Bioimpacts. 2025;15:30255.
doi: 10.34172/bi.30255
Original Article
Simultaneous effect of miR-21 suppression and miR-143 restoration on inhibition of proliferation and migration in SW-480 colorectal cancer cells
Maryam Tohidast Data curation, Formal analysis, Investigation, Software, Visualization, Writing – original draft, Writing – review & editing, 1, 2 
Mohammad Amini Data curation, Formal analysis, Methodology, Software, Validation, 2
Mohammad Amin Doustvandi Data curation, Formal analysis, 2
Seyed Samad Hosseini Investigation, Writing – original draft, Writing – review & editing, 2 
Farzaneh Bilan Investigation, 2
Nazila Mozammel Investigation, 2
Pouryia Sameti Investigation, 2
Amir Ali Mokhtarzadeh Conceptualization, Formal analysis, Methodology, Project administration, Supervision, Validation, Visualization, Writing – review & editing, 2, * 
Behzad Baradaran Conceptualization, Project administration, Supervision, 2, * 
Author information:
1Department of Biotechnology, Higher Education Institute of Rab-Rashid, Tabriz, Iran
2Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
Abstract
Introduction:
Colorectal cancer (CRC) is regarded as a serious global issue and is presently ranked second in the classification of gastrointestinal (GI) malignancies, with fast incidence and high mortality patterns. As the key "gene expression regulators", miRNAs critically contribute to tumor progression and development. For example, miR-21 (an oncomiR) and miR-143 (a tumor suppressor) are dysregulated through colorectal tumorigenesis. Accordingly, this study assesses the concomitant therapeutic impacts of "miR-21 suppression" (anti-miR-21) and "miR-143 restoration" (miR-143) on CRC cell proliferation and migration.
Methods:
SW-480 cell lines (with overexpressed "miR-21" and downregulated "miR-143") were transfected via "anti-miR-21" and "miR-143" mimics, either independently or in combination. Next, cell viability assessment was performed through MTT assay. Then, apoptosis induction was examined with "Annexin V-FITC Kit", and via Propidium Iodide (PI) assay and DAPI staining. In the next step, "cell cycle condition" and "autophagy induction" were studied through flow cytometry. "Wound-healing assay" and "clonogenic assay" were employed to investigate the migration and proliferation of tumor cells. Ultimately, qRT-PCR was utilized to quantify the intensity of the effects of "anti-miR-21" and "miR-143" on gene expression profiles.
Results:
Downregulation of "miR-21" expression and overexpression of "miR-143" were found to synergistically reduce the viability (while elevating apoptosis) of SW-480 cells by modulating Bcl-2 and Bax expression profiles. Combined therapy increased the number of cells in the sub-G1 phase and reduced cell proliferation by modulating expression levels of PTEN and AKT-1. Additionally, miR-21 suppression and miR-143 restoration concomitantly reduced cell migration by modulating the expression of MMP-9.
Conclusion:
Considering anti-cancer effects on cell growth, survival, and migration, it can be concluded that the concomitant suppression of "anti-miR-21" and "miR-143 restoration" might be introduced as a promising method for the therapy of CRC.
Keywords: Colorectal cancer, miR-21, miR-143, Cell proliferation, Migration
Copyright and License Information
© 2025 The Author(s).
This work is published by BioImpacts as an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (
http://creativecommons.org/licenses/by-nc/4.0/). Non-commercial uses of the work are permitted, provided the original work is properly cited.
Funding Statement
The authors are grateful for the financial supports of Immunology Research Center, Tabriz University of Medical Sciences (Grant no. 64490).
Introduction
Colorectal cancer (CRC) is still parading as a global issue, with a cumulative lifetime risk of 5% in the general population.1 As with other types of digestive tract cancers, CRC is a multi-step process2 and is instigated by gradually accumulated genetic and epigenetic changes that ultimately lead to uncontrolled cell proliferation.3 The poor prognosis of CRC patients is the main reason for their low survival rate.4 Thus, during the first 5 years of incidence, 30% to 50% of CRC patients develop post-operation metastasis to other organs (particularly the liver), culminating in therapy failures.5 Besides the surgery, common therapies for CRC include chemotherapy and radiotherapy. However, despite their relative efficacy, these therapies suffer from some shortcomings such as side effects and development of resistance all preventing achievement of measurable results.6 Thus, identifying molecular mechanisms involved in colorectal tumorigenesis is crucial to develop tailored therapies for getting these barriers addressed and ensuring the successful treatment of CRC.7
miRNAs are small (~19 to 22 nucleotides) and endogenous ncRNAs that, by targeting 3′-UTR of intended mRNAs, function to control gene expression post-transcription.8,9 They serve a unique function in a multitude of biological and pathological events by adjusting cell growth, survival, differentiation, proliferation, and apoptosis.4,10 Further, research advocates that miRNAs are dysregulated during tumorigenesis and (while serving as oncomiRs and/or suppressors of tumors) contribute to the progression and development of human malignancies such as CRC, suggesting their therapeutic and diagnostic importance.7,11
“miR-21” and “miR-143” are supposed to be dysregulated through colorectal tumorigenesis and are thus involved in its pathogenesis.12,13 miR-21 is a key miRNA and is viewed as a potent target for CRC prognosis, diagnosis, and treatment.8 Mediating the progression of tumor and cell growth, miR-21 expression is supposed to be upregulated in a sort of human malignancies (e.g., pancreatic cancers, lung cancers, leukemia and osteosarcoma, gastric cancers, and brain tumors).8,14,15 miR-21 is further overexpressed in CRC and serves as an oncomiR by modulating multiple pathways, culminating in non-regulated cell proliferation, apoptosis blockade, as well as stimulation of tumor growth and metastasis.4,16 For example, miR-21 is capable of downregulating “PTEN” expression and elevating the activation of “PI3K/AKT” plus “NF-πB” pathways, thereby promoting cancer cell proliferation.17 Similarly, research advocates that “miR-143” serves a unique function in gene expression regulation through multiple cellular processes.18 This miRNA appears in “miR-143-5p” and “miR-143-3p” isoforms, which are expressed in most human tissues.19 miR-143 is involved in regulating biological events such as cell survival, growth, apoptosis, and differentiation by affecting multiple target genes (e.g., ELK1, Bcl-2, DNMT3A, ERK5, MYO6, and K-Ras).19,20 Furthermore, considering its target mRNAs, miR-143 can modulate multiple signaling pathways through tumorigenesis.19 In CRC cells, “miR-143” is downregulated and functions as a tumor inhibitor, while its overexpression could remarkably prevent CRC cell proliferation and growth, implying its ability as a therapeutic target for CRC.9
RNA oligonucleotides exhibit certain features that cause drug design and efficacy to be adversely affected by shortcomings, including (i) poor penetration in the cell membrane, (ii) degradation by nucleases in biological systems, (iii) entrapment in the endosome, (iv) low binding affinity for complementary sequences (CS), (v) off-target and unwanted toxicities, (vi) inadequate delivery to target tissues, and (vii) activation of innate immune responses. Emerging as a novel therapeutic modality, miRNA delivery has enabled addressing several associated challenges using versatile procedures. For example, the problem of rapid degradation of naked miRNAs by nucleases can be handled by introducing chemical modifications to the oligonucleotides. Yet, a deeper understanding of the functional and biological mechanisms and chemistry of miRNAs and their bioengineering will assuredly promote the versatility of this new therapeutic modality.21
Accordingly, given the importance of miR-21 and miR-143 through colorectal tumorigenesis, this study investigates the concomitant influence of “suppressing miR-21 and restoring miR-143” on CRC cell proliferation, apoptosis, and migration as a new therapeutic modality. The findings obtained imply that anti-miR-21 and miR-143 can supportively hamper tumorigenesis of CRC cells by regulating the expression profiles of various target genes.
Materials and Methods
Cell culture and selection of cell lines
“HCT-116”, “HT-29”, and “SW-480” (i.e., cell lines of CRC) were procured from IPI (Pasteur Institute of Iran-National Cell Bank) and grown in T25 flasks for cell culture with a medium of “RPMI-1640” (USA Gibco,) with 10% GibcoTM FBS and “10 000 U/mL penicillin” and “1000 μg/mL streptomycin” (as antibiotics). The cells were then kept under controlled conditions (i.e., Tm: 37°C; (%) of CO2: 5; (%) of humidity: 95). The appropriate cell line was chosen by evaluating the rates of “miR-21” and “miR-143” expressed in the concerned cell lines using qRT-PCR.22
Cell transfection
Due to its high expression level of miR-21 and low expression level of miR-143, the SW-480 cell line was selected. Both “anti-miR-21” and “miR-143” mimics (at various concentrations of 10, 20, and 40 pmol) along with “FITC conjugate controls” (GenePharma Co, Shanghai) were transferred into “SW-480 cells” at the concentration of 1 × 106 utilizing the Bio-Rad Gene Pulser Xcell System in accordance with the specified protocols (TC = 12.5ms, and Volts = 160v). Totally, 2 × 105 cells were then cultured into six-well plates. The best transfection time (duration) and dosage were determined based on the qRT-PCR-based evaluation of the amounts of miRNA expression in the transfected groups. Transfection efficiency was assessed by employing a MACSQuant® Analyzer10 flow cytometry (Germany).23
RNA extraction and qRT -PCR
The process of total RNA isolation from the concerned cell lines was accomplished by employing the “Trizol RNA Extraction Kit” (GeneAll, S-Korea). RNAs were measured for their purity and concentration by the “Thermo Scientific NanoDrop Spectrophotometer” based on absorbance at “260” and “280” nm. The intended cDNA was then obtained via the reverse-transcription of the extracted mRNAs by the “Universal cDNA Synthesis miRNACURY LNATM kit”. For measuring the expression amounts for the target genes, cDNA was synthesized utilizing the “Takara PrimeScript RT Master Mix”. Next, the expression profiles for “Bcl-2”, “Bax ”, “MMP-9”, “PTEN”, “AKT”, “miR-21”, and “miR-143” were determined with a “BioFACTTM 2X RT-PCR Master Mix” (S-Korea) in a “US Applied Biosystems qRT-PCR instrument”. Ultimately, endogenous housekeeping genes “U6” and “GAPDH” were employed to normalize the expressed rates for concerned genes and miRNAs through the 2-ΔΔCt methodology. The related primers as well as their forward and reverse sequences are reported in Table 1.24
Table 1.
