Bioimpacts. 12(1):9-20.
doi: 10.34172/bi.2021.23630
Review
Antiviral and anti-inflammatory drugs to combat COVID-19: Effects on cardiac ion channels and risk of ventricular arrhythmias
Luigi X Cubeddu 1, *  , Daisy de la Rosa 1, Michele Ameruoso 2
, Daisy de la Rosa 1, Michele Ameruoso 2
Author information:
1Nova SE University, Health Professions Division, 3200 S, University Drive, Davie, FL 33328, USA
2Laboratorios Pan-Quimica, Prados del Este, Caracas, Venezuela
Abstract
Introduction:
Drugs with no indication for the treatment of cardiovascular diseases (e.g., drugs employed to treat COVID-19) can increase the risk of arrhythmias. Of interest, a six-fold increase in the number of arrhythmic events was reported in patients with severe COVID-19. In this study, we reviewed (i) the pro-arrhythmic action of drugs given to patients with COVID-19 infection, and (ii) the effects of inflammatory cytokines on cardiac ion channels and possible generation of arrhythmias.
Methods:
We conducted a literature search on the drugs with purported or demonstrated efficacy against COVID-19 disease, emphasizing the mechanisms by which anti-COVID-19 drugs and inflammatory cytokines interfere with cardiac ion channels.
Results:
Antibiotics (azithromycin), antimalarials (hydroxychloroquine, chloroquine), antivirals (ritonavir/lopinavir, atazanavir), and some of the tyrosine kinase inhibitors (vandetanib) could induce long QT and increase risk for ventricular arrhythmias. The pro-arrhythmic action results from drug-induced inhibition of Kv11.1 (hERG) channels interfering with the repolarizing potassium IKr currents, leading to long QT and increased risk of triggered arrhythmias. At higher concentrations, these drugs may interfere with IKs, IK1, and/or Ito potassium currents, and even inhibit sodium (INa) and calcium (ICa) currents, inducing additional cardiac toxicity. Ibrutinib, an inhibitor of Bruton’s TK, increased the incidence of atrial fibrillation and ventricular tachycardia associated with a short QT interval. Inflammatory cytokines IL-6 and TNF-α inhibit IKr and Ito repolarizing potassium currents. High levels of inflammatory cytokines could contribute to the arrhythmic events. For remdesivir, favipiravir, dexamethasone, tocilizumab, anakinra, baricitinib, and monoclonal antibodies (bamlanivimab, etesevimab, and casirivimab), no evidence supports significant effects on cardiac ion channels, changes in the QT interval, and increased risk for ventricular arrhythmias.
Conclusion:
This study supports the concept of hERG channel promiscuity. Different drug classes given to COVID-19 patients might delay repolarization, and increase the risk of ventricular arrhythmias. The presence of comorbid pro-arrhythmic disease states, and elevated levels of pro-arrhythmic cytokines, could increase the risk of ventricular arrhythmias. Discontinuation of nonessential drugs and correction of electrolyte abnormalities could prevent severe ventricular arrhythmias. Altogether, the most effective therapies against COVID-19 (remdesivir, dexamethasone, monoclonal antibodies) lack pro-arrhythmic activity.
Keywords: Drugs and COVID-19, Torsade de Pointes, Torsade de Pointes, hERG potassium channels., Long QT, Cytokines, hERG potassium channels, Tyrosine kinase inhibitors
Copyright and License Information
© 2022 The Author(s)
This work is published by BioImpacts as an open access article distributed under the terms of the Creative Commons Attribution License (
http://creativecommons.org/licenses/by-nc/4.0/). Non-commercial uses of the work are permitted, provided the original work is properly cited.
Introduction
The severe acute respiratory syndrome caused by the new β-coronavirus named SARS-CoV-2, later labeled by the World Health Organization (WHO) as COVID-19, has reached pandemic proportions.
1
As of January 10, 2021, approximately 89 million subjects worldwide were reported infected with COVID-19, with 50 million patients recovered and 1.9 million deceased; a number that increased to 228 million and 4.6 million, respectively, by September 21, 2021.
1
The clinical presentation of COVID-19 infection varies from asymptomatic, to mild, moderate, and severe, and even death.
2
Besides the direct pathogenic actions of SARS-CoV-2 and its variants, an abnormal inflammatory, immune, and pro-coagulant response determines the severe course of the disease.
3
Due to the lack of effective treatments to treat severe COVID-19, the FDA issued compassionate and emergency use authorizations of drugs and antibodies to treat this novel disease. Consequently, early during the pandemic, patients received treatments whose proposed efficacy relied on in vitro antiviral activity, and on prior use in inflammatory and autoimmune diseases. A wide range of compounds, with different chemical structures and mechanisms of action, were and are being used, either alone or in combination, in an attempt to cure or minimize disease severity.
4-6
The list includes antibiotics (azithromycin, doxycycline), antimalarials (hydroxychloroquine, chloroquine), antivirals (ritonavir/lopinavir, remdesivir, favipiravir), anti-inflammatory drugs such as steroids (dexamethasone), IL-6 antagonists (tocilizumab), interleukin-1 (IL-1) antagonists (anakinra), June kinase-JAK1 and JAK2 inhibitors (baricitinib), tyrosine kinase inhibitors (TKIs) (ibrutinib), and monoclonal antibodies (bamlanivimab, etesevimab, casirivimab, and imdevimab). We learned that many of these agents were ineffective, with a high risk-to-benefit ratio. As of today, remdesivir and dexamethasone gained FDA approval, while monoclonal antibodies and baricitinib are under EUA.
Old age, hypertension, obesity, diabetes, cardiovascular disease, chronic lung disease, and immunosuppression predict the severe course of COVID-19, risk of hospitalization, admissions to ICU, and mortality.
2
Besides, associated comorbidities require additional drug therapies, often in high doses, increasing the risk of drug-induced toxicity. Several of the anti-COVID19 drugs interfere with cardiac ion channels, and their use is associated with severe ventricular arrhythmias
4-6
(Table 1, Fig. 1). Moreover, the pro-arrhythmic potential of drugs increases in patients with comorbidities known to predict severe disease.
7-9
In addition, in hospitalized COVID-19 patients, 16.7% experienced arrhythmias. Patients that were admitted to the ICU due to more severe illness had a greater incidence of arrhythmias than the non-ICU patients (44.4% vs 6.9%).
10
Because of its clinical relevance, we investigated the potential pro-arrhythmic action of drugs used in the treatment of COVID-19. Here, we discuss the cardiac channels and ion fluxes affected, and the mechanisms of channel inhibition when available, for drugs that were and are used to treat COVID-19 disease, including the role of inflammatory cytokines.
Table 1.
Effects on cardiac ion channels and pro-arrhythmic actions of drugs with purported or demonstrated efficacy in the treatment of COVID-19
|
Drug
|
Drug class
|
In vitro
effects on ion channels/currents
|
Arrhythmia reports
|
| Lopinavir, ritonavir, nelfinavir, saquinavir, atazanavir |
Antivirals/protease inhibitors |
Inhibition of KCNH2-hERG and
IKr
.
Atazanivir also decreased hERG channel density
|
Long QTc or TdP, or both reported. |
| Remdisivir, fapinavir |
Inhibitors of RNA polymerase |
No evidence found. One case report. Dedicated QT studies are needed. |
No reports |
| Chloroquine, OH-chloroquine |
Anti-malarial |
KCNJ2-Kir2.1-
IK1
,
KCNH2-hERG-Kv11.1-
IKr
,
At higher concentrations inhibits
INa
&
Ica
.
|
Reports of Long QT, T wave abnormalities, V Tach/TdP /cardiac arrest |
| Azithromycin |
Antibiotic |
KCNH2-hERG-
IKr
|
Long QT, T wave abnormalities, V Tach/TdP |
| IL-6, TNF-α, Anti-Ro/SSA antibodies, nuclear factor-β |
Cytokines/pro-inflammatory antibodies |
IL-6 inhibits
IKr
currents, and decreases KCNH2- hERG channel mRNA and protein expression (via JAK pathway and/or ROS production).
TNF-α inhibits
IKr
currents and reduces the amount of membrane hERG channels. TNF-α also inhibits
Ito
currents and the amount of α-subunit Kv4.2 protein.
Anti-Ro antibodies against antigen Ro52/TRIM21, inhibits KCNH2-hERG channels and
IKr
.
NF-β inhibited
Ito,f
.
|
|
| Anakinra, canakinumab, rinolacet |
Interfere with IL-1 actions |
No data found |
No reports |
| Sarilumab, siltuximab, tocilizumab |
IL-6 antagonists |
No evidence.
May shorten long QT.
|
No reports |
| Axitinib, cabozantinib imatinib, pazopanib, ponatinib, regorafenib, sorafenib, sunitinib, vandetanib, |
TKIs |
Inhibit hERG/KCNH2 channels and
IKr
.
Vandetanib also inhibits the
IKs
and
IK
1, INaand ICa,at higher concentrations.
Sunitinib also inhibits
INa
currents at higher conc.
|
Long QTc and increased risk for TdP and SCD. |
| Ibrutinib |
BKTI |
Disrupted mitochondrial and ER calcium handling |
Increases risk of A Fib, V tach, SV tach, HF, HT. Associated with short QT and delayed afterdepolarizations. |
| Baricitinib, ruxolutinib |
JAKI |
No evidence |
No reports |
Casirivimab + imdevimab
bamlanivimab + ezetivimab.
|
Monoclonal antibodies |
No evidence |
No reports |
Abbreviations: TKIs, tyrosine kinase inhibitors; BTKI, Bruton’s tyrosine kinase inhibitors; JAKI, janus kinase inhibitors; TNF, tumor necrosis factor; IL-6, interleukin-1.
Currents are shown in bold italic font. IKr: rapid inward rectifying potassium current that develops through phases 2 and 3 of the AP, and is needed for membrane repolarization. IKs: slow inward rectifying potassium current that develops through phases 2 and 3 of the AP, and is needed for membrane repolarization. IK1: inwardly rectifying current that develops after the AP, and maintains the negative resting membrane potential during diastole. Ito,f: transient outward rapid repolarizing potassium current, involved on the notch of the AP. INa:
rapid inward sodium current that determines the phase zero of the AP and membrane depolarization. ICa: voltage-dependent calcium current during the plateau of the AP, needed for contraction.
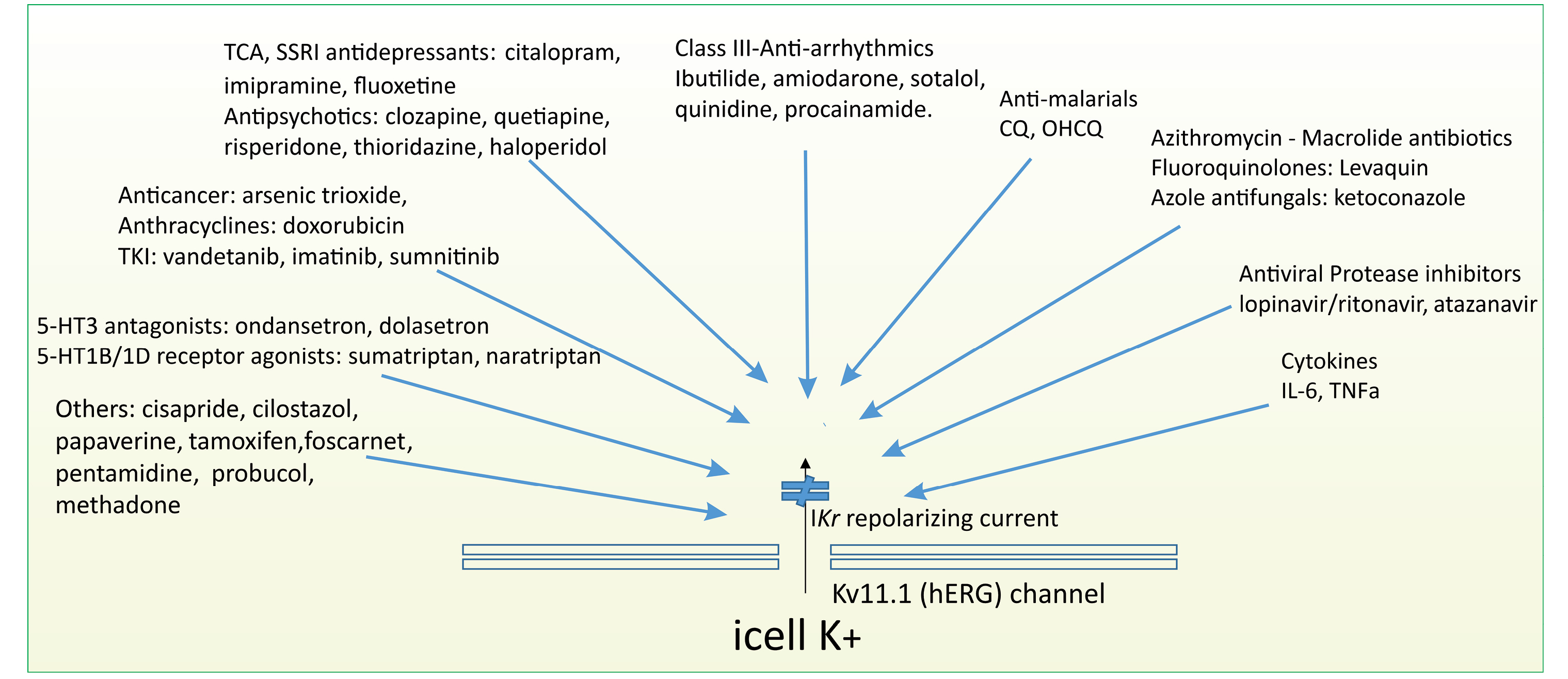
Fig. 1.
The promiscuity of the hERG channels: Inhibitors of the hERG (Kv11.1) channels and IKr currents. The drugs depicted in the figure have been shown to inhibit cardiac hERG (Kv11.1) channels and the IKr current that moves through the channels. Some prototype drugs are provided. For further details, see references 8 and 9. Abbreviations: VEGFR: vascular endothelial growth factor receptor; TKI, tyrosine kinase inhibitor; TCA: tricyclic antidepressants; SSRI, selective serotonin reuptake inhibitors; IL-6, Interleukin-6; TNFα: tumor necrosis alpha; 5-HT: 5-hydroxytryptamine (serotonin).
.
The promiscuity of the hERG channels: Inhibitors of the hERG (Kv11.1) channels and IKr currents. The drugs depicted in the figure have been shown to inhibit cardiac hERG (Kv11.1) channels and the IKr current that moves through the channels. Some prototype drugs are provided. For further details, see references 8 and 9. Abbreviations: VEGFR: vascular endothelial growth factor receptor; TKI, tyrosine kinase inhibitor; TCA: tricyclic antidepressants; SSRI, selective serotonin reuptake inhibitors; IL-6, Interleukin-6; TNFα: tumor necrosis alpha; 5-HT: 5-hydroxytryptamine (serotonin).
Torsade de Pointes, cardiac potassium-ion channels, long QT and SCD
Torsade de Pointes (TdP) is the most common cause of drug-induced ventricular tachycardia and sudden cardiac death (SCD). TdP presents as a polymorphic ventricular tachycardia, commonly most often accompanied by a long QTc interval. TdP most often results from inhibition of the IKr potassium currents conducted through the human ether-a-go-go-related gene (hERG) channels.
4,9
Multiple, structurally different drugs act upon the hERG channel to inhibit Ikr, (Fig. 1), prolonging repolarization. This manifests with widening the ventricular action potential, long QT, increased risk of afterdepolarizations, and ventricular arrhythmias. The long QTc is often associated with greater QT dispersion, irregularities of the T waves, presence of U waves, and of T wave alternant, which further increases the risk of developing TdP.
11
Ion channels are composed of alpha-subunits that are the pore-forming section of the channel. The alpha subunits together with the beta-subunits and other proteins form a complex, which regulates the channel activity. There are several potassium channels that open and close during the action potential.
4,12,13
For a normal function, the channels must open and close in a coordinated sequence. Inhibition, known as loss of function or activation (gain of function) of the channels, is linked to a greater risk of severe arrhythmias. The channels are identified by the gene encoding the channel, the alpha subunit (the pore or channel), and the current that carries it through.
13
Immediately following the upstroke of the action potential (phase 0) due to rapid entry of sodium (INa), two small potassium currents are activated, the Ito,f (fast) and the Ito,s (slow) currents producing the notch in action potential.
12,13
The KCND2 and KCND3 genes encode for the alpha subunits Kv4.2 and Kv4.3 responsible for the rapid repolarizing current Ito,f.
14
The KCNA4 gene encodes for the channel Kv1.4, responsible for the transient outward of the slow repolarizing current, Ito,s.
3,12
Quinidine exerts inhibitory actions on these channels prolonging repolarization.
15
IKr is a rapid inward rectifier repolarizing current, that runs through phase 2 and the beginning of phase 3 of the action potential. This current moves through the alpha subunit pore Kv11.1. The KCNH2/hERG gene encodes the Kv11.1subunit.
12,13
Inhibition ofIKr delays the process of repolarization, which manifests with a long QTc interval on the ECG. The slow repolarization allows sufficient time for the inactive calcium channels to reactivate, leading to early calcium influx (early afterdepolarizations) that, if of sufficient magnitude, may trigger an arrhythmia.
4,9
Kv11.1-channel inhibition or decreases in the number of mature functional channels is associated with long QTc
3,14,16
The effect of IKr inhibitors increases at low heart rates and low extracellular potassium concentrations. This explains the greater QTc prolongation and increased risk of TdP reported when hERG channel inhibitors are given in patients with bradycardia and/or hypokalemia.
17
Drugs may directly inhibit the hERG channels and/or decrease the amount of functional, mature, channels in the plasma membrane, decreasing IKr currents.
4,8,9
The number of mature channels is determined by the rates of synthesis, trafficking, and degradation of the channels. The half-life of the hERG channels is nearly 11 hours.
18
The channels are glycosylated in the endoplasmic