
L |
L |
L |
L |
| Jimeenez-Osorio et al, 2016 |
L |
L |
L |
L |
L |
L |
| Khatami et al, 2016 |
L |
L |
L |
L |
L |
L |
| Koay et al, 2021 |
L |
L |
L |
L |
L |
L |
| Noori et al, 2013 |
S |
L |
L |
L |
L |
S |
| Sattarinezhad et al, 2018 |
L |
L |
L |
L |
L |
L |
| Sun et al, 2016 |
S |
L |
L |
L |
L |
S |
| Tan et al, 2019 |
L |
L |
L |
L |
L |
L |
| Tan et al, 2018 |
L |
L |
L |
L |
L |
L |
| Voroneanu et al, 2017 |
S |
L |
L |
L |
L |
S |
| Yang et al, 2018 |
S |
L |
L |
L |
L |
S |
Note: H: high risk of bias, S: some concerns, L: low risk of bias
The search results were individually reviewed by two independent researchers. Any disagreements were resolved by two other researchers. This rigorous approach aimed to minimize potential bias in the review process. The review included a comprehensive examination of the literature, including randomized trials that evaluated the effects of antioxidant therapy on individuals with diabetic nephropathy.
Types of antioxidants
Various antioxidant groups were used in the studies (see Table 3), including flavonoids, polyphenols, vitamins, antioxidant minerals, and fatty acids. The flavonoid group consisted of silymarin, baicalin, and epigallocatechin gallate. The polyphenol group used were curcumin and resveratrol. Vitamin E (tocotrienol) and B were also used as antioxidant vitamins, while selenium was used as an antioxidant mineral. Several studies used α-lipoic acid as an antioxidant fatty acid. Furthermore, the studies included in this systematic review used both single and combined antioxidant agents. The single antioxidant agents used were silymarin, baicalin, epigallocatechin gallate, vitamin E, selenium, curcumin, α-lipoic acid, and tocotrienol-rich vitamin E. Meanwhile, the combined antioxidant agents were antioxidant α-lipoic acid with vitamin B6 and resveratrol with losartan. All of these antioxidant compounds were evaluated for their ability to inhibit the development of diabetic nephropathy.
Table 3.
Effect size
|
Compound (group)
|
Author
|
Outcome
|
Result
|
Conclusion
|
|
Single
|
|
|
|
|
| Silymarin (flavonoid) |
Fallahzadeh et al |
UACR decreased |
Placebo: - 219 mg/g
Intervention: -566 mg/g
Significantly different between the two groups |
Silymarin reduced UACR, TNF-α, and MDA levels significantly |
| Urinary and serum TNF-α decreased |
Placebo
Serum: -1.27 pg/mg
Urinary: -0.78 pg/mg
Intervention
Serum: -2.39 pg/mg
Urinary: -3.45 pg/mg
Significantly different between the two groups |
| Urinary and serum TGF-β decreased |
Placebo
Serum: 0.64 ng/mL
Urinary: -1.38pg/mg
Intervention
Serum: -0.57 ng/mL
Urinary: -3.97 pg/mg
No significant difference between the two groups |
| Urinary and serum MDA decreased |
Placebo
Serum: 0.99 µmol/L
Urinary: 0.4 nmol/mg
Intervention
Serum: -3.43 µmol/L
Urinary: -1.5 nmol/mg
Significantly different between the two groups |
| Voroneanu et al |
eGFR decreased |
Placebo: -11.5 ml/min/1.73 m2
Intervention: -13.46 ml/min/1.73 m2
No significant difference between the two groups |
| No significant changes in proteinuria |
Placebo: -0.03 g/day
Intervention: + 0.24 g/day
No significant difference between the two groups |
Baicalin
(flavonoid) |
Yanget al |
Urinary microalbumin decreased |
Placebo: -28.1 mg/day
Intervention: -135.1 mg/day
Significantly different between the two groups |
Baicalin decreased urinary microalbumin, UAER, and NF-κB significantly
Baicalin increased SOD and GSH-Px significantly |
| Urinary albumin excretion rate (UAER) decreased. |
Placebo: -4.9µg/min
Intervention: -18.9µg/min
Significantly different between the two groups |
| SOD increased |
Placebo: + 1 U/ml
Intervention: + 6.1U/ml
Significantly different between the two groups |
| GSH-Px increased |
Placebo: + 1.8 U/l
Intervention: + 5.9 U/l
Significantly different between the two groups |
| TGF-β decreased |
Placebo: -6.8 pg/ml
Intervention: -6.7 pg/ml
No significant difference between the two groups |
| NF-κB decreased |
Placebo: -2.2 %
Intervention: -3.9 %
Significantly different between the two groups |
| Epigallocatechin gallate (flavonoid) |
Borgeset al |
UACR decreased |
Placebo: -25 mg/g
Intervention: -77 mg/g
Significantly different between the two groups |
Epigallocatechin gallate significantly decreased UACR, DKK-1, and TNF-α |
| eGFR decreased |
Placebo: -0.01 ml/min/1.73 m2
Intervention: -0.07 ml/min/1.73 m2
No significant difference between the two groups |
| Plasma DKK-1 decreased |
Placebo: -0.01 pg/ml
Intervention: -0.39 pg/ml
Significantly different between the two groups |
| Serum TNF-α decreased |
Placebo: 0.06 pg/ml
Intervention: -0.16 pg/ml
Significantly different between the two groups |
| CRP increased |
Placebo: + 0.15
Intervention: 0.00
No significant difference between the two groups |
| Curcumin (polyphenol) |
Jimenez-Osorio et al |
Serum creatinine increased |
Placebo: + 0.3
Intervention: + 0.1
No significant difference between the two groups |
Curcumin significantly attenuated proteinuria and plasma MDA, and improved antioxidant activity |
| eGFR decreased |
Placebo: -2.5 ml/min/1.73m2
Intervention: -0.8 ml/min/1.73m2
Significantly different between the two groups |
| Proteinuria decreased |
Placebo: -1 g/day
Intervention: -0.2 g/day
Significantly different between the two groups |
| Plasma MDA decreased |
Placebo: -1 nM
Intervention: -0.7 nM.
Significantly different between the two groups |
| Antioxidant activity increased |
Placebo: not mentioned (histogram)
Intervention: not mentioned (histogram)
Significantly different between the two groups |
Vitamin E
(vitamin) |
Khatami et al |
Urine creatinine decreased |
Placebo: -0.9 mg/dL
Intervention: 0.00 mg/dL
No significant difference between the two groups |
Vitamin E significantly decreased TNF-α, MMP-2, MMP-9, MDA, AGEs, total cholesterol, and LDL
Vitamin E significantly increased total antioxidant capacity, plasma glutathione, and HDL |
| TNF-α decreased |
Placebo: + 5.6 ng/L
Intervention: -35.4 ng/L
Significantly different between the two groups |
| MMP-2 decreased |
Placebo: 60.4 ng/mL
Intervention: -556.7 ng/mL
Significantly different between the two groups |
| MMP-9 decreased |
Placebo: + 225.7 ng/L
Intervention: -1461.5 ng/L
Significantly different between the two groups |
| MDA decreased |
Placebo: -0.3 μmol/L
Intervention: -0.9μmol/L
Significantly different between the two groups |
| AGEs decreased |
Placebo: + 177.3AU
Intervention: -1832.2 AU
Significantly different between the two groups |
| Aghadavod et al |
Total antioxidant capacity increased |
Placebo: + 37.0 mmol/l
Intervention: + 84.9 mmol/l
Significantly different between the two groups |
| Plasma glutathione increased |
Placebo: -42.8 μmol/L
Intervention: + 72.7 μmol/L
Significantly different between the two groups |
| Total cholesterol decreased |
Placebo: -0.8 mg/dl
Intervention: -14.3 mg/dl
Significantly different between the two groups |
| LDL decreased |
Placebo: + 0.1 mg/dl
Intervention: -16.4 mg/dl
Significantly different between the two groups |
| HDL increased |
Placebo: -2.1 mg/dl
Intervention: + 1.4 mg/dl
Significantly different between the two groups |
| Tocotrienol-rich vitamin E (Vitamin) |
Tan et al |
UACR decreased |
Placebo: -61.77 mg/mmol
Intervention: + 19.03 mg/mmol
Significantly different between the two groups |
Tocotrienol-rich vitamin E significantly decreased UACR, creatinine, BUN, and MDA
Tocotrienol-rich vitamin E significantly increased eGFR |
| Serum creatinine decreased |
Placebo: -5.54 µmol/l
Intervention: -0.44 µmol/l
Significantly different between the two groups |
| BUN decreased |
Placebo: -1.07 mmol/l
Intervention: -0.96 mmol/l
No significant difference between the two groups |
| eGFR increased |
Placebo: + 17.39 ml/min/1.73m2
Intervention: + 0.47 ml/min/1.73m2
Significantly different between the two groups |
| AGEs decreased |
Placebo: -28.54 µg/mL
Intervention: - 46.28 µg/mL
Significantly different between the two groups |
| Tan et al |
Serum creatinine decreased |
Placebo: + 5.4 µmol/l
Intervention: –3.3 µmol/l
Significantly different between the two group |
| eGFR increased |
Placebo: -2.9 ml/min/1.73m2
Intervention: 1.5 ml/min/1.73m2
Significantly different between the two groups |
| VCAM-1 increased |
Placebo: + 30.0 ng/mL
Intervention: + 4.1 ng/mL
Significantly different between the two groups |
| MDA decreased |
Placebo: -232 ng/mL
Intervention: -102 ng/mL
No significant difference between the two groups |
| HbA1c decreased |
Placebo: -0.38 %
Intervention: -0.60 %
No significant difference between the two group |
| Koay et al |
UACR increased |
Placebo: + 0.56 mg/mmol
Intervention: + 1.78 mg/mmol
No significant difference between the two groups |
| Serum creatinine decreased |
Placebo: + 0.84 µmol/l
Intervention: -7.85 µmol/l
Significantly different between the two groups |
| eGFR increased |
Placebo: = 1.45 ml/min/1.73m2
Intervention: + 4.83ml/min/1.73m2
Significantly different between the two groups |
| TGF-β decreased |
Placebo: -8.4 ng/mL
Intervention: -4.3 ng/mL
No significant difference between the two groups |
| Selenium (Mineral antioxidant) |
Bahmani et al |
Insulin serum decreased |
Placebo: 0.5μIU/ml
Intervention: -3.1μIU/ml
Significantly different between the two groups |
Selenium significantly decreased MMP-2, MDA, and hs-CRP
Selenium significantly increased glutathione peroxidase and total antioxidant capacity |
| HOMA-IR decreased |
Placebo: 0.1
Intervention: -0.9
Significantly different between the two groups |
| HOMA-B decreased |
Placebo: + 2.3
Intervention: -11.3
Significantly different between the two groups |
| Glutathione peroxidase increased |
Placebo: -27.7 U/ml
Intervention: + 2.3 U/ml
Significantly different between the two groups |
| Bahmani et al |
TGF-β decreased |
Placebo: -70.9 ng/L
Intervention: -77.7 ng/L
No significant difference between the two groups |
| MMP-2 decreased |
Placebo: + 76.0 ng/mL
Intervention: -612.3 ng/mL
Significantly different between the two groups |
| NO increased |
Placebo: + 4.3 μmol/L
Intervention: + 47.9 μmol/L
No significant difference between the two groups |
| MDA decreased |
Placebo: + 0.4 µmol/l
Intervention: -0.1 µmol/l
Significantly different between the two groups |
| AGEs decreased |
Placebo: + 571.0 AU/g protein
Intervention -100.0 AU/g protein
No significant difference between the two groups |
| Total antioxidant capacity increased |
Placebo: + 15.8 mmol/l
Intervention: + 174.9 mmol/l
Significantly different between the two groups |
| Plasma glutathione increased |
Placebo: -20.7 µmol/l
Intervention: + 241.5 µmol/l
No significant difference between the two groups |
| hs-CRP decreased |
Placebo: -135.3 ng/mL
Intervention: -1069.2 ng/mL
Significantly different between the two groups |
|
Combination
|
|
|
|
|
Lipoic acid + vit B6
(alpha-lipoic acid and vitamin B) |
Noori et al |
Urinary creatinine decreased |
Placebo: 0.0 mg/dL
Intervention: 0.02 mg/dL
Significantly different between the two groups |
Alpha-lipoic acid and vitamin B significantly decreased urinary creatinine, microalbumin, and MDA
Alpha-lipoic acid and vitamin B significantly increased NO |
| Urinary microalbumin decreased |
Placebo: + 27.0 mg/g creatinine
Intervention: -74.0 mg/g creatinine
Significantly different between the two groups |
| NO increased |
Placebo: -0.3μmol/L
Intervention: 1.0 μmol/L
Significantly different between the two groups |
| MDA decreased |
Placebo: 0.0 μmol/L
Intervention: -1.2 μmol/L
Significantly different between the two groups |
| Resveratrol + losartan (Polyphenol) |
Sattarinezhad et al |
UACR decreased |
Placebo: + 25.3 mg/gcreatinine
Intervention: -46.4 mg/gcreatinine
Significantly different between the two groups |
Resveratrol and losartan significantly decreased UACR, serum creatinine, and MDA
Resveratrol and losartan significantly increased NO, GSH-Px, SOD activity, and catalase activity |
| eGFR increased |
Placebo: -4.0 ml/min/1.73 m2
Intervention: + 1.7 ml/min/1.73 m2
No significant difference between the two groups |
| Serum creatinine decreased |
Placebo: + 0.1 mg/dL
Intervention: -0.0 mg/dL
No significant difference between the two groups |
| MDA decreased |
Placebo: + 0.9 nmol/ml
Intervention: -0.4 nmol/ml
Significantly different between the two groups |
| NO increased |
Placebo: -0.5 µmol/L
Intervention: + 4.4 µmol/L
Significantly different between the two groups |
| GSH-Px increased |
Placebo: -5.7 U/L
Intervention: + 13.4 U/L
Significantly different between the two groups |
| SOD activity increased |
Placebo: -4.2 U/L
Intervention: 4.8 U/L
Significantly different between the two groups |
| Catalase increased |
Placebo: -2.7 U/L
Intervention: + 5.9 U/L
Significantly different between the two groups |
| MDA decreased |
Placebo: + 0.9 nmol/mL
Intervention: -0.4 nmol/mL
Significantly different between two groups |
Abbreviations: UACR: urine albumin-creatinine ratio; UAER: urinary albumin excretion rate; MDA: malondialdehyde; TNF-α: tumor necrosis factor alpha; TGF-β: transforming growth factor beta; eGFR: estimated glomerular filtration rate; NO: nitrite oxide; SOD: super oxide dismutase; AGEs: advanced glycation end products; Hs-CRP: high sensitivity C-reactive protein; GSH-Px: plasma glutathione peroxidase; HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; HOMA-B: Homeostatic Model Assessment Beta; MPO: myeloperoxidase.
The effect of antioxidants on patients with diabetic nephropathy
Oxidative stress plays a crucial role in pathophysiological development of diabetes mellitus. Prolonged hyperglycemia results in the excessive production of ROS, leading to a reduction in endogenous antioxidants, such as glutathione and superoxide dismutase. Excessive production of ROS is implicated in the alteration of renal metabolic pathways and is associated with the development of diabetic nephropathy, resulting in inflammation, fibrosis, and endothelial dysfunction.4
Malondialdehyde (MDA) and advanced glycation end products (AGEs) are markers of oxidative stress in cells. Six studies reported significant reductions in MDA levels in patients with diabetic nephropathy who received antioxidant therapy, including silymarin, curcumin, vitamin E, tocotrienol-rich vitamin E, selenium, and a combination of lipoic acid with B vitamins.13-20 Three studies also reported reductions in AGEs with antioxidant therapy. Treatment with vitamin E and tocotrienol-rich vitamin E resulted in a significant reduction,13,21 while treatment with selenium showed a reduction, but no significant difference between the intervention and placebo groups.15
Transforming growth factor beta (TGF-β) is a growth factor that contributes to the accumulation of extracellular matrix (ECM). It induces an increase in ECM production and stability by suppressing its degradation, a process that leads to the development of glomerulosclerosis and interstitial fibrosis in diabetic nephropathy. Four studies reported that antioxidant therapy with silymarin, baicalin, tocotrienol-rich vitamin E, and selenium reduced TGF-β levels in patients with diabetic nephropathy, but the reduction was not significantly different from placebo.11,16,22,23
Tumor necrosis factor alpha (TNF-α) is a proinflammatory cytokine that is essential in the pathogenesis and clinical outcome of diabetic nephropathy. Three studies reported that antioxidant therapy with silymarin, epigallocatechin gallate, and vitamin E helped reduce TNF-α levels. The results showed a significant difference between the intervention and placebo groups.11,13,24
Matrix metalloproteinase-2 (MMP-2) and matrix metalloproteinase-9 (MMP-9) are inflammatory markers involved in the pathophysiology of diabetic nephropathy. Two studies reported that vitamin and selenium treatment could reduce the levels of MMP-2 and MMP-9 in patients with diabetic nephropathy. Moreover, the reductions differed significantly between the intervention and placebo groups.13,16
Based on the analysis, four studies reported that antioxidant treatment reduced UACR levels in 101 patients.11,19,21,22 However, one study found no significant decrease in UACR levels.21
Furthermore, three studies showed a decrease in the levels of proteinuria, albuminuria, and microalbuminuria. A significant decrease in proteinuria levels was observed with curcumin intervention.12 However, another study found no significant changes in proteinuria levels with silymarin intervention.25 On the other hand, a significant decrease in albuminuria and microalbuminuria was observed with a single baicalin and a combination of lipoic acid and B vitamins.14,17
Antioxidant treatment affects creatinine levels. Six studies reported that antioxidants, including vitamin E, tocotrienol-rich vitamin E, a combination of lipoic acid and B vitamins, and a combination of resveratrol and losartan, could reduce creatinine levels.14,16,17,21 However, two of these studies using vitamin E and a combination of resveratrol and losartan as antioxidant treatments found that the reductions were insignificant.13,26 In other words, curcumin as antioxidants were found to have an effect on the increase in creatinine, but not significant.12
The effect of antioxidant therapy on increasing estimated glomerular filtration rate (eGFR) levels was investigated in six studies. The tocotrienol-rich vitamin E and the combination of resveratrol with losartan were found to have a positive effect on increasing eGFR levels in patients. The intervention group showed a significant increase in eGFR levels with tocotrienol-rich vitamin E treatment compared to the placebo group. However, the intervention group did not show a significant difference in eGFR levels with the combination of resveratrol with losartan compared to the placebo group.14,19,21,24 On the contrary, eGFR levels decreased with curcumin, silymarin and epigallocatechin gallate antioxidant therapy.22,23
Discussion
This study aims to investigate the types of antioxidant agents, either single or combined, that inhibit the development of diabetic nephropathy and the effectiveness of antioxidant agent interventions to repair kidney structure and function. This study observed changes in the levels of UACR, creatinine, AGE production, inflammatory and fibrosis markers, and antioxidants in patients received antioxidant therapy and those who did not. The results of this study suggested that antioxidant therapy can improve kidney function by reducing oxidative stress levels through increased antioxidant capacity and can affect various markers of inflammation and fibrosis.
Antioxidant reduces oxidative stress induced by hyperglycemia
Hyperglycemia induces oxidative stress, leading to an increase in the amount of ROS in cells due to the diversion of alternative glucose pathways and its metabolites, such as fructose-1,6-bisphosphate and glyceraldehyde-3-phosphate. For example, increased sorbitol levels induce cell damage and reduces the antioxidant glutathione. Meanwhile, increased AGEs modify lipids, proteins, and nucleic acids. In addition, AGEs trigger the formation of intracellular free radicals and activate proinflammatory factors such as interleukin (IL)-1 and TGF-β. Furthermore, increased protein kinase C and angiotensin II exacerbate conditions caused by hyperglycemia.27
The continuous increase in ROS in cells creates an imbalance between ROS and endogenous antioxidants. As a result, numerous studies have investigated the potential of external antioxidants to reduce ROS and oxidative stress in cells. Antioxidants derived from medicinal plants are promising candidates for reducing ROS activity in hyperglycemia. Several studies reviewed in this study used various groups of antioxidant compounds, including flavonoids, polyphenols, vitamins, antioxidant minerals, and fatty acids. These compounds are known to reduce free radicals (ROS). In addition, the studies examine MDA and AGEs as markers of oxidative stress. MDA is a product of lipid peroxidation that indicates the interaction between oxygen molecules and polyunsaturated fatty acids.28 Polyunsaturated fatty acids are present in cell membranes, making them highly susceptible to peroxidation caused by ROS. Previous studies have shown that patients with diabetes mellitus have increased MDA levels.29,30 Silymarin, curcumin, vitamin E, tocotrienol-rich vitamin E, selenium, and a combination of alpha-lipoic acid (ALA) with vitamin B have been found to significantly reduce serum MDA levels. Curcumin works by inhibiting MDA production through the sirtuin 1, 2, and 3 pathways.31
Furthermore, curcumin may counteract the heightened effects of AGEs caused by ROS by interfering with the activation of leptin signaling by AGEs.32 Curcumin can also inhibit ROS and increase the expression of antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH). Previous studies have reported that vitamin E can reduce oxidative stress in hemodialysis patients.33 Vitamin E and selenium have been shown to play an essential role in the interaction between enzymatic and non-enzymatic pathways to prevent lipid peroxidation of circulating lipoproteins and cell membranes.34-36 They also prevent the glycation of hemoglobin and the accumulation of AGEs.37 The administration of vitamin E affects the balance of ROS by increasing antioxidants, leading to a decrease in MDA levels.38 The ability of ALA to reduce oxidative stress contributes to the reduction of AGEs and MDA production in patients with diabetic nephropathy. This ability is associated with its role as a scavenger of various types of ROS. Alpha-lipoic acid has the potential to be a scavenger of hydroxyl radicals (OH), hypochlorous acid, and singlet oxygen.39 Studies have also supported the effectiveness of ALA in improving endothelial function. ALA has shown the ability to improve endothelial function by reducing plasma MDA and aortic tissue levels in patients with diabetes.40 It is an agent that inhibits endothelial dysfunction and improves insulin sensitivity in skeletal muscle and liver.41,42
Antioxidant capacity may be associated with the activation of antioxidant enzymes. For example, SOD and glutathione peroxidase-1 (GPx) are antioxidant enzymes involved in the intrinsic defense mechanism that protects cells when exposed to free radicals, namely ROS.43 The alleviation of kidney disease depends significantly on the essential role played by three SOD isoforms.44 Superoxide dismutase 1 (SOD1) reduces ROS and improves kidney function by decreasing the levels of TNF-α and IL-1 in kidney failure.45 Meanwhile, superoxide dismutase 2 (SOD2) is associated with improved kidney function and tubular necrosis. Finally, superoxide dismutase 3 (SOD3) results in a significant increase in both glomerular and tubular areas.46
Plant extract antioxidants, such as baicalin, have been shown to alleviate oxidative stress in conditions such as diabetic nephropathy.47 The administration of baicalin improved the endogenous SOD and plasma glutathione peroxidase (GPx). Baicalin contains multiple -OH groups that are susceptible to oxidation by ROS. This property may mitigate the detrimental effects of ROS on other cellular components and to partially alleviate oxidative stress. Baicalin exhibits dose-dependent effectiveness in increasing total antioxidant capacity and superoxide anion scavenging activity. This suggests that baicalin may neutralize ROS and increase SOD.48 As antioxidant activity increases, kidney function appears to improve, as evidenced by reduced levels of microalbuminuria and UACR. By reducing oxidative stress and mitochondrial ROS, GPx1 improves glomerulosclerosis.49 The expression of GPx1 and GPx4 isoforms is primarily detected in podocytes and mesangial cells.50 This is consistent with the results indicating reduced albuminuria following treatment with the flavonoid group, namely baicalin and silymarin.17,20
Role of antioxidants in inflammation
Kidney damage begins with podocyte injury, leading to tubular epithelium and interstitial damage characterized by proteinuria. Proteins such as albumin, transferrin, complement components, and lipoproteins, that pass through the tubules cause lysosomal rupture and energy depletion in tubular cells, resulting in tubulointerstitial injury.51 Complement components activate tubular epithelial cells, inducing a proinflammatory state by releasing arachidonic acid products and proinflammatory cytokines such as IL-6, IL-1β, and TNF-α.52 Albumin induces the release of TNF-α, IL-8, monocyte chemotactic protein-1 (MCP-1), and platelet-derived growth factor (PDGF) from epithelial cells, which can lead to interstitial inflammation and fibrosis.53,54 Meanwhile, cytokines play a critical role in the development of tubulointerstitial injury.
Based on the above description, several biomarkers are involved in the pathology of diabetic kidney disease, including creatinine, urea, uric acid, lipid peroxidation, lactate dehydrogenase (LDH), IL-6, TNF-α, prostaglandin E₂ (PGE-2), inducible nitric oxide synthase (iNOS), and reduced levels of antioxidant enzymes.55 However, TNF-α, TGF-β, and cyclooxygenase-2 (COX-2) are the most potent biomarkers, as their overexpression is responsible for causing kidney pathology in diabetic patients.56,57 Tumor necrosis factor-alpha (TNF-α) is a pro-inflammatory cytokine that contributes to microvascular complications in diabetes, including nephropathy.58 Previous studies have shown that TNF-α has cytotoxic effects on glomerular, mesangial, and epithelial cells, resulting in direct kidney damage.59 The high expression of TNF-α and COX-2 in the kidneys is induced by the activation of the nuclear transcription factor nuclear factor kappa B (NF-κB).60
Flavonoids, such as silymarin, baicalin, epigallocatechin gallate, and vitamin E, have been shown to reverse the process of renal fibrosis by inhibiting inflammation. Silymarin, baicalin, epigallocatechin gallate, and vitamin E may reduce the expression of TNF-α by inhibiting the NF-κB pathway.61,62 For instance, Morishima et al (2010) found that silymarin inhibits the proliferation and proinflammatory cytokine secretion of the T cells. Silymarin effectively blocks the activation of NF-kB. Similarly, baicalin inhibits inflammation by targeting the NF-κB signaling pathway.60,63 This inhibitory effect is mediated by preventing the phosphorylation and degradation of IκBα.59,64
Role of antioxidant in fibrosis
Chronic diabetic nephropathy manifests as renal fibrosis, which is characterized by glomerulosclerosis, tubulointerstitial fibrosis, inflammatory infiltration, and loss of renal parenchyma. This loss is characterized by tubular atrophy, capillary loss, and podocyte thinning. At the cellular level, mesangial cells and fibroblasts are activated, tubular cells undergo epithelial-mesenchymal transition (EMT), monocyte/macrophage and T cells infiltrate, and cells undergo apoptosis.65
Transforming growth factor-β (TGF- β) plays a crucial role in the development of glomerulosclerosis and interstitial fibrosis observed in end-stage renal disease (ESRD).66 TGF-β enhances the expression of connective tissue growth factor (CTGF) via Smad activation in fibroblasts, leading to the development of fibrosis both in vitro and in vivo.67 The activation of the TGF-β signaling pathway can induce dedifferentiation, apoptosis, necrosis, and ROS in tubular epithelial cells, followed by a decline in kidney function and infiltration of inflammatory cells.68 Transforming growth factor-β1 (TGF- β1), a specific isoform of TGF-β, regulates the activity of other transcription factors involved in ECM synthesis. The direct stimulation of ECM transcription by TGF-β1 contributes to the accumulation of ECM proteins observed in fibrotic conditions, including diabetic nephropathy.69
Silymarin, baicalin, tocotrienol-rich vitamin E, and selenium did not significantly reduce TGF-β levels in the intervention group compared to the placebo group. However, reducing TGF-β levels is crucial in inhibiting the fibrotic process in the kidney. The results showed the impact of the bioactive compounds on the development of renal fibrosis, specifically by affecting the TGF-β signaling pathways. These suggested that the bioactive compounds may have potential in treating renal fibrosis. This could be used as a preventive approach for individuals in the early stages of diabetes mellitus, although further research is necessary if the intervention period is extended.
Previous research has shown that TGF-β1 plays an important role in the development and progression of diabetic nephropathy. TGF-β1 is an essential protein that binds to the cell membrane in the TGF-β signaling pathway facilitated by Smads. In the diabetic nephropathy rats, The TGF-β/Smad signaling pathway is specifically activated in the kidneys of the DN model rats, resulting in increased expression of TGF-β1 and Smad2/3 proteins, while the expression of Smad6/7 is decreased. The administration of silymarin decreases the expression of Smad2/3 and TGF-β1 proteins and increases the expression of Smad6/7. In diabetic nephropathy rats, silymarin was found to have a protective effect on kidney tissues by regulating the JAK2/STAT3/SOCS1 and Smad/TGF-β signaling pathways. This regulation resulted in decreased expression of inflammatory factors and renal fibrosis in the respective pathways.70
In addition, baicalin has been shown to have potential in alleviating diabetic nephropathy by increasing the expression of sirtuin 1 in podocytes exposed to high glucose and inhibiting the NF-κB pathway. Baicalin prevents renal fibrosis in diabetic nephropathy mice by increasing the levels of miR-124 and blocking the downstream TLR4/NF-κB pathway.60 It also inhibits inflammation by targeting the NF-κB signaling pathway and prevents extracellular matrix (ECM) accumulation through the TGF-β/Smad3 pathway.20
The ECM is crucial for providing structural support to tissues and organs, including the kidneys, and maintaining normal physiological processes. Excessive accumulation of ECM components is a hallmark of the morphological features of diabetic nephropathy. MMPs are essential for the turnover and remodeling of the ECM, as they can break down nearly all of its components. This activity is tightly regulated to maintain normal tissue homeostasis by balancing the synthesis of ECM proteins.67 The accumulation of ECM is caused by an imbalance between the synthesis and degradation of matrix proteins. In diabetic nephropathy, the production of matrix proteins is increased, but their degradation is decreased. This is primarily mediated by MMPs. Matrix metalloproteinases-2 (MMP-2) plays a dual role in promoting the transition of renal tubular epithelium to the myofibroblastic phenotype, which marks the initiation of renal interstitial fibrosis in diabetic nephropathy. In this process, MMP-2 is an essential and self-sufficient factor.
On the other hand, the expression of matrix metalloproteinases-9 increases in patients with diabetic nephropathy according to the degree of albuminuria.71 These proteases modify the components of the basal lamina, resulting in cell death.72 Selenium appears to have an effect on the reduction of MMP-2 secretion in patients with diabetic nephropathy.16 Previous studies have shown that this is related to the activation of ERK1/2 pathway, which increases MMP-2 secretion. Selenium is believed to inhibit the activation of the ERK1/2 pathway, thereby reducing MMP-2 secretion.73 Additionally, TNF-α affects the increase in ERK1/2 phosphorylation, which affects the secretion of MMP-1, MMP-3, and MMP-9. The effects of TNF-α can be eliminated by a mitogen-activated protein kinase (MEK) inhibitor in the mitogen-activated protein kinase (MAPK) pathway. Moreover, vitamin E appears to reduce TNF-α expression and significantly decreases MMP-2 and MMP-9 secretion.13,71
The enhancement of the extracellular matrix encompasses the elevation of type 4 collagen and α-smooth muscle actin within mesangial cells. Inflammatory processes can have a detrimental effect on the body. For example, glomerulosclerosis can lead to an increase in the mesangial extracellular matrix, resulting in a subsequent decrease in the glomerular filtration rate (eGFR).74 Albuminuria is an indication of impaired glomerular filtration barrier or tubular reabsorption.75 In addition, tocotrienol-enriched vitamin E with the simultaneous use of resveratrol and losartan increases eGFR levels. However, curcumin, silymarin, and epigallocatechin gallate antioxidants did not show a similar effect.
Interconnectivity
The interconnectivity of diabetic nephropathy refers to the complex relationship among different factors and mechanisms contributing to the development and progression of kidney damage in individuals with diabetes. It involves a network of interconnected pathways and processes that interact and influence each other, such as hyperglycemia, oxidative stress, inflammation, fibrosis, and apoptosis.
Persistent high blood glucose levels in diabetes lead to the activation of several pathways, including the polyol pathway, AGE formation, protein kinase C activation, and the hexosamine pathway. These pathways interact with each other and contribute to the development of kidney damage. Hyperglycemia and other metabolic disorders in diabetes promote the production of ROS and oxidative stress. Oxidative stress, in turn, can cause cellular damage and inflammation, contributing to the progression of diabetic nephropathy. Chronic low-grade inflammation is a hallmark of diabetic nephropathy. Inflammatory cytokines, chemokines, and immune cells play a role in the inflammatory response, exacerbating kidney damage.
The renin-angiotensin-aldosterone system (RAAS) regulates blood pressure and fluid balance. In diabetes, dysregulation of the renin-angiotensin system (RAS) can lead to increased production of angiotensin II, a potent vasoconstrictor and proinflammatory mediator. Angiotensin II contributes to kidney injury by promoting inflammation, oxidative stress, and fibrosis.76 However, no clinical studies have been conducted on this topic. Therefore, further research is needed to determine the potential of antioxidants in the regulation of the RAS.
Kidney fibrosis is a common characteristic of progressive kidney damage characterized by excessive scarring and deposition of extracellular matrix proteins occur in the kidney tissues. This condition is a hallmark of diabetic nephropathy and disrupts the normal kidney structure, impairing its function. In diabetic kidney disease (DKD), prolonged exposure to high blood glucose levels and other metabolic diseases can cause cellular dysfunction and kidney damage. Apoptosis of renal cells, including tubular epithelial cells, podocytes, and mesangial cells, is observed in DKD. This apoptotic cell death contributes to the loss of functional kidney cells and the progression of kidney damage. Multiple factors, such as oxidative stress, inflammation, and activation of various signaling pathways, are believed to contribute to the initiation and progression of apoptosis in DKD. Although studies have been conducted on the conditions that cause fibrosis, few observations have been made regarding apoptosis markers. Research in this area is necessary to enable the development of treatments that target the apoptotic pathways.
Potential clinical significance
This study suggested that antioxidant therapy may be effective in reducing the risk of ESRD. Although the available data from the studies are limited, evidence suggested that antioxidant therapy may reduce oxidative stress markers and inflammation associated with the development of diabetic nephropathy. However, its effectiveness in preventing the progression of fibrosis remains unclear. Among all antioxidants, vitamin E has been shown to lower serum creatinine and improve eGFR, but it does not significantly reduce TGF-β levels, a fibrosis marker. This may be due to the duration of diabetes mellitus in the patients. The relationship between the use of antioxidant therapy and the progression of diabetic nephropathy needs to be clarified. While antioxidants may have a preventive effect, they may not be effective as a treatment. Adverse events associated with antioxidant therapy have been minimal. No evidence that antioxidants are harmful to individuals with diabetic nephropathy has been found.
Limitations
The limitations of this review lie in the utilization of only three databases during the article search. The articles primarily focus on RCTs. However, the heterogeneity of antioxidant agent types, doses, and duration presents challenges in comparing the outcomes, particularly the effectiveness of antioxidants on expected outcomes. Out of the selected studies, ten studies stated that the limitations of their research were the small sample size and the insufficient duration of antioxidant administration to the patients. Additionally, six studies noted the lack of laboratory-based outcomes, such as the control of various biomarkers of inflammation, oxidative stress, and several proteins associated with the development of diabetic nephropathy, which was attributed to financial constraints. One study found that patient compliance was an uncontrollable factor that affected the results. Another study used a single-blind design, which allowed laboratory measurements and clinical assessments to be independently assessed by the researchers. Our analyses were based on published data, thereby limiting our ability to thoroughly explore the potential advantages and disadvantages of antioxidant therapy in individuals with diabetic nephropathy. Further research is needed to validate the reproducibility and generalizability.
Currently, there are no theories addressing the potential side effects of antioxidants on patients with diabetic nephropathy. Ongoing research focuses on the use of antioxidants for diabetic nephropathy due to increased oxidative stress in hyperglycemic conditions. Studies have shown that daily intake of flavonoid groups, particularly flavonols (68 mg) and isoflavones (20–240 mg), does not result in adverse effects. Silymarin has been confirmed to be safe for human consumption at therapeutic doses. A high dosage of 700 mg three times daily for 24 weeks was found to be well-tolerated.77
Concerns have been raised about the use of synthetic vitamin E in antioxidant therapy. The maximum tolerated daily dose is 300 mg, while doses exceeding 2 g/day are considered toxic in the United States. Long-term intake at doses over 240–480 mg/day may lead to adverse effects, which typically subside shortly after reducing the dose.78 Resveratrol is well-tolerated in experimental models with no significant adverse effects. It has been reported that a safe dose for a person weighing 60 kg is 450 mg/day.79
Conclusion
This systematic review identifies flavonoids, vitamins, fatty acids, and antioxidant minerals as potential agents for improving diabetic nephropathy. The results of this study suggest that these antioxidant agents may reduce oxidative stress and inflammation markers associated with the development of diabetic nephropathy. In addition, various antioxidants may contribute to reducing fibrosis markers, but their effect does not appear to be statistically significant.
Recommendations
The studies analyzed in this systematic review are currently restricted to research on the benefits of antioxidants in improving kidney function, reducing oxidative stress markers, and repairing inflammation and fibrosis. Other aspects related to the role of antioxidants in regulating RAS and apoptosis, which are also crucial for the development of diabetic nephropathy, have not yet been investigated. Understanding the interplay between these factors is essential to develop effective strategies for the prevention and management of diabetic nephropathy. Targeting multiple pathways may be necessary to break the vicious cycle of kidney disease and halt its progression. Further research with larger sample sizes and longer duration is considered necessary. The administration of antioxidants should begin early in the onset of diabetes mellitus, as oxidative stress increases from the early stages. Ideally, antioxidants should be used for prevention rather than therapy. Furthermore, research on potential side effects of antioxidant therapy is necessary.
Acknowledgements
The authors are responsible for the contents of this manuscript.
Competing Interests
The authors have no conflicts of interest to declare.
Ethical Statement
Since this is a literature review, no ethical approval was required.
Supplementary files
Supplementary file 1. Search strategy
(pdf)
References
- Leutner M, Haug N, Bellach L, Dervic E, Kautzky A, Klimek P. Risk of typical diabetes-associated complications in different clusters of diabetic patients: analysis of nine risk factors. J Pers Med 2021; 11:328. doi: 10.3390/jpm11050328 [Crossref] [ Google Scholar]
- International Diabetes Federation (IDF). IDF Diabetes Atlas. 10th ed. IDF; 2021.
- Farah RI, Al-Sabbagh MQ, Momani MS, Albtoosh A, Arabiat M, Abdulraheem AM. Diabetic kidney disease in patients with type 2 diabetes mellitus: a cross-sectional study. BMC Nephrol 2021; 22:223. doi: 10.1186/s12882-021-02429-4 [Crossref] [ Google Scholar]
- Thomas MC, Brownlee M, Susztak K, Sharma K, Jandeleit-Dahm KA, Zoungas S. Diabetic kidney disease. Nat Rev Dis Primers 2015; 1:15018. doi: 10.1038/nrdp.2015.18 [Crossref] [ Google Scholar]
- Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol 2018; 14:88-98. doi: 10.1038/nrendo.2017.151 [Crossref] [ Google Scholar]
- Zsom L, Zsom M, Salim SA, Fülöp T. Estimated glomerular filtration rate in chronic kidney disease: a critical review of estimate-based predictions of individual outcomes in kidney disease. Toxins (Basel) 2022; 14:127. doi: 10.3390/toxins14020127 [Crossref] [ Google Scholar]
- Miranda-Díaz AG, Pazarín-Villaseñor L, Yanowsky-Escatell FG, Andrade-Sierra J. Oxidative stress in diabetic nephropathy with early chronic kidney disease. J Diabetes Res 2016; 2016:7047238. doi: 10.1155/2016/7047238 [Crossref] [ Google Scholar]
- Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res 2010; 107:1058-70. doi: 10.1161/circresaha.110.223545 [Crossref] [ Google Scholar]
- Schaffer SW, Jong CJ, Mozaffari M. Role of oxidative stress in diabetes-mediated vascular dysfunction: unifying hypothesis of diabetes revisited. Vascul Pharmacol 2012. 57: 139-49. 10.1016/j.vph.2012.03.005.
- Bolignano D, Cernaro V, Gembillo G, Baggetta R, Buemi M, D'Arrigo G. Antioxidant agents for delaying diabetic kidney disease progression: a systematic review and meta-analysis. PLoS One 2017; 12:e0178699. doi: 10.1371/journal.pone.0178699 [Crossref] [ Google Scholar]
- Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ 2021; 372:n160. doi: 10.1136/bmj.n160 [Crossref] [ Google Scholar]
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 2019; 366:l4898. doi: 10.1136/bmj.l4898 [Crossref] [ Google Scholar]
- Fallahzadeh MK, Dormanesh B, Sagheb MM, Roozbeh J, Vessal G, Pakfetrat M. Effect of addition of silymarin to renin-angiotensin system inhibitors on proteinuria in type 2 diabetic patients with overt nephropathy: a randomized, double-blind, placebo-controlled trial. Am J Kidney Dis 2012; 60:896-903. doi: 10.1053/j.ajkd.2012.06.005 [Crossref] [ Google Scholar]
- Jiménez-Osorio AS, García-Niño WR, González-Reyes S, Álvarez-Mejía AE, Guerra-León S, Salazar-Segovia J. The effect of dietary supplementation with curcumin on redox status and Nrf2 activation in patients with nondiabetic or diabetic proteinuric chronic kidney disease: a pilot study. J Ren Nutr 2016; 26:237-44. doi: 10.1053/j.jrn.2016.01.013 [Crossref] [ Google Scholar]
- Golriz Khatami P, Soleimani A, Sharifi N, Aghadavod E, Asemi Z. The effects of high-dose vitamin E supplementation on biomarkers of kidney injury, inflammation, and oxidative stress in patients with diabetic nephropathy: a randomized, double-blind, placebo-controlled trial. J Clin Lipidol 2016; 10:922-9. doi: 10.1016/j.jacl.2016.02.021 [Crossref] [ Google Scholar]
- Tan GCJ, Tan SMQ, Phang SCW, Ng YT, Ng EY, Ahmad B. Tocotrienol-rich vitamin E improves diabetic nephropathy and persists 6-9 months after washout: a phase IIa randomized controlled trial. Ther Adv Endocrinol Metab 2019; 10:2042018819895462. doi: 10.1177/2042018819895462 [Crossref] [ Google Scholar]
- Bahmani F, Kia M, Soleimani A, Asemi Z, Esmaillzadeh A. Effect of selenium supplementation on glycemic control and lipid profiles in patients with diabetic nephropathy. Biol Trace Elem Res 2016; 172:282-9. doi: 10.1007/s12011-015-0600-4 [Crossref] [ Google Scholar]
- Bahmani F, Kia M, Soleimani A, Mohammadi AA, Asemi Z. The effects of selenium supplementation on biomarkers of inflammation and oxidative stress in patients with diabetic nephropathy: a randomised, double-blind, placebo-controlled trial - expression of concern. Br J Nutr 2022; 127:155. doi: 10.1017/s000711452100204x [Crossref] [ Google Scholar]
- Noori N, Tabibi H, Hosseinpanah F, Hedayati M, Nafar M. Effects of combined lipoic acid and pyridoxine on albuminuria, advanced glycation end-products, and blood pressure in diabetic nephropathy. Int J Vitam Nutr Res 2013; 83:77-85. doi: 10.1024/0300-9831/a000147 [Crossref] [ Google Scholar]
- Aghadavod E, Soleimani A, Hamidi G, Keneshlou F, Heidari A, Asemi Z. Effects of high-dose vitamin E supplementation on markers of cardiometabolic risk and oxidative stress in patients with diabetic nephropathy: a randomized double-blinded controlled trial. Iran J Kidney Dis 2018; 12:156-62. [ Google Scholar]
- Tan SM, Chiew Y, Ahmad B, Kadir KA. Tocotrienol-rich vitamin E from palm oil (Tocovid) and its effects in diabetes and diabetic nephropathy: a pilot phase II clinical trial. Nutrients 2018; 10:1315. doi: 10.3390/nu10091315 [Crossref] [ Google Scholar]
- Yang M, Kan L, Wu L, Zhu Y, Wang Q. Effect of baicalin on renal function in patients with diabetic nephropathy and its therapeutic mechanism. Exp Ther Med 2019; 17:2071-6. doi: 10.3892/etm.2019.7181 [Crossref] [ Google Scholar]
- Koay YY, Tan GC, Phang SC, Ho JI, Chuar PF, Ho LS. A phase IIb randomized controlled trial investigating the effects of tocotrienol-rich vitamin E on diabetic kidney disease. Nutrients 2021; 13:258. doi: 10.3390/nu13010258 [Crossref] [ Google Scholar]
- Borges CM, Papadimitriou A, Duarte DA, Lopes de Faria JM, Lopes de Faria JB. The use of green tea polyphenols for treating residual albuminuria in diabetic nephropathy: a double-blind randomised clinical trial. Sci Rep 2016; 6:28282. doi: 10.1038/srep28282 [Crossref] [ Google Scholar]
- Voroneanu L, Siriopol D, Dumea R, Badarau S, Kanbay M, Afsar B. Addition of silymarin to renin-angiotensin system blockers in normotensive patients with type 2 diabetes mellitus and proteinuria: a prospective randomized trial. Int Urol Nephrol 2017; 49:2195-204. doi: 10.1007/s11255-017-1697-5 [Crossref] [ Google Scholar]
- Sattarinezhad A, Roozbeh J, Shirazi Yeganeh B, Omrani GR, Shams M. Resveratrol reduces albuminuria in diabetic nephropathy: a randomized double-blind placebo-controlled clinical trial. Diabetes Metab 2019; 45:53-9. doi: 10.1016/j.diabet.2018.05.010 [Crossref] [ Google Scholar]
- Vasavada N, Agarwal R. Role of oxidative stress in diabetic nephropathy. Adv Chronic Kidney Dis 2005; 12:146-54. doi: 10.1053/j.ackd.2005.01.001 [Crossref] [ Google Scholar]
- Slater TF. Free-radical mechanisms in tissue injury. Biochem J 1984; 222:1-15. doi: 10.1042/bj2220001 [Crossref] [ Google Scholar]
- Chang JM, Kuo MC, Kuo HT, Chiu YW, Chen HC. Increased glomerular and extracellular malondialdehyde levels in patients and rats with diabetic nephropathy. J Lab Clin Med 2005; 146:210-5. doi: 10.1016/j.lab.2005.05.007 [Crossref] [ Google Scholar]
- Sauriasari R, Andrajati R, Azizahwati Azizahwati, Dharmeizar Dharmeizar, Saputri DA, Muris RU. Marker of lipid peroxidation related to diabetic nephropathy in Indonesian type 2 diabetes mellitus patients. Diabetes Res Clin Pract 2015; 108:193-200. doi: 10.1016/j.diabres.2014.12.016 [Crossref] [ Google Scholar]
- Alizadeh M, Kheirouri S. Curcumin reduces malondialdehyde and improves antioxidants in humans with diseased conditions: a comprehensive meta-analysis of randomized controlled trials. Biomedicine (Taipei) 2019; 9:23. doi: 10.1051/bmdcn/2019090423 [Crossref] [ Google Scholar]
- Tang Y, Chen A. Curcumin eliminates the effect of advanced glycation end-products (AGEs) on the divergent regulation of gene expression of receptors of AGEs by interrupting leptin signaling. Lab Invest 2014; 94:503-16. doi: 10.1038/labinvest.2014.42 [Crossref] [ Google Scholar]
- Bergin P, Leggett A, Cardwell CR, Woodside JV, Thakkinstian A, Maxwell AP. The effects of vitamin E supplementation on malondialdehyde as a biomarker of oxidative stress in haemodialysis patients: a systematic review and meta-analysis. BMC Nephrol 2021; 22:126. doi: 10.1186/s12882-021-02328-8 [Crossref] [ Google Scholar]
- Galli F, Azzi A, Birringer M, Cook-Mills JM, Eggersdorfer M, Frank J. Vitamin E: emerging aspects and new directions. Free Radic Biol Med 2017; 102:16-36. doi: 10.1016/j.freeradbiomed.2016.09.017 [Crossref] [ Google Scholar]
- Galli F, Bonomini M, Bartolini D, Zatini L, Reboldi G, Marcantonini G. Vitamin E (alpha-tocopherol) metabolism and nutrition in chronic kidney disease. Antioxidants (Basel) 2022; 11:989. doi: 10.3390/antiox11050989 [Crossref] [ Google Scholar]
- Niki E. Lipid oxidation that is, and is not, inhibited by vitamin E: consideration about physiological functions of vitamin E. Free Radic Biol Med 2021; 176:1-15. doi: 10.1016/j.freeradbiomed.2021.09.001 [Crossref] [ Google Scholar]
- Song Q, Liu J, Dong L, Wang X, Zhang X. Novel advances in inhibiting advanced glycation end product formation using natural compounds. Biomed Pharmacother 2021; 140:111750. doi: 10.1016/j.biopha.2021.111750 [Crossref] [ Google Scholar]
- Zappe K, Pointner A, Switzeny OJ, Magnet U, Tomeva E, Heller J. Counteraction of oxidative stress by vitamin E affects epigenetic regulation by increasing global methylation and gene expression of MLH1 and DNMT1 dose dependently in Caco-2 cells. Oxid Med Cell Longev 2018; 2018:3734250. doi: 10.1155/2018/3734250 [Crossref] [ Google Scholar]
- Gorąca A, Huk-Kolega H, Piechota A, Kleniewska P, Ciejka E, Skibska B. Lipoic acid - biological activity and therapeutic potential. Pharmacol Rep 2011; 63:849-58. doi: 10.1016/s1734-1140(11)70600-4 [Crossref] [ Google Scholar]
- Sena CM, Nunes E, Louro T, Proença T, Fernandes R, Boarder MR. Effects of alpha-lipoic acid on endothelial function in aged diabetic and high-fat fed rats. Br J Pharmacol 2008; 153:894-906. doi: 10.1038/sj.bjp.0707474 [Crossref] [ Google Scholar]
- Sari DP, Susilo I, Khotib J. The mechanism of alpha lipoic acid on reducing the MDA level and MCP-1 expression in endothelial dysfunction of hypercholesterolemia rat (Rattus norvegicus) model. Folia Medica Indonesiana 2016; 52:154-9. doi: 10.20473/fmi.v52i3.5444 [Crossref] [ Google Scholar]
- Muellenbach EA, Diehl CJ, Teachey MK, Lindborg KA, Archuleta TL, Harrell NB. Interactions of the advanced glycation end product inhibitor pyridoxamine and the antioxidant alpha-lipoic acid on insulin resistance in the obese Zucker rat. Metabolism 2008; 57:1465-72. doi: 10.1016/j.metabol.2008.05.018 [Crossref] [ Google Scholar]
- Maritim AC, Sanders RA, Watkins JB 3rd. Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol 2003; 17:24-38. doi: 10.1002/jbt.10058 [Crossref] [ Google Scholar]
- Hong YA, Lim JH, Kim MY, Kim Y, Park HS, Kim HW. Extracellular superoxide dismutase attenuates renal oxidative stress through the activation of adenosine monophosphate-activated protein kinase in diabetic nephropathy. Antioxid Redox Signal 2018; 28:1543-61. doi: 10.1089/ars.2017.7207 [Crossref] [ Google Scholar]
- Yin M, Wheeler MD, Connor HD, Zhong Z, Bunzendahl H, Dikalova A. Cu/Zn-superoxide dismutase gene attenuates ischemia-reperfusion injury in the rat kidney. J Am Soc Nephrol 2001; 12:2691-700. doi: 10.1681/asn.v12122691 [Crossref] [ Google Scholar]
- Pisani A, Sabbatini M, Riccio E, Rossano R, Andreucci M, Capasso C. Effect of a recombinant manganese superoxide dismutase on prevention of contrast-induced acute kidney injury. Clin Exp Nephrol 2014; 18:424-31. doi: 10.1007/s10157-013-0828-2 [Crossref] [ Google Scholar]
- Ma L, Wu F, Shao Q, Chen G, Xu L, Lu F. Baicalin alleviates oxidative stress and inflammation in diabetic nephropathy via Nrf2 and MAPK signaling pathway. Drug Des Devel Ther 2021; 15:3207-21. doi: 10.2147/dddt.s319260 [Crossref] [ Google Scholar]
- Dai J, Liang K, Zhao S, Jia W, Liu Y, Wu H. Chemoproteomics reveals baicalin activates hepatic CPT1 to ameliorate diet-induced obesity and hepatic steatosis. Proc Natl Acad Sci U S A 2018; 115:E5896-905. doi: 10.1073/pnas.1801745115 [Crossref] [ Google Scholar]
- Chu Y, Lan RS, Huang R, Feng H, Kumar R, Dayal S. Glutathione peroxidase-1 overexpression reduces oxidative stress, and improves pathology and proteome remodeling in the kidneys of old mice. Aging Cell 2020; 19:e13154. doi: 10.1111/acel.13154 [Crossref] [ Google Scholar]
- Wiedenmann T, Dietrich N, Fleming T, Altamura S, Deelman LE, Henning RH. Modulation of glutathione peroxidase activity by age-dependent carbonylation in glomeruli of diabetic mice. J Diabetes Complications 2018; 32:130-8. doi: 10.1016/j.jdiacomp.2017.11.007 [Crossref] [ Google Scholar]
- Menzel S, Moeller MJ. Role of the podocyte in proteinuria. Pediatr Nephrol 2011; 26:1775-80. doi: 10.1007/s00467-010-1725-5 [Crossref] [ Google Scholar]
- Gaarkeuken H, Siezenga MA, Zuidwijk K, van Kooten C, Rabelink TJ, Daha MR. Complement activation by tubular cells is mediated by properdin binding. Am J Physiol Renal Physiol 2008; 295:F1397-403. doi: 10.1152/ajprenal.90313.2008 [Crossref] [ Google Scholar]
- Rangan GK, Wang Y, Tay YC, Harris DC. Differential effects of albumin on cytokine gene expression in proximal tubular epithelial cells. Nephrol Dial Transplant 2005; 20:1013-4. doi: 10.1093/ndt/gfh733 [Crossref] [ Google Scholar]
- Tang S, Leung JC, Abe K, Chan KW, Chan LY, Chan TM. Albumin stimulates interleukin-8 expression in proximal tubular epithelial cells in vitro and in vivo. J Clin Invest 2003; 111:515-27. doi: 10.1172/jci16079 [Crossref] [ Google Scholar]
- Rehman MU, Tahir M, Khan AQ, Khan R, Lateef A, Oday OH. Chrysin suppresses renal carcinogenesis via amelioration of hyperproliferation, oxidative stress and inflammation: plausible role of NF-κB. Toxicol Lett 2013; 216:146-58. doi: 10.1016/j.toxlet.2012.11.013 [Crossref] [ Google Scholar]
- Cheng H, Fan X, Moeckel GW, Harris RC. Podocyte COX-2 exacerbates diabetic nephropathy by increasing podocyte (pro)renin receptor expression. J Am Soc Nephrol 2011; 22:1240-51. doi: 10.1681/asn.2010111149 [Crossref] [ Google Scholar]
- Fernández-Real JM, Vendrell J, García I, Ricart W, Vallès M. Structural damage in diabetic nephropathy is associated with TNF-α system activity. Acta Diabetol 2012; 49:301-5. doi: 10.1007/s00592-011-0349-y [Crossref] [ Google Scholar]
- Navarro JF, Mora-Fernández C. The role of TNF-alpha in diabetic nephropathy: pathogenic and therapeutic implications. Cytokine Growth Factor Rev 2006; 17:441-50. doi: 10.1016/j.cytogfr.2006.09.011 [Crossref] [ Google Scholar]
- Bertani T, Abbate M, Zoja C, Corna D, Perico N, Ghezzi P. Tumor necrosis factor induces glomerular damage in the rabbit. Am J Pathol 1989; 134:419-30. [ Google Scholar]
- Suryavanshi SV, Kulkarni YA. NF-κβ: a potential target in the management of vascular complications of diabetes. Front Pharmacol 2017; 8:798. doi: 10.3389/fphar.2017.00798 [Crossref] [ Google Scholar]
- Kaur G, Athar M, Alam MS. Dietary supplementation of silymarin protects against chemically induced nephrotoxicity, inflammation and renal tumor promotion response. Invest New Drugs 2010; 28:703-13. doi: 10.1007/s10637-009-9289-6 [Crossref] [ Google Scholar]
- Zheng XP, Nie Q, Feng J, Fan XY, Jin YL, Chen G. Kidney-targeted baicalin-lysozyme conjugate ameliorates renal fibrosis in rats with diabetic nephropathy induced by streptozotocin. BMC Nephrol 2020; 21:174. doi: 10.1186/s12882-020-01833-6 [Crossref] [ Google Scholar]
- Morishima C, Shuhart MC, Wang CC, Paschal DM, Apodaca MC, Liu Y, et al. Silymarin inhibits in vitro T-cell proliferation and cytokine production in hepatitis C virus infection. Gastroenterology 2010; 138: 671-81.e2. 10.1053/j.gastro.2009.09.021.
- Manna SK, Mukhopadhyay A, Van NT, Aggarwal BB. Silymarin suppresses TNF-induced activation of NF-kappa B, c-Jun N-terminal kinase, and apoptosis. J Immunol 1999; 163:6800-9. doi: 10.4049/jimmunol.163.12.6800 [Crossref] [ Google Scholar]
- Liu Y. Cellular and molecular mechanisms of renal fibrosis. Nat Rev Nephrol 2011; 7:684-96. doi: 10.1038/nrneph.2011.149 [Crossref] [ Google Scholar]
- Braga Gomes K, Fontana Rodrigues K, Fernandes AP. The role of transforming growth factor‐beta in diabetic nephropathy. Int J Med Genet 2014; 2014:180270. doi: 10.1155/2014/180270 [Crossref] [ Google Scholar]
- Yao Y, Yuan Y, Lu Z, Ma Y, Xie Y, Wang M. Effects of Nervilia fordii Extract on pulmonary fibrosis through TGF-β/Smad signaling pathway. Front Pharmacol 2021; 12:659627. doi: 10.3389/fphar.2021.659627 [Crossref] [ Google Scholar]
- Gentle ME, Shi S, Daehn I, Zhang T, Qi H, Yu L. Epithelial cell TGFβ signaling induces acute tubular injury and interstitial inflammation. J Am Soc Nephrol 2013; 24:787-99. doi: 10.1681/asn.2012101024 [Crossref] [ Google Scholar]
- Gomes LR, Terra LF, Wailemann RA, Labriola L, Sogayar MC. TGF-β1 modulates the homeostasis between MMPs and MMP inhibitors through p38 MAPK and ERK1/2 in highly invasive breast cancer cells. BMC Cancer 2012; 12:26. doi: 10.1186/1471-2407-12-26 [Crossref] [ Google Scholar]
- Chen Y, Chen L, Yang T. Silymarin nanoliposomes attenuate renal injury on diabetic nephropathy rats via co-suppressing TGF-β/Smad and JAK2/STAT3/SOCS1 pathway. Life Sci 2021; 271:119197. doi: 10.1016/j.lfs.2021.119197 [Crossref] [ Google Scholar]
- Thrailkill KM, Clay Bunn R, Fowlkes JL. Matrix metalloproteinases: their potential role in the pathogenesis of diabetic nephropathy. Endocrine 2009; 35:1-10. doi: 10.1007/s12020-008-9114-6 [Crossref] [ Google Scholar]
- Cabral-Pacheco GA, Garza-Veloz I, Castruita-De la Rosa C, Ramirez-Acuña JM, Perez-Romero BA, Guerrero-Rodriguez JF. The roles of matrix metalloproteinases and their inhibitors in human diseases. Int J Mol Sci 2020; 21:9739. doi: 10.3390/ijms21249739 [Crossref] [ Google Scholar]
- Conley SM, Bruhn RL, Morgan PV, Stamer WD. Selenium's effects on MMP-2 and TIMP-1 secretion by human trabecular meshwork cells. Invest Ophthalmol Vis Sci 2004; 45:473-9. doi: 10.1167/iovs.03-0767 [Crossref] [ Google Scholar]
- Mima A. Prediction of decreased estimated glomerular filtration rate using liver fibrosis markers: a renal biopsy-based study. Sci Rep 2022; 12:17630. doi: 10.1038/s41598-022-22636-9 [Crossref] [ Google Scholar]
- Casanova AG, López-Hernández FJ, Vicente-Vicente L, Morales AI. Are antioxidants useful in preventing the progression of chronic kidney disease?. Antioxidants (Basel) 2021; 10:1669. doi: 10.3390/antiox10111669 [Crossref] [ Google Scholar]
- Chawla T, Sharma D, Singh A. Role of the renin angiotensin system in diabetic nephropathy. World J Diabetes 2010; 1:141-5. doi: 10.4239/wjd.v1.i5.141 [Crossref] [ Google Scholar]
- Soleimani V, Delghandi PS, Moallem SA, Karimi G. Safety and toxicity of silymarin, the major constituent of milk thistle extract: an updated review. Phytother Res 2019; 33:1627-38. doi: 10.1002/ptr.6361 [Crossref] [ Google Scholar]
- Rutkowski M, Grzegorczyk K. Adverse effects of antioxidative vitamins. Int J Occup Med Environ Health 2012; 25:105-21. doi: 10.2478/S13382-012-0022-x [Crossref] [ Google Scholar]
- Shaito A, Posadino AM, Younes N, Hasan H, Halabi S, Alhababi D. Potential adverse effects of resveratrol: a literature review. Int J Mol Sci 2020; 21:2084. doi: 10.3390/ijms21062084 [Crossref] [ Google Scholar]