Primer sequences
|
Gene
|
Forward(F)/Reverse(R)
|
Primer sequence
|
|
U6
|
F |
5ˊ-CTTCGGCAGCACATATACTAAAATTGG-3ˊ |
| R |
5ˊ-TCATCCTTGCGCAGGGG-3ˊ |
|
GAPDH
|
F |
5ˊ-CAAGATCATCAGCAATGCCT-3ˊ |
| R |
5ˊ-GCCATCACGCCACAGTTTCC-3ˊ |
|
Bcl-2
|
F |
5ˊ-CTGTGGATGACTGAGTACCTG-3ˊ |
| R |
5ˊ-GAGACAGCCAGGAGAAATCA-3 ˊ |
|
Bax
|
F |
5′-GACTCCCCCCGAGAGGTCTT-3′ |
| R |
5′-ACAGGGCCTTGAGCACCAGTT-3′ |
|
MMP-9
|
F |
5ˊ-TTGACAGCGACAAGAAGTGG-3ˊ |
| R |
5ˊ-GCCATTCACGTCGTCCTTAT-3ˊ |
|
AKT
|
F |
5ˊ-GCTGCACAAACGAGGGGAG-3ˊ |
| R |
5ˊ-CCGCTCCGTCTTCATCAGCT-3ˊ |
|
PTEN
|
F |
5′- TGAGTTCCCTCAGCCGTTACCT-3′ |
| R |
5′- GAGGTTTCCTCTGGTCCTGGTA-3′ |
|
miR-21
|
Target sequence |
5ˊ-CAACACCAGUCGAUGGGCUGU-3′ |
|
miR-143
|
Target sequence |
5′-GGUGCAGUGCUGCAUCUCUGGU-3′ |
MTT assay
MTT assay was implemented for visualizing the cytotoxicity of “miR-143” and “anti-miR-21” against the studied SW-480 cells. Briefly, these cells were first transfected with miRNAs (in combination or alone) and then cultured (i.e., 1 × 104 cells in each single well) in 96-w plates. Two days post-transfection, the wells underwent incubation (Tm: 37°C) with a “50 μL solution of MTT”. Following culture medium removal to dissolve the formazan crystals, each well was treated with “dimethyl sulfoxide” (100 μl). After 30 min of incubation, the optical density (OD) value of each well was read (at 570 nm) using a “Switzerland Tecan Microplate Reader. All steps were replicated three times.25
Wound healing assay
The scratch analysis was conducted for assessing the impacts of combination therapy via miRNA transfection on cell migration ability. For this purpose, SW-480 cells were transfected with anti-miR-21 and miR-143 mimics. Next, each single well was supplied with 3 × 105 transfected cells in 12-well plates (having RPMI-1640 enriched with 10% FBS). When the concerned cells reached a confluency of about 70–80%, cellular monolayers were scratched from the central point via a sterile “yellow pipette tip” for creating a “wound-like gap” through the cells. The wounds were imaged at specific time points (i.e., 0, and 48 hours) via an “OPTIKA Series Inverted Microscope”. The rate of migration was quantified in ImageJ.26
Apoptosis assay
“Annexin V-FITC Kit” and “PI staining”
The rate of apoptosis instigation was evaluated via the “Annexin V-FITC/PI Staining Kit” (Exbio- Czech). Upon transfecting SW-480 cells using “anti-miR-21” and “miR-143”, the cells were transported to six-well plates (i.e., 2 × 105 cells in each single well). Two days post-incubation, the cells were first detached with trypsin/EDTA and subsequently harvested. Next, the flow cytometer instrument (Milteny BiotecTM, Germany) was employed to analyze samples in accordance with the suggested protocols. FlowJo (v10.10, USA) was further utilized to evaluate the rate of apoptosis. All experiments were repeated three times.27
DAPI staining
The DAPI staining method was performed as a qualitative way to further investigate the induction of apoptosis in treatment groups based on chromatin condensation and nuclear fragmentation. After seeding transfected cells into 96-well plates followed by two days of incubation, the cells underwent fixation with 100 μL of 5% paraformaldehyde for 60 min, and subsequently three washes with PBS. Afterward, a 10-minute permeabilization process was achieved using “Triton-X-100” (0.1%), continued with additional washes and staining with a “0.1% DAPI solution” (Sigma Aldrich, USA) for 10 min under darkness. Ultimately, the “Biotek Cytation-5 FI System” was employed to visualize cell morphology.26
Cell cycle assay
Flow cytometry analysis (FCA) evaluated the combined influence of “anti-miR-21” and “miR-143” on the progression of the cell cycle. It was initiated by seeding the transfected cells in six-well plates (i.e., 2 × 105 cells in every single well), followed (after 48 hours) by collecting the cells and fixing them with ethanol (70%) and incubation overnight at Tm: -20°C. After 24 hours, the cells were incubated for 30 min with “1% RNase A” (Bioneer, Daejeon, Korea), underwent permeabilization with “0.1% Triton-X-100”, and stained by “0.1% DAPI solution” for half an hour at 25°C under darkness. Finally, the FCA quantified the amount of cells (by %) arrested in the different phases, with the results analyzed in FlowJo.28
Colony formation assay (CFA)
The clonogenic evaluation was conducted to scrutinize the impact of the “anti-miR-21 and miR-143” combination on the colony-forming ability of SW-480 cells (as a distinctive feature of CSCs – cancer stem cells). To conduct the experiment, SW-480 cells were transfected and seeded into six-well plates at a low (i.e., 1 × 10³) density of cells in each well. The cells were cultured for a weak, followed by PBS-based cell rinsing and then cell fixing with “5% paraformaldehyde”. The cells (post-fixation) were stained for half an hour with “0.5% crystal violet”, followed by washing the plates and counting the colonies after 20 min using an inverted microscope.29
Autophagy assay
The cells were first transfected with miRNAs (combined and alone); subsequently, each well was supplied by 2 × 105 cells in six-well plates. Two days post-transfection, the cells underwent PBS washing and were stained with monodansylcadaverine (MDC, 50 µM). After a 10-minute incubation, PBS was used for rinsing, and trypsin was employed to detach and harvest the cells. The samples then underwent FCA assessment by means of an FCA instrument. The collected data were subsequently analyzed in FlowJo.29
Statistical analysis
All tests were conducted three times. The obtained data for continuous variables were reported as “mean ± SD”. Further, the “T-test” and “one-way ANOVA” were run to identify the significance of differences between study variables. All analyses were done in GraphPad Prism 9, and results with a P < 0.05 were supposed to be statistically significant.30
Results
Levels of “miR-21” and “miR-143” expression in CRC cell lines
The expression rates for both “miR-21” and “miR-143” in three concerned cell lines of CRC (i.e., i) SW-480, ii) HT-29, and iii) HCT-116) were investigated using qRT-PCR. The obtained results (Fig. 1) revealed no statistically significant difference in expressed of “miR-143” between the cell lines of CRC, with the SW-480 cell line exhibiting significantly higher miR-21 expression levels than other cell lines (P < 0.0001). Therefore, according to these results, malignant features of the SW-480 cell line were suggested to be more influenced by miR-21, it was thus preferred (as the optimum option) for additional examinations.
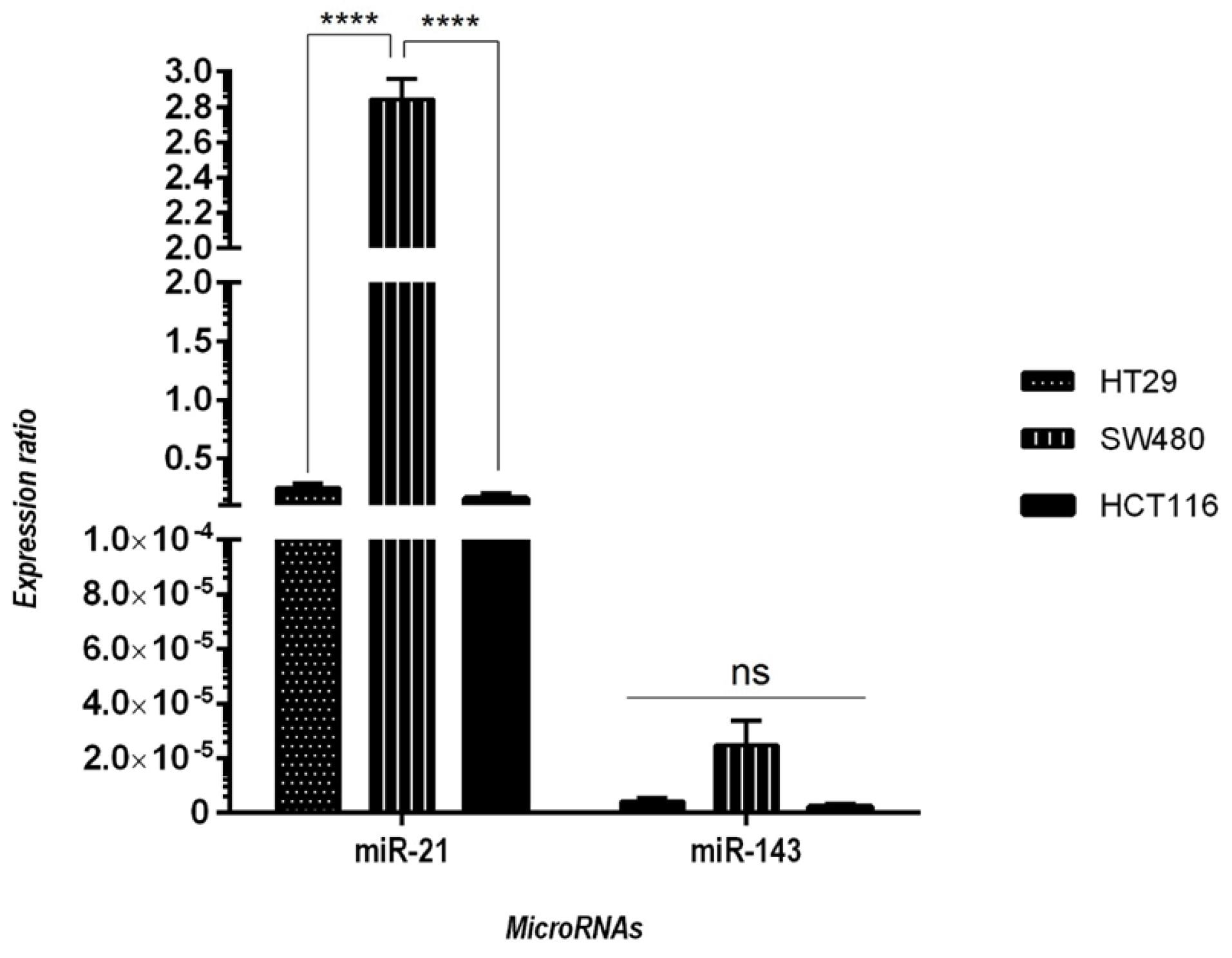
Fig. 1.
The expressed amounts for both “miR-21” and “miR-143” in “SW-480”, “HT-29”, and “HCT-116”, as opposed to each other utilizing qRT-PCR. As demonstrated, the expression rate for “miR-21” in the SW-480 cell line is significantly higher than its expression level in other cell lines. The data are presented as “mean ± SD”; (no. of replications: three); (****P < 0.0001; ns = non-significant).
.
The expressed amounts for both “miR-21” and “miR-143” in “SW-480”, “HT-29”, and “HCT-116”, as opposed to each other utilizing qRT-PCR. As demonstrated, the expression rate for “miR-21” in the SW-480 cell line is significantly higher than its expression level in other cell lines. The data are presented as “mean ± SD”; (no. of replications: three); (****P < 0.0001; ns = non-significant).
Evaluation of transfection efficiency in SW-480 cells
The transfection efficiency of miRNAs was assessed through flow cytometry using a control miR conjugated with FITC. The results (Fig. 2A) indicated that the transfection rate of FITC‐conjugated control miR in SW-480 cells is about 96.6%. qRT-PCR was run to measure the optimal transfection dose and time for anti-miR-21 plus miR-143 in SW-480 cells. As displayed in Fig. 2B, 20 pmol of “anti-miR-21” and “miR-143” could significantly modulate their expression as opposed to other groups (P < 0.0001). In addition, it was found that after 48 hours, the miR-21 expression was significantly downregulated, whilst that of miR-143 was significantly upregulated (P < 0.0001). Thus, based on these results, a dose of “20 pmol” of miRNAs and a duration of “48 hours” were selected as the optimal dose and incubation time, respectively.
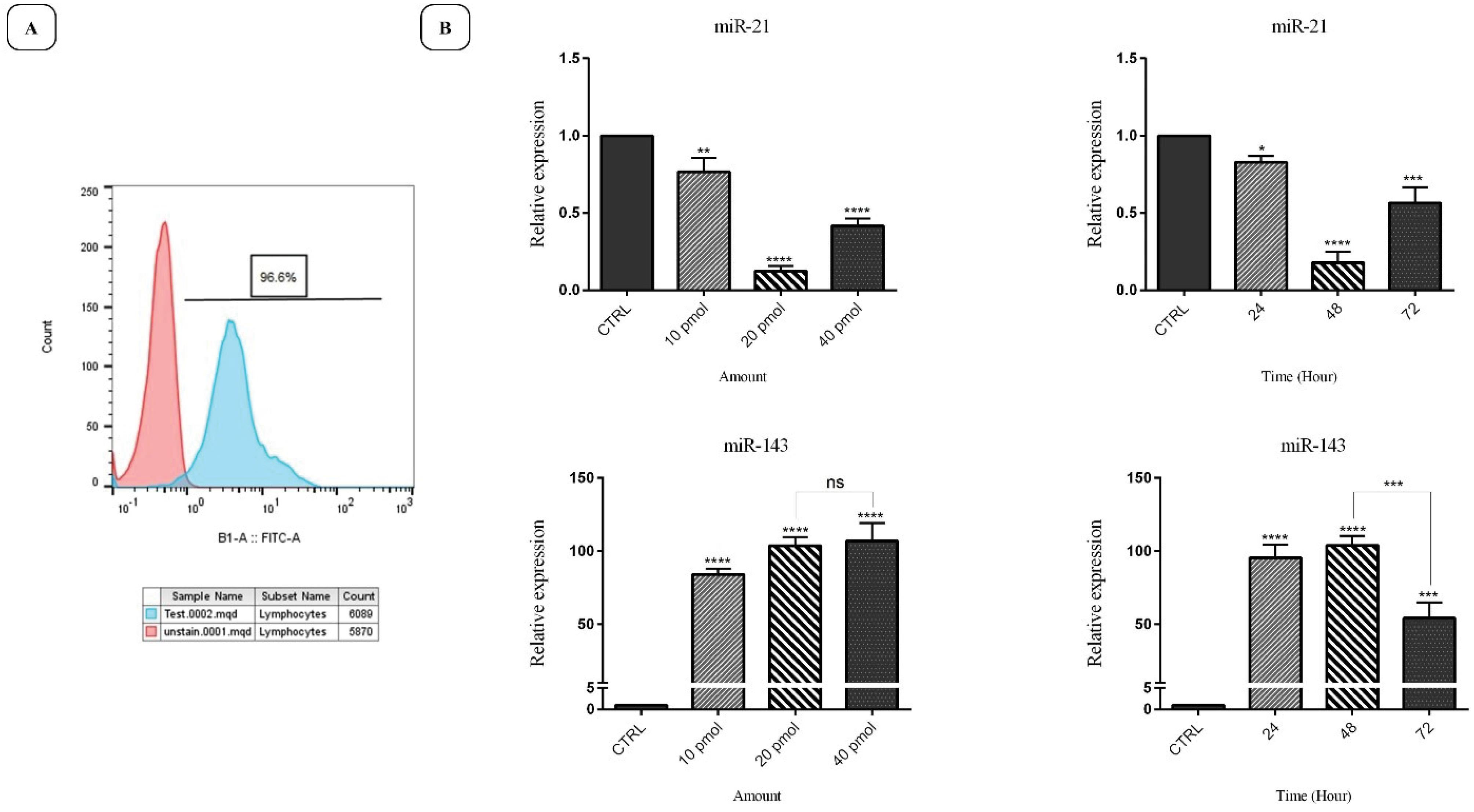
Fig. 2.
A) The efficiency of transfection of miR-21 and miR-143 evaluated using flow cytometry. As shown, the transfection rate is estimated at 96.6%. B) SW-480 cells have been treated with three doses (10, 20, and 40 pmol) of miRNAs and incubated for varying durations (24, 48, and 72 hours). The dose of “20 pmol” and duration of “48 hours” for “anti-miR-21” and “miR-143” were evaluated by qRT-PCR; (****P < 0.0001; *** P < 0.001; ** P < 0.01; * P < 0.05; “ns = non-significant”).
.
A) The efficiency of transfection of miR-21 and miR-143 evaluated using flow cytometry. As shown, the transfection rate is estimated at 96.6%. B) SW-480 cells have been treated with three doses (10, 20, and 40 pmol) of miRNAs and incubated for varying durations (24, 48, and 72 hours). The dose of “20 pmol” and duration of “48 hours” for “anti-miR-21” and “miR-143” were evaluated by qRT-PCR; (****P < 0.0001; *** P < 0.001; ** P < 0.01; * P < 0.05; “ns = non-significant”).
“miR-21 suppression + miR-143 restoration” reduces SW-480 cell viability
The influences of “anti-miR-21” and “miR-143” transfection (alone or simultaneously) on SW-480 cell survival were examined through MTT assay. As demonstrated in Fig. 3, miR-21 suppression significantly diminished cell survival in transfected cells than in “control” and “negative control” groups (P < 0.0001). Likewise, exogenous overexpressed rates of “miR-143” led to a noticeable decline in the viability of SW-480 cells (P < 0.0001) than in the control groups. However, in the combination treatments, cell survival dropped noticeably (P < 0.0001) than in alone treatments.
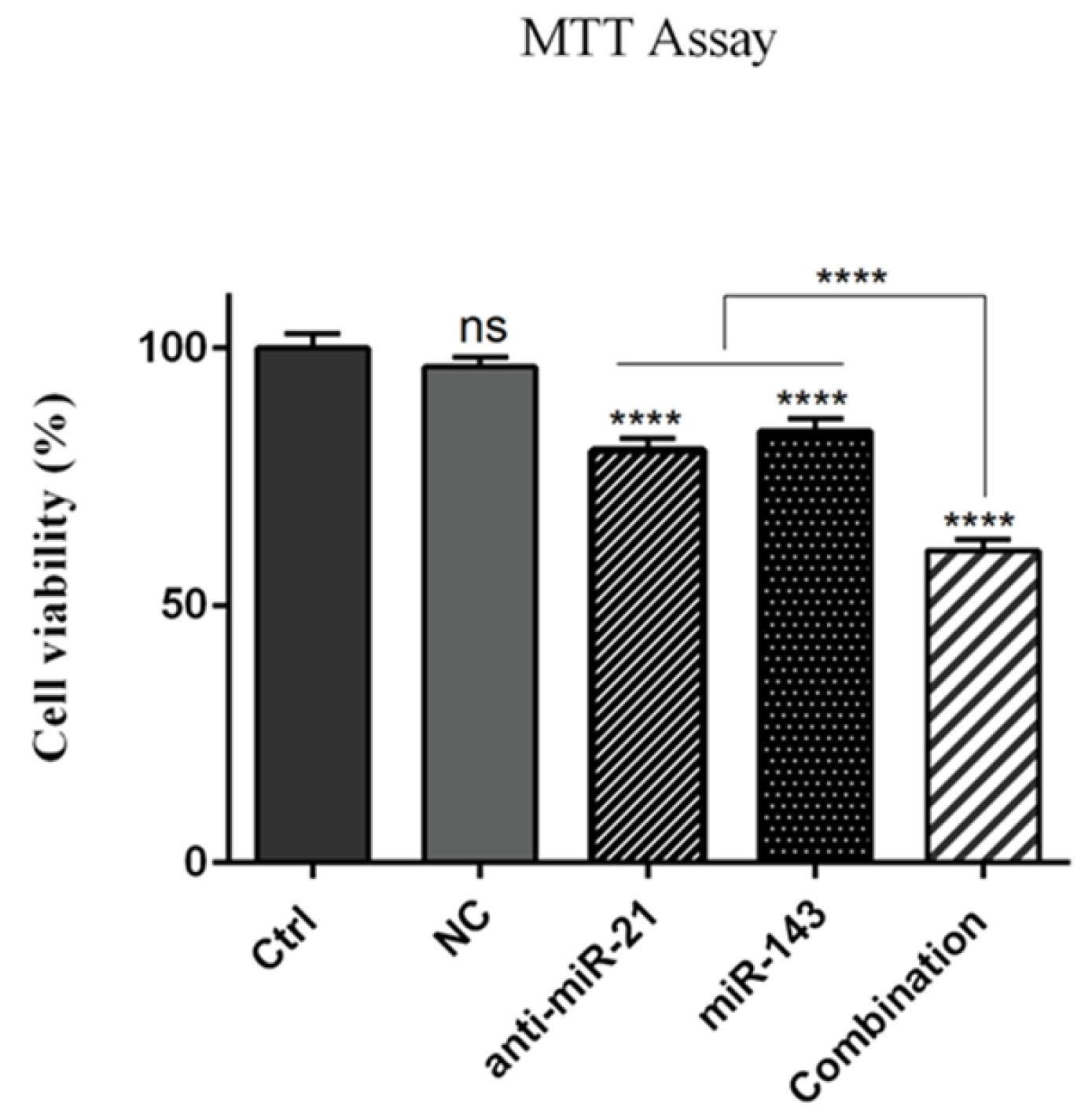
Fig. 3.
Cell survival assessment in various treatment groups using MTT assay. SW-480 cells have been treated with “anti-miR-21” and with “miR-143” (alone or concomitantly). As can be seen, the combined group exhibits a significant reduction in cell survival rate than the individual groups treatments; (****P < 0.0001; “ns = non-significant”).
.
Cell survival assessment in various treatment groups using MTT assay. SW-480 cells have been treated with “anti-miR-21” and with “miR-143” (alone or concomitantly). As can be seen, the combined group exhibits a significant reduction in cell survival rate than the individual groups treatments; (****P < 0.0001; “ns = non-significant”).
“Anti-miR-21 + miR-143” induces apoptosis in SW-480 cells
Annexin V-FITC/PI Staining assay revealed that “miR-143 overexpression” and “miR-21 suppression” remarkably triggered apoptosis induction in SW-480 cells (i.e., up to 12.17% and 15.03%, respectively) compared to the corresponding 3.52% in the control group (P < 0.0001). Contrary to single groups, the apoptosis rate in combination treatments was significantly increased to 32.08% (P < 0.0001) (Fig. 4A). Additionally, DAPI staining revealed that miR-21 suppression and miR-143 overexpression can synergistically enhance the rate of DNA fragmentation more than that in cells treated with single miRNA (Fig. 4B).
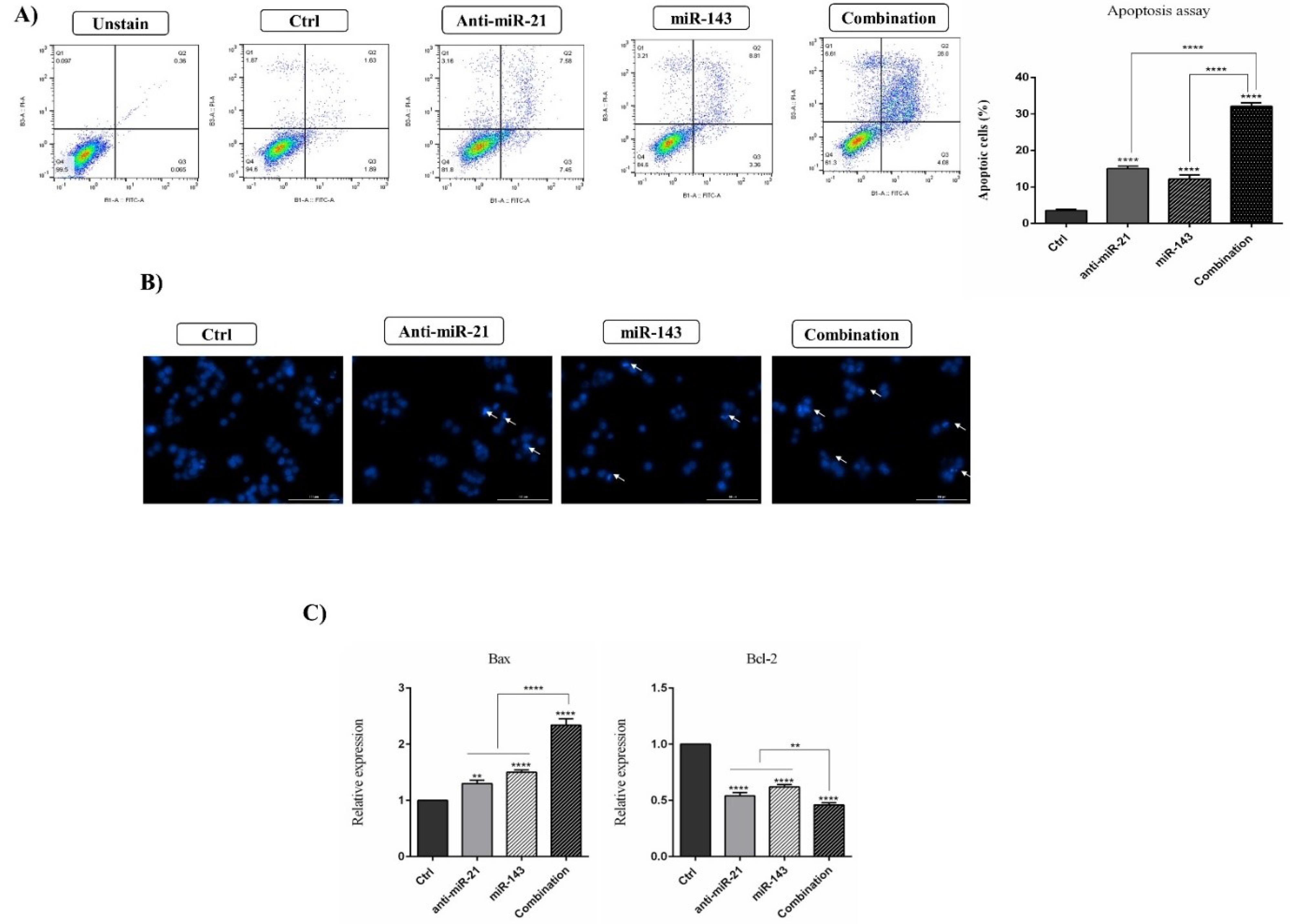
Fig. 4.
“anti-miR-21 + miR-143” have simultaneously induced apoptosis in SW-480 cells. A) Graphically illustrated results for the induction of apoptosis in diverse treatments as evaluated by the “Annexin V-FITC/PI Staining” assay. Transfecting cells with “anti-miR-21” or “miR-143” can remarkably enhance apoptosis rate, but in the combination group, this increase has been significantly higher than in the single groups (****P < 0.0001). B) Fragmentation of chromatin in transfected cells followed by DAPI staining. C) A qRT-PCR assessment of expressed levels for Bcl-2 and Bax genes in the transfected groups than in controls; (****P < 0.0001; *** P < 0.001).
.
“anti-miR-21 + miR-143” have simultaneously induced apoptosis in SW-480 cells. A) Graphically illustrated results for the induction of apoptosis in diverse treatments as evaluated by the “Annexin V-FITC/PI Staining” assay. Transfecting cells with “anti-miR-21” or “miR-143” can remarkably enhance apoptosis rate, but in the combination group, this increase has been significantly higher than in the single groups (****P < 0.0001). B) Fragmentation of chromatin in transfected cells followed by DAPI staining. C) A qRT-PCR assessment of expressed levels for Bcl-2 and Bax genes in the transfected groups than in controls; (****P < 0.0001; *** P < 0.001).
Furthermore, aiming to verify apoptosis induction in the transfected cells, the expressed rates for genes that regulate the apoptosis process were examined by qRT-PCR. As presented in Fig. 4C, the expression rates for the anti-apoptotic gene “Bcl-2” have significantly declined in cells individually transfected with “anti-miR-21” and/or “miR-143” (P < 0.0001) than in the control group. Contrarily, co-transfection of “anti-miR-21” and “miR-143” noticeably lowered the expression levels of Bcl-2 (P < 0.0001), compared to single transfections. It was also found that the levels of Bax mRNA (a proapoptotic gene) increased via cell transfection by “anti-miR-21” (P < 0.001) and “miR-143” mimics (P < 0.0001), contrary to controls. However, in combined treatments, Bax expression levels were higher (P < 0.0001) compared to single treatments.
Inhibition of SW-480 cell migration by merged “anti-miR-21 + miR-143”
In this study, scratch evaluation was performed to assess the influence of “anti-miR-21” and “miR-143” on cell migration ability. It was found that “suppression of miR-21 + overexpression of miR-143” can significantly reduce the migration intensity in SW-480 cells relative to the individually treated groups (P < 0.0001) (Fig. 5A).
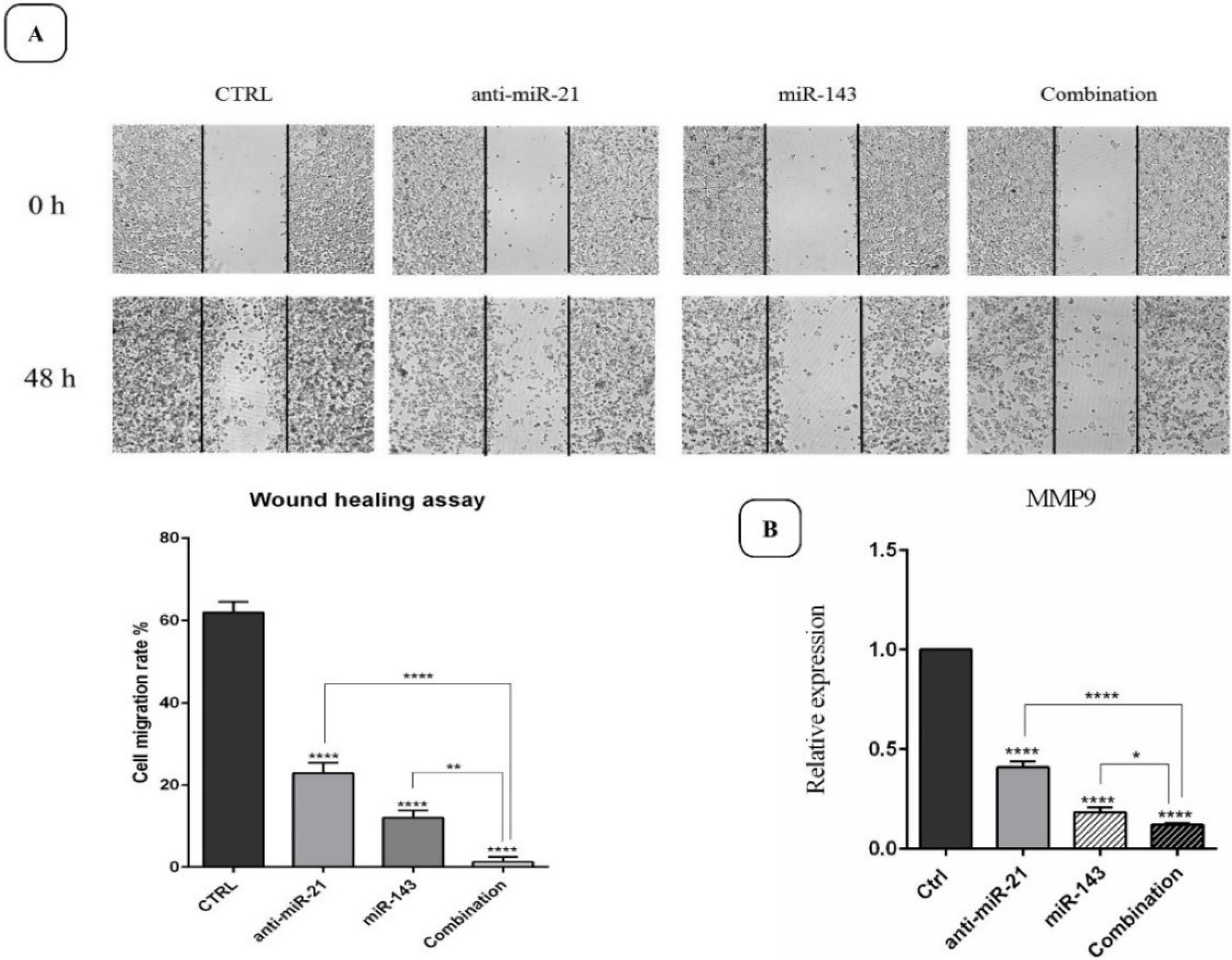
Fig. 5.
The status of “migration inhibition” in different treatment groups evaluated using a wound healing assay, with the results demonstrated graphically. A) 48 h post-incubation, the combination group contributed to a significantly higher migration inhibition than other groups. B) qRT-PCR-based assessment of MMP-9 gene expression levels in the transfected groups in contrast to the controls; (****P < 0.0001; *** P < 0.001; and ** P < 0.01).
.
The status of “migration inhibition” in different treatment groups evaluated using a wound healing assay, with the results demonstrated graphically. A) 48 h post-incubation, the combination group contributed to a significantly higher migration inhibition than other groups. B) qRT-PCR-based assessment of MMP-9 gene expression levels in the transfected groups in contrast to the controls; (****P < 0.0001; *** P < 0.001; and ** P < 0.01).
To further clarify the influences of polytherapy on cell migration, the expression rate for the metastatic gene “MMP-9” was investigated. As displayed in Fig. 5B, the downregulated expression for “MMP-9” is evident in SW-480 cells transfected utilizing “anti-miR-21” and “miR-143” mimics individually, compared to cells in the control group (P < 0.0001). On the other hand, the combination therapy culminated in a significantly lower expression of MMP-9 than cells transfected individually. Collectively, we postulate that “anti-miR-21” and “mimic-miR-143” may prevent CRC migration by regulating metastasis-associated genes such as “MMP-9”.
“Anti-miR-21 + miR-143” effect on cell cycle advancement
FCA was performed to evaluate the influence of “polytherapy” on cell cycle status in cells. As illustrated (Fig. 6), the cell cycle was arrested at the “sub-G1 phase” in transfected cells than in the control group. “Anti-miR-21” and “miR-143” can be expressively increased in the sub-G1 cell population from 0.35% (in the control group) to 6.20% and 5.92%, respectively (P < 0.001). Subsequently, it was found that simultaneous “suppression of miR-21” and “miR-143 overexpression” more effectively triggered the arrest of the cell cycle at the sub-G1 phase (13.4%), compared to the separately transfected cells (P < 0.0001).
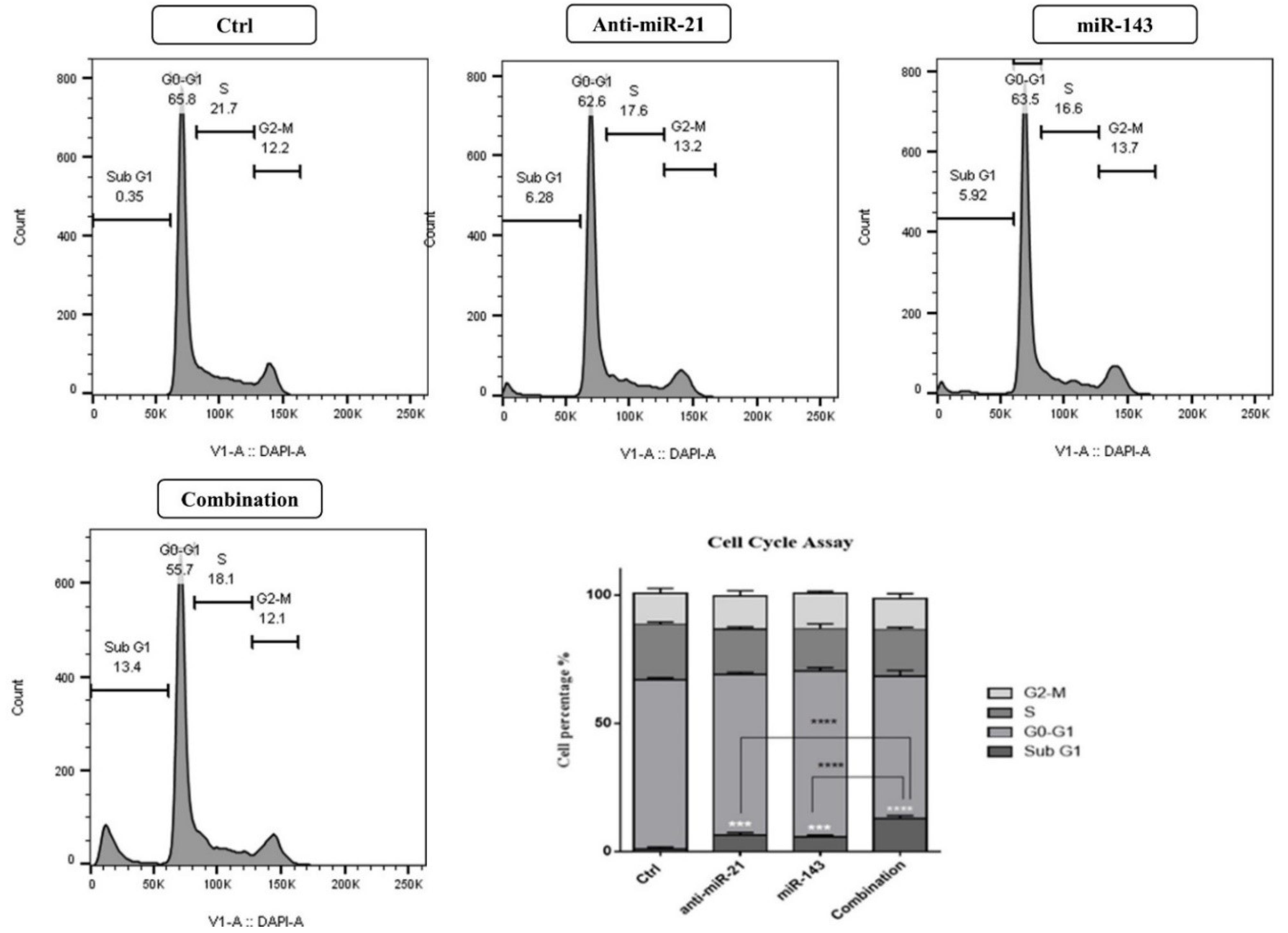
Fig. 6.
The status of the cell cycle in transfected cells assessed utilizing FCA compared with the control group. As shown, combination therapy can provoke the arrest of the cell cycle more effectively at the sub-G1 phase, than separately transfected cells; (****P < 0.0001).
.
The status of the cell cycle in transfected cells assessed utilizing FCA compared with the control group. As shown, combination therapy can provoke the arrest of the cell cycle more effectively at the sub-G1 phase, than separately transfected cells; (****P < 0.0001).
“Suppression of miR-21 + miR-143 overexpression” reduces clonogenic power of SW-480 cells
Considering the antiproliferative effects of “anti-miR-21” and “miR-143”, the clonogenic capacity of SW-480 cells was measured via CFA. Transfection of anti-miR-21 and miR-143 lowered “colony formation capacity” in SW-480 cells than in controls, as demonstrated in Fig. 7A (P < 0.0001). Further, the combined therapy revealed a highly noticeable suppressive impact on the capacity of SW-480 cells to form colonies than anti-miR-21 or miR-143 alone (P < 0.0001). For further evaluation of the impact of polytherapy in CRC cell proliferation, a qRT-PCR analysis quantified the expression amounts of genes that contribute to cell growth. The results in Fig. 7B indicate that transfection with “anti-miR-21” (P < 0.01) and “miR-143” (P < 0.0001) reduces the expression amount of the pro-survival gene “AKT-1” than in controls. In addition, AKT-1 gene expression was more significantly downregulated in cells simultaneously transfected utilizing “anti-miR-21 + miR-143”, in contrast to single treatments (P < 0.0001). Likewise, the expressed amount for the tumor-suppressing gene “PTEN” was enhanced in cells treated with anti-miR-21, in contrast to controls (P < 0.0001). Contrarily, no significant difference was observed in the expression of this gene in cells transfected utilizing miR-143 than in cells in the control group. However, the expression rate for PTEN was uplifted more in the polytherapy group than in the alone treatments (P < 0.0001). Eventually, it was revealed that combination therapy with anti-miR-21 and miR-143 mimics can suppress SW-480 cell “growth” and “proliferation” via modulating PTEN and AKT-1 expression.
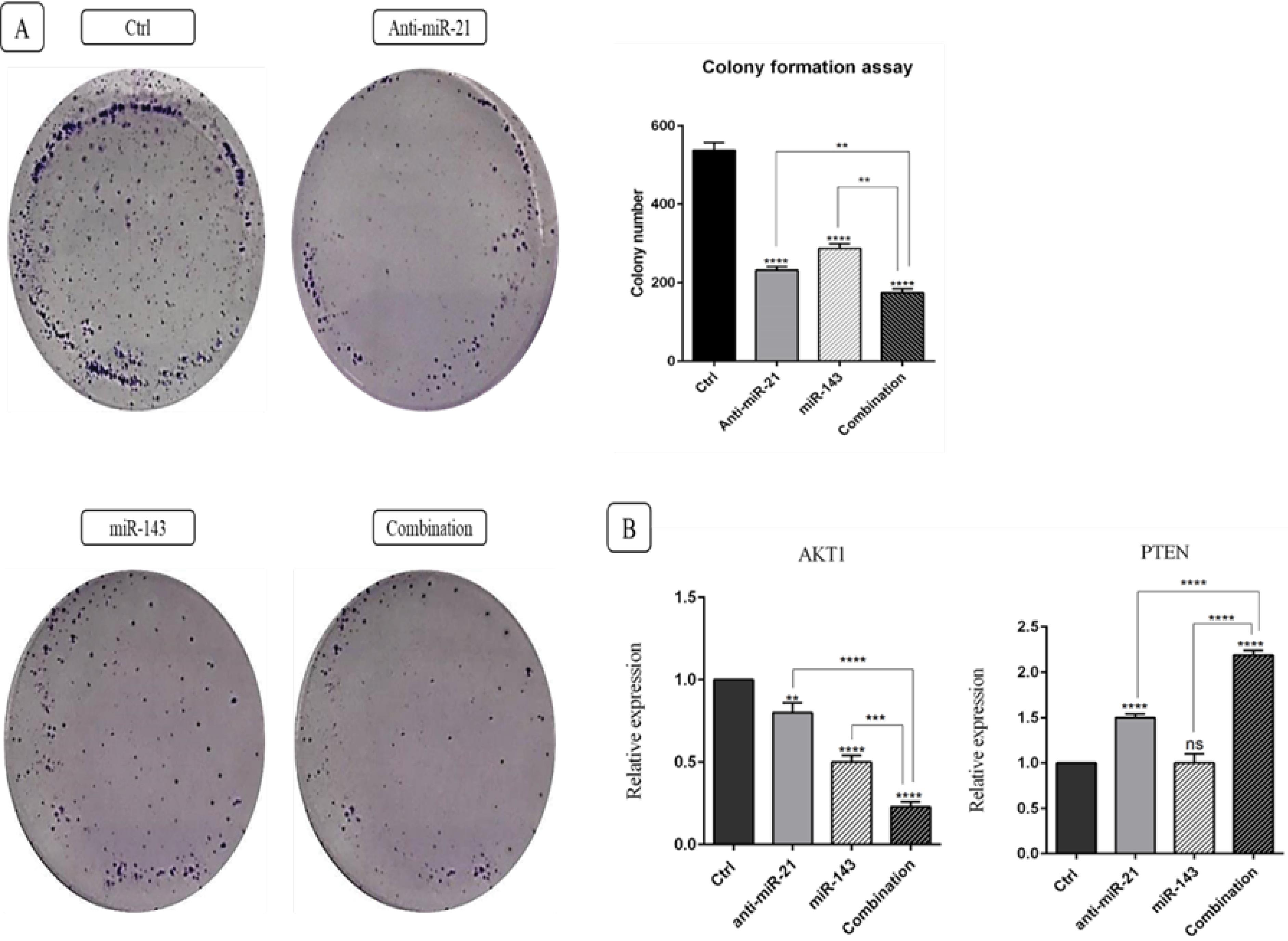
Fig. 7.
A) Transfection by “anti-miR-21” and “miR-143” culminates in a reduction of the colony formation capacity of SW-480 cells, as opposed to controls. However, the combined therapy exhibits a highly noticeable suppressive impact on the colony formation capacity of SW-480 cells when compared with anti-miR-21 or miR-143 alone (****P < 0.0001). B) qRT-PCR assessment of AKT-1 and PTEN gene expression levels in the transfected groups, contrary to controls; (****P < 0.0001; ***P < 0.001; and **P < 0.01; “ns = non-significant”).
.
A) Transfection by “anti-miR-21” and “miR-143” culminates in a reduction of the colony formation capacity of SW-480 cells, as opposed to controls. However, the combined therapy exhibits a highly noticeable suppressive impact on the colony formation capacity of SW-480 cells when compared with anti-miR-21 or miR-143 alone (****P < 0.0001). B) qRT-PCR assessment of AKT-1 and PTEN gene expression levels in the transfected groups, contrary to controls; (****P < 0.0001; ***P < 0.001; and **P < 0.01; “ns = non-significant”).
“anti-miR-21 + miR-143” effect on autophagy
In this study, MDC staining and FCA were used to evaluate autophagy induction in SW-480 cells. The findings revealed that “suppressed miR-21” and “restored miR-143” alone can remarkably induce autophagy in SW-480 cells (P < 0.0001). The autophagy rate was elevated from 0.26% (in the control group) up to 10.7% and 13.5% in anti-miR-21 and mimic miR-143 transfected cells, respectively. In addition, the combination therapy exhibited a higher rate of autophagy induction (30.6%) compared to the “anti-miR-21” and “miR-143” groups (Fig. 8).
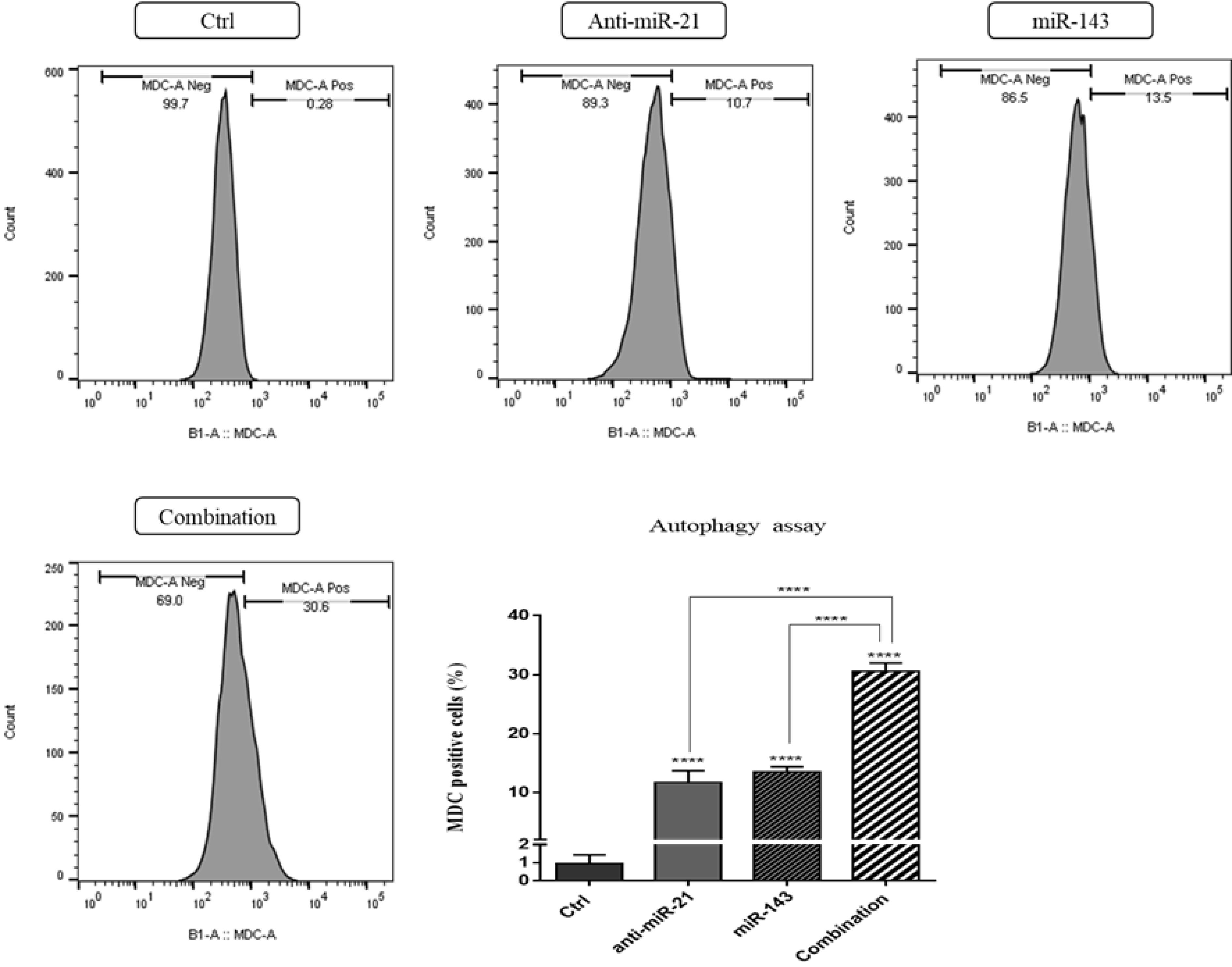
Fig. 8.
Induction of autophagy in different treatment groups based on results from MDC staining. As shown, the combination therapy culminates in a higher autophagy induction rate than the anti-miR-21 and miR-143 groups; (****P < 0.0001).
.
Induction of autophagy in different treatment groups based on results from MDC staining. As shown, the combination therapy culminates in a higher autophagy induction rate than the anti-miR-21 and miR-143 groups; (****P < 0.0001).
Discussion
CRC still remains a serious health threat due to poor prognosis and failure of conventional treatments. Thus, identifying novel therapeutic settings and modalities that can improve the outcomes for CRC patients is desperately required.31,32 Today, miRNA replacement therapy is pondered as a versatile gene therapy methodology to develop new treatment strategies for human cancers such as CRC.33,34 Recent research advocates that miRNAs can directly influence the expression of tumor suppressors/oncogenes and subsequently modulate multiple signaling pathways. In other words, impaired expression of miRNAs can contribute to the progression of an array of human cancers by regulating cell invasion, metastasis, angiogenesis, and resistance to chemotherapy.35 Thus, miRNAs are considered promising therapeutic targets for developing novel treatment strategies for malignancies such as CRC.36
Based on earlier reports, dysregulated “miR-21” and “miR-143” serve a unique function in CRC progression.10,37 Previous studies on miR-21 have shown that this miRNA (serving as an oncomiR, especially in CRC) holds great potential as a diagnostic, prognostic, and therapeutic target. As claimed, upregulated expression of miR-21 may foster the process of metastasis to the liver and lymph nodes in patients suffering from CRC.37 Research further demonstrates that miR-21 upregulation by impairing the expression of several proteins (e.g., Pdcd4 and Sec23A) increases migration and invasion of SW-480 cells.38,39 In addition, miR-21 has been illustrated to be an important predictor for colorectal adenocarcinoma and to contribute to cancer progression by regulating profiles of expression for genes such as ABCB1, HPGD, Bcl-2, TIAM1, TLR3, and PDCD4.40 Likewise, functioning as a tumor suppressor, miR-143 is believed to exhibit meaningful assistance in tumorigenesis of CRC and various cancers. Decreased expression of “miR-143”, which is deemed as a promising diagnostic biomarker for CRC, is shown to be correlated with tumor progression, chemotherapy (CHT) resistance, metastasis, and poor prognosis of patients.2,41 Research shows that miR-143 could block cell proliferation and migration in CRC cells by modulating several genes, including Bcl-2, ERBB3I, K-Ras, MMP-9, c- Myc, and MACC1.42 Therefore, given the importance of both “miR-21” and “miR-143”, this study investigated the impact of suppressing miR-21 using antisense sequence along with exogenous overexpression of miR-143 expression through mimic sequences on CRC cells as a promising therapy.
In this study, MTT assay illustrated that suppression of miR-21 or miR-143 overexpression would lower SW-480 CRC cell viability and that co-transfection of these miRNAs can markedly reduce cell proliferation. In line with these findings, research reports that the suppression of “miR-21” in multiple myeloma cell lines can notably reduce cell survival.43 According to Nabipoorashrafi et al, transfecting miR-143 results in decreased proliferation of WM-115 melanoma cells in vitro.44
Further analysis with Annexin V-FITC/PI and DAPI staining supported that a decline in cell viability in transfection groups can be attributed to apoptosis induction. It was discovered that “anti-miR-21 + miR-143” simultaneous transfection can improve apoptosis induction in SW-480 cells, which further qualifies the combination therapy as a more effective strategy to overcome cancer cell progression. Further, qRT-PCR results indicated upregulation of Bax expression and lowered levels of Bcl-2 in transfection groups, confirming apoptosis induction by the combination therapy. In a similar study, Xiong et al. postulated that the apoptosis rate is enhanced in HCT-116 cells that are transfected with an anti-miR-21 inhibitor.14 Further, miR-143 overexpression was shown to hamper SW-480 CRC cells from proliferation through apoptosis provocation and modulating Bax/Bcl-2 expression ratio.45 Liu et al found that overexpression of miR-143 in HeLa cells reduced Bcl-2 expression, and knocking down miR-143 with anti-miR-143 increased Bcl-2 expression.20
Cell cycle analysis results revealed that simultaneous transfection of SW-480 cells with anti-miR-21 and miR-143 would inhibit cell cycle progression at the sub-G1 phase, more effectively than transfections with single treatments. These findings support those in previous studies that indicate miR-21 overexpression can increase CRC cell growth by mitigating “sub G1 cell cycle arrest” as well as promote resistance to 5-fluorouracil treatment by downregulating the expression of hMSH2.46 Similar results imply “miR-143 overexpression” would contribute to the accumulation of cells in the sub-G1 cell cycle phase in cervical cancer.26
Consistent with previous results, CFA evidenced that miR-21 suppression and miR-143 overexpression can cooperatively diminish the self-renewal and growth of SW-480 cells. Considering the synergistic anti-growth effects of anti-miR-21 and miR-143, we further investigated AKT1 and PTEN expression in treatment groups. Notably, the “PTEN/AKT signaling pathway” plays a decisive role in CRC advancement by influencing multiple cellular events, including cell growth and proliferation. The tumor-suppressing gene PTEN (phosphatase and tensin homolog) can inhibit cell proliferation through dephosphorylation and deactivation of AKT.47 Activated AKT in turn promotes cell proliferation and migration in a sort of human malignancies such as CRC.48 Furthermore, the present study showed that the combination of “suppressed miR-21 + miR-143 overexpression” would lead to PTEN upregulation and AKT-1 downregulation in SW-480 CRC cells. Consistently, PTEN has been documented as a “direct target” for miR-21. Again, miR-21 overexpression can induce the proliferative potential of cancerous cells (e.g., CRC cells) by regulating the PTEN/AKT pathway.11,49 It has also been denoted that the expression of miR-21 is directly linked with the expression of AKT, PTEN, and CD44, which ultimately regulate the growth and migration of keloid keratinocytes.50 In addition, miR-21 suppression through restoring PTEN gene expression leads to reduced proliferation in liver cancer cells.51 As reported by Noguchi et al. overexpressed “miR-143” in human bladder cancer (BC; T24 cells) can significantly reduce AKT expression by modulating PI3K/Akt plus MAPK signaling pathways.52 Thus, it can be assumed that inhibition of miR-21 expression (plus overexpressed miR-143) can hinder the abnormal growth of cancer cells by increasing PTEN tumor suppressor and reducing AKT expression.
Epithelial mesenchymal transition (EMT) is viewed as one of the key stages in the transformation of primary (initial) tumors into metastatic forms of colon cancer.53 In this process, the adhesion properties of epithelial cells are reduced, and, as a result, the process of migration and invasion of tumors to other organs occurs.54 According to the scratch assay results in this study, low levels of miR-21 (due to anti-miR-21) reduce cell migration, as opposed to controls. Further, a similar finding was obtained for increasing the expression of miR-143 using mimic-miR-143. In particular, results demonstrated a substantial decline in the potency of SW-480 cells to migrate and metastasis in the case of simultaneous transfection of “anti-miR-21” and “miR-143” relative to single treatments. Extensive research has explored the function of miR-21 and miR-143 in cell migration in different cancers, whose results concur with the present research findings. In a similar study, it has been reported that up-expressed “miR-21” is associated with the capabilities of CRC cells to migrate and invade. Findings have shown that miR-21 directly hinders tumor-suppressive genes such as TIMP3, PDCD4, PTEN, TPM1, RECK, and Maspin. Such an inhibition triggers several degradation enzymes, thereby increasing cell migration and invasion as well as promoting distant metastases.55 Similarly, miR-143 overexpression hampers migration, cell growth, and invasion in CRC cells by affecting MACC1 (i.e., metastasis-associated in colon cancer-1). Thus, in CRC tissues, an inverse association exists between the level of “miR-143” and the expression of MACC1 mRNA.42 For example, Li et al illustrated that miR-143 overexpression can hamper CRC cell proliferation, invasion, and EMT by downregulating the expression of the HMGA2 gene. This finding suggests that miR-143 reduces the malignant progression of CRC by suppressing HMGA2.56
Research advocates that cancer cell migration inhibition is accomplished by influencing the different genes (e.g., MMP-9) that participate in the metastasis process, which was further analyzed in this study. Matrix metalloproteinase-9 (MMP-9) degrades important extracellular matrix proteins (e.g., collagen proteins) and enhances the migration ability of various cancer cells. Further, MMP-9 activity enables tumor cells to access blood vessels and migrate to other areas of the body, eventually causing metastasis.57 Importantly, MMP-9 upregulation has been recognized to be closely linked with cell metastasis and poor prognosis in CRC patients. We further displayed that suppression of “miR-21” and restoration of “miR-143” cooperatively reduced MMP-9 expression in SW-480 cells. In line with our results, restoring miR-143 expression has been shown to diminish SW-480 cell migration through downregulating MMP-9 expression.43 Wang et al stated that miR-21 leads to cystic fibrosis by downregulating the expression of the MMP-9 gene.57 Again, research has found that increasing the expression of miR-143 would lower the migration of oral squamous cell carcinomas by declining the expression of invasive genes including MMP-9, C- Myc, and K-Ras.58 Further, miR-21 was previously reported to directly target tissue inhibitors of metalloproteinase-3 (TIMP3), which indeed can increase the expression of both “MMP-9” and “MMP-2”, thereby promoting the migration and tube formation of endothelial cells.59 Research shows that overexpression of miR-143 in melanoma cancer cells suppresses metastasis-related genes, including E-cadherin, Vimentin, CXCR4, and MMP-9.44 Thus, it can be stated that the combined use of anti-miR-21 and miR-143 might be a promising technique to significantly reduce metastasis and invasion in CRC cells.
This study evaluated the effect of suppressing miR-21 expression along with increasing miR-143 expression on autophagy induction in SW-480 cells. Under normal cellular conditions, autophagy is a mechanism for maintaining biological activity through the recovery of damaged proteins and organs. However, in cancer cells, it may inhibit tumorigenesis by reducing cell proliferation and inducing cell death.60 Our results also revealed a significantly enhanced autophagy rate in the groups that were transfected utilizing “anti-miR-21” and “miR-143”, as opposed to the control groups. However, suppression of miR-21 expression along with increased miR-143 expression further elevated the induction of autophagy in SW-480 cells. Despite our limited knowledge about the contribution of miRNAs to autophagy induction, it was shown that targeting miR-21 increased autophagy-linked proteins “Vps34”, “Beclin-1”, and “LC3-II” as well as induced autophagy in leukemia cells.61 Wei et al demonstrated that miR-143 serves a decisive function in regulating autophagy by targeting ATG2B and suppressing gene expression in autophagy in NSCLC (i.e., non-small cell lung cancer) cells.62
Conclusion
Collectively, the findings reported here indicated that simultaneous “suppression of miR-21 + restoration of miR-143” can effectively and cooperatively inhibit in vitro tumorigenesis in SW-480 CRC cells. The designed combination therapy was found to hamper cell viability through Bcl-2/ Bax expression and subsequent apoptosis induction. Again, it was found that the “anti-miR-21 and mimic-miR-143” combination would reduce cell migration by downregulating MMP-9 and inhibit cell growth by modulating AKT-1 and PTEN expression. It is suggested that anti-miR-21 and mimic-miR-143 functions are far more pronounced when they are used in combination. Thus, their simultaneous use can be deemed as a novel therapeutic technique to improve the treatment of CRC.
Research Highlights
What is the current knowledge?
What is new here?
Acknowledgments
The authors thank support from the TUMS-IRC (Immunology Research Center of TUMS; Tabriz, Iran).
Competing Interests
The authors assert no conflict of interest.
Ethical Statement
This study was approved by the ethics committee of Tabriz University of Medical Sciences, Tabriz, Iran (Ethic Code No: IR.TBZMED.REC.1398.1178)
References
- Chapelle N, Martel M, Toes-Zoutendijk E, Barkun AN, Bardou M. Recent advances in clinical practice: colorectal cancer chemoprevention in the average-risk population. Gut 2020; 69:2244-55. doi: 10.1136/gutjnl-2020-320990 [Crossref] [ Google Scholar]
- Sahami-Fard MH, Kheirandish S, Sheikhha MH. Expression levels of miR-143-3p and -424-5p in colorectal cancer and their clinical significance. Cancer Biomark 2019; 24:291-7. doi: 10.3233/cbm-182171 [Crossref] [ Google Scholar]
- Porru M, Pompili L, Caruso C, Biroccio A, Leonetti C. Targeting KRAS in metastatic colorectal cancer: current strategies and emerging opportunities. J Exp Clin Cancer Res 2018; 37:57. doi: 10.1186/s13046-018-0719-1 [Crossref] [ Google Scholar]
- Hejazi M, Baghbani E, Amini M, Rezaei T, Aghanejad A, Mosafer J. MicroRNA-193a and taxol combination: a new strategy for treatment of colorectal cancer. J Cell Biochem 2020; 121:1388-99. doi: 10.1002/jcb.29374 [Crossref] [ Google Scholar]
- Scheer A, Auer RA. Surveillance after curative resection of colorectal cancer. Clin Colon Rectal Surg 2009; 22:242-50. doi: 10.1055/s-0029-1242464 [Crossref] [ Google Scholar]
- Wang H, Nie L, Wu L, Liu Q, Guo X. NR2F2 inhibits Smad7 expression and promotes TGF-β-dependent epithelial-mesenchymal transition of CRC via transactivation of miR-21. BiochemBiophys Res Commun 2017; 485:181-8. doi: 10.1016/j.bbrc.2017.02.049 [Crossref] [ Google Scholar]
- Dunbar CE, High KA, Joung JK, Kohn DB, Ozawa K, Sadelain M. Gene therapy comes of age. Science 2018; 359:eaan4672. doi: 10.1126/science.aan4672 [Crossref] [ Google Scholar]
- Li T, Leong MH, Harms B, Kennedy G, Chen L. MicroRNA-21 as a potential colon and rectal cancer biomarker. World J Gastroenterol 2013; 19:5615-21. doi: 10.3748/wjg.v19.i34.5615 [Crossref] [ Google Scholar]
- Liu Q, Yang W, Luo Y, Hu S, Zhu L. Correlation between miR-21 and miR-145 and the incidence and prognosis of colorectal cancer. J BUON 2018; 23:29-35. [ Google Scholar]
- Akao Y, Nakagawa Y, Hirata I, Iio A, Itoh T, Kojima K. Role of anti-oncomirs miR-143 and -145 in human colorectal tumors. Cancer Gene Ther 2010; 17:398-408. doi: 10.1038/cgt.2009.88 [Crossref] [ Google Scholar]
- Wu Y, Song Y, Xiong Y, Wang X, Xu K, Han B. MicroRNA-21 (miR-21) promotes cell growth and invasion by repressing tumor suppressor PTEN in colorectal cancer. Cell PhysiolBiochem 2017; 43:945-58. doi: 10.1159/000481648 [Crossref] [ Google Scholar]
- Chi Y, Zhou D. MicroRNAs in colorectal carcinoma--from pathogenesis to therapy. J Exp Clin Cancer Res 2016; 35:43. doi: 10.1186/s13046-016-0320-4 [Crossref] [ Google Scholar]
- Hosseini SS, Nazifi P, Amini M, Zargari F, Yari AH, Baradaran B. Crocin suppresses colorectal cancer cell proliferation by regulating miR-143/145 and KRAS/RREB1 pathways. Anticancer Agents Med Chem 2023; 23:1916-23. doi: 10.2174/1871520623666230718145100 [Crossref] [ Google Scholar]
- Xiong B, Cheng Y, Ma L, Zhang C. MiR-21 regulates biological behavior through the PTEN/PI-3 K/Akt signaling pathway in human colorectal cancer cells. Int J Oncol 2013; 42:219-28. doi: 10.3892/ijo.2012.1707 [Crossref] [ Google Scholar]
- Bilan F, Amini M, Doustvandi MA, Tohidast M, Baghbanzadeh A, Hosseini SS. Simultaneous suppression of miR-21 and restoration of miR-145 in gastric cancer cells; a promising strategy for inhibition of cell proliferation and migration. Bioimpacts 2024; 14:27764. doi: 10.34172/bi.2023.27764 [Crossref] [ Google Scholar]
- Yu W, Zhu K, Wang Y, Yu H, Guo J. Overexpression of miR-21-5p promotes proliferation and invasion of colon adenocarcinoma cells through targeting CHL1. Mol Med 2018; 24:36. doi: 10.1186/s10020-018-0034-5 [Crossref] [ Google Scholar]
- Liu H, Wang J, Tao Y, Li X, Qin J, Bai Z. Curcumol inhibits colorectal cancer proliferation by targeting miR-21 and modulated PTEN/PI3K/Akt pathways. Life Sci 2019; 221:354-61. doi: 10.1016/j.lfs.2019.02.049 [Crossref] [ Google Scholar]
- Bajhan E, Mansoori B, Mohammadi A, Shanehbandi D, Khaze Shahgoli V, Baghbani E. MicroRNA-143 inhibits proliferation and migration of prostate cancer cells. Arch PhysiolBiochem 2022; 128:1323-9. doi: 10.1080/13813455.2020.1769678 [Crossref] [ Google Scholar]
- Karimi L, Mansoori B, Shanebandi D, Mohammadi A, Aghapour M, Baradaran B. Function of microRNA-143 in different signal pathways in cancer: new insights into cancer therapy. Biomed Pharmacother 2017; 91:121-31. doi: 10.1016/j.biopha.2017.04.060 [Crossref] [ Google Scholar]
- Liu L, Yu X, Guo X, Tian Z, Su M, Long Y. miR-143 is downregulated in cervical cancer and promotes apoptosis and inhibits tumor formation by targeting Bcl-2. Mol Med Rep 2012; 5:753-60. doi: 10.3892/mmr.2011.696 [Crossref] [ Google Scholar]
- Segal M, Slack FJ. Challenges identifying efficacious miRNA therapeutics for cancer. Expert Opin Drug Discov 2020; 15:987-92. doi: 10.1080/17460441.2020.1765770 [Crossref] [ Google Scholar]
- Tohidast M, Memari N, Amini M, Hosseini SS, Jebelli A, Doustvandi MA. MiR-145 inhibits cell migration and increases paclitaxel chemosensitivity in prostate cancer cells. Iran J Basic Med Sci 2023; 26:1350-9. doi: 10.22038/ijbms.2023.70878.15397 [Crossref] [ Google Scholar]
- Mozammel N, Baghbani E, Amini M, Jodeiry Zaer S, Baghay Esfandyari Y, Tohidast M. The simultaneous effects of miR-145-5p and hsa-let-7a-3p on colorectal tumorigenesis: in vitro evidence. Adv Pharm Bull 2024; 14:231-40. doi: 10.34172/apb.2024.004 [Crossref] [ Google Scholar]
- Jodeiry Zaer S, Aghamaali M, Amini M, Doustvandi MA, Hosseini SS, Baradaran B. Cooperatively inhibition effect of miR-143-5p and miR-145-5p in tumorigenesis of glioblastoma cells through modulating AKT signaling pathway. Bioimpacts 2024; 14:29913. doi: 10.34172/bi.2023.29913 [Crossref] [ Google Scholar]
- Hosseini SS, Zare Reihani R, Doustvandi MA, Amini M, Zargari F, Baradaran B. Synergistic anticancer effects of curcumin and crocin on human colorectal cancer cells. Mol Biol Rep 2022; 49:8741-52. doi: 10.1007/s11033-022-07719-0 [Crossref] [ Google Scholar]
- Baghay Esfandyari Y, Doustvandi MA, Amini M, Baradaran B, Jodeiry Zaer S, Mozammel N. MicroRNA-143 sensitizes cervical cancer cells to cisplatin: a promising anticancer combination therapy. Reprod Sci 2021; 28:2036-49. doi: 10.1007/s43032-021-00479-5 [Crossref] [ Google Scholar]
- Ahangar NK, Khalaj-Kondori M, Alizadeh N, Mokhtarzadeh A, Baghbanzadeh A, Abdoli Shadbad M. Silencing tumor-intrinsic HHLA2 potentiates the anti-tumoral effect of paclitaxel on MG63 cells: another side of immune checkpoint. Gene 2023; 855:147086. doi: 10.1016/j.gene.2022.147086 [Crossref] [ Google Scholar]
- Aghajani M, Mokhtarzadeh A, Aghebati-Maleki L, Mansoori B, Mohammadi A, Safaei S. CD133 suppression increases the sensitivity of prostate cancer cells to paclitaxel. Mol Biol Rep 2020; 47:3691-703. doi: 10.1007/s11033-020-05411-9 [Crossref] [ Google Scholar]
- Mansoori B, Silvestris N, Mohammadi A, Khaze V, Baghbani E, Mokhtarzadeh A. miR-34a and miR-200c have an additive tumor-suppressive effect on breast cancer cells and patient prognosis. Genes (Basel) 2021; 12:267. doi: 10.3390/genes12020267 [Crossref] [ Google Scholar]
- Baghbanzadeh A, Baghbani E, Hajiasgharzadeh K, Noorolyai S, Khaze V, Mansoori B. microRNA-193a-5p suppresses the migratory ability of human KATO III gastric cancer cells through inhibition of vimentin and MMP-9. Adv Pharm Bull 2022; 12:169-75. doi: 10.34172/apb.2022.018 [Crossref] [ Google Scholar]
- Yamamoto H, Mori M. MicroRNAs as therapeutic targets and colorectal cancer therapeutics. Adv Exp Med Biol 2016; 937:239-47. doi: 10.1007/978-3-319-42059-2_13 [Crossref] [ Google Scholar]
- Bayat Mokhtari R, Homayouni TS, Baluch N, Morgatskaya E, Kumar S, Das B. Combination therapy in combating cancer. Oncotarget 2017; 8:38022-43. doi: 10.18632/oncotarget.16723 [Crossref] [ Google Scholar]
- Asadzadeh Z, Mansoori B, Mohammadi A, Aghajani M, Haji-Asgarzadeh K, Safarzadeh E. microRNAs in cancer stem cells: biology, pathways, and therapeutic opportunities. J Cell Physiol 2019; 234:10002-17. doi: 10.1002/jcp.27885 [Crossref] [ Google Scholar]
- Aumeeruddy MZ, Mahomoodally MF. Combating breast cancer using combination therapy with 3 phytochemicals: piperine, sulforaphane, and thymoquinone. Cancer 2019; 125:1600-11. doi: 10.1002/cncr.32022 [Crossref] [ Google Scholar]
- Wuchty S, Arjona D, Bozdag S, Bauer PO. Involvement of microRNA families in cancer. Nucleic Acids Res 2012; 40:8219-26. doi: 10.1093/nar/gks627 [Crossref] [ Google Scholar]
- Thomas J, Ohtsuka M, Pichler M, Ling H. MicroRNAs: clinical relevance in colorectal cancer. Int J Mol Sci 2015; 16:28063-76. doi: 10.3390/ijms161226080 [Crossref] [ Google Scholar]
- Basati G, Emami Razavi A, Abdi S, Mirzaei A. Elevated level of microRNA-21 in the serum of patients with colorectal cancer. Med Oncol 2014; 31:205. doi: 10.1007/s12032-014-0205-3 [Crossref] [ Google Scholar]
- Asangani IA, Rasheed SA, Nikolova DA, Leupold JH, Colburn NH, Post S. MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor PDCD4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene 2008; 27:2128-36. doi: 10.1038/sj.onc.1210856 [Crossref] [ Google Scholar]
- Li C, Zhao L, Chen Y, He T, Chen X, Mao J. MicroRNA-21 promotes proliferation, migration, and invasion of colorectal cancer, and tumor growth associated with down-regulation of sec23a expression. BMC Cancer 2016; 16:605. doi: 10.1186/s12885-016-2628-z [Crossref] [ Google Scholar]
- Saheb Sharif-Askari N, Saheb Sharif-Askari F, Guraya SY, Bendardaf R, Hamoudi R. Integrative systematic review meta-analysis and bioinformatics identifies microRNA-21 and its target genes as biomarkers for colorectal adenocarcinoma. Int J Surg 2020; 73:113-22. doi: 10.1016/j.ijsu.2019.11.017 [Crossref] [ Google Scholar]
- Yu B, Liu X, Chang H. MicroRNA-143 inhibits colorectal cancer cell proliferation by targeting MMP7. Minerva Med 2017; 108:13-9. doi: 10.23736/s0026-4806.16.04651-6 [Crossref] [ Google Scholar]
- Zhang Y, Wang Z, Chen M, Peng L, Wang X, Ma Q. MicroRNA-143 targets MACC1 to inhibit cell invasion and migration in colorectal cancer. Mol Cancer 2012; 11:23. doi: 10.1186/1476-4598-11-23 [Crossref] [ Google Scholar]
- Leone E, Morelli E, Di Martino MT, Amodio N, Foresta U, Gullà A. Targeting miR-21 inhibits in vitro and in vivo multiple myeloma cell growth. Clin Cancer Res 2013; 19:2096-106. doi: 10.1158/1078-0432.ccr-12-3325 [Crossref] [ Google Scholar]
- Nabipoorashrafi SA, Shomali N, Sadat-Hatamnezhad L, Mahami-Oskouei M, Mahmoudi J, Sandoghchian Shotorbani B. miR-143 acts as an inhibitor of migration and proliferation as well as an inducer of apoptosis in melanoma cancer cells in vitro. IUBMB Life 2020; 72:2034-44. doi: 10.1002/iub.2345 [Crossref] [ Google Scholar]
- Karimi L, Zeinali T, Hosseinahli N, Mansoori B, Mohammadi A, Yousefi M. miRNA-143 replacement therapy harnesses the proliferation and migration of colorectal cancer cells in vitro. J Cell Physiol 2019; 234:21359-68. doi: 10.1002/jcp.28745 [Crossref] [ Google Scholar]
- Valeri N, Gasparini P, Braconi C, Paone A, Lovat F, Fabbri M. MicroRNA-21 induces resistance to 5-fluorouracil by down-regulating human DNA MutS homolog 2 (hMSH2). Proc Natl Acad Sci U S A 2010; 107:21098-103. doi: 10.1073/pnas.1015541107 [Crossref] [ Google Scholar]
- Sun Y, Tian H, Wang L. Effects of PTEN on the proliferation and apoptosis of colorectal cancer cells via the phosphoinositol-3-kinase/Akt pathway. Oncol Rep 2015; 33:1828-36. doi: 10.3892/or.2015.3804 [Crossref] [ Google Scholar]
- Tokunaga E, Oki E, Egashira A, Sadanaga N, Morita M, Kakeji Y. Deregulation of the Akt pathway in human cancer. Curr Cancer Drug Targets 2008; 8:27-36. doi: 10.2174/156800908783497140 [Crossref] [ Google Scholar]
- Liu H, Huang X, Liu X, Xiao S, Zhang Y, Xiang T. miR-21 promotes human nucleus pulposus cell proliferation through PTEN/AKT signaling. Int J Mol Sci 2014; 15:4007-18. doi: 10.3390/ijms15034007 [Crossref] [ Google Scholar]
- Yan L, Cao R, Liu Y, Wang L, Pan B, Lv X. MiR-21-5p links epithelial-mesenchymal transition phenotype with stem-like cell signatures via AKT signaling in keloid keratinocytes. Sci Rep 2016; 6:28281. doi: 10.1038/srep28281 [Crossref] [ Google Scholar]
- Liu H, Cheng L, Cao D, Zhang H. Suppression of miR-21 expression inhibits cell proliferation and migration of liver cancer cells by targeting phosphatase and tensin homolog (PTEN). Med Sci Monit 2018; 24:3571-7. doi: 10.12659/msm.907038 [Crossref] [ Google Scholar]
- Noguchi S, Yasui Y, Iwasaki J, Kumazaki M, Yamada N, Naito S. Replacement treatment with microRNA-143 and -145 induces synergistic inhibition of the growth of human bladder cancer cells by regulating PI3K/Akt and MAPK signaling pathways. Cancer Lett 2013; 328:353-61. doi: 10.1016/j.canlet.2012.10.017 [Crossref] [ Google Scholar]
- Salehi S, Mansoori B, Mohammadi A, Davoudian S, Musavi Shenas SM, Shajari N. An analysis of suppressing migratory effect on human urinary bladder cancer cell line by silencing of snail-1. Biomed Pharmacother 2017; 96:545-50. doi: 10.1016/j.biopha.2017.10.044 [Crossref] [ Google Scholar]
- Toiyama Y, Yasuda H, Saigusa S, Tanaka K, Inoue Y, Goel A. Increased expression of slug and vimentin as novel predictive biomarkers for lymph node metastasis and poor prognosis in colorectal cancer. Carcinogenesis 2013; 34:2548-57. doi: 10.1093/carcin/bgt282 [Crossref] [ Google Scholar]
- Faltejskova P, Besse A, Sevcikova S, Kubiczkova L, Svoboda M, Smarda J. Clinical correlations of miR-21 expression in colorectal cancer patients and effects of its inhibition on DLD1 colon cancer cells. Int J Colorectal Dis 2012; 27:1401-8. doi: 10.1007/s00384-012-1461-3 [Crossref] [ Google Scholar]
- Li X, Zhang H, Cui T, Wu Y, Wang S. MiR-143-5p inhibits proliferation, invasion, and epithelial to mesenchymal transition of colorectal cancer cells by downregulation of HMGA2. Trop J Pharm Res 2021; 20:1337-43. doi: 10.4314/tjpr.v20i7.3 [Crossref] [ Google Scholar]
- Wang J, Gao Y, Ma M, Li M, Zou D, Yang J. Effect of miR-21 on renal fibrosis by regulating MMP-9 and TIMP1 in kk-ay diabetic nephropathy mice. Cell BiochemBiophys 2013; 67:537-46. doi: 10.1007/s12013-013-9539-2 [Crossref] [ Google Scholar]
- Mesgarzadeh AH, Aali M, Farhadi F, Noorolyai S, Baghbani E, Mohammadnejad F. Transfection of microRNA-143 mimic could inhibit migration of HN-5 cells through down-regulating of metastatic genes. Gene 2019; 716:144033. doi: 10.1016/j.gene.2019.144033 [Crossref] [ Google Scholar]
- Hu J, Ni S, Cao Y, Zhang T, Wu T, Yin X. The angiogenic effect of microRNA-21 targeting TIMP3 through the regulation of MMP2 and MMP9. PLoS One 2016; 11:e0149537. doi: 10.1371/journal.pone.0149537 [Crossref] [ Google Scholar]
- Yun CW, Lee SH. The Roles of Autophagy in Cancer. Int J Mol Sci 2018; 19:3466. doi: 10.3390/ijms19113466 [Crossref] [ Google Scholar]
- Seca H, Lima RT, Lopes-Rodrigues V, Guimaraes JE, Almeida GM, Vasconcelos MH. Targeting miR-21 induces autophagy and chemosensitivity of leukemia cells. Curr Drug Targets 2013; 14:1135-43. doi: 10.2174/13894501113149990185 [Crossref] [ Google Scholar]
- Wei J, Ma Z, Li Y, Zhao B, Wang D, Jin Y. miR-143 inhibits cell proliferation by targeting autophagy-related 2B in non-small cell lung cancer H1299 cells. Mol Med Rep 2015; 11:571-6. doi: 10.3892/mmr.2014.2675 [Crossref] [ Google Scholar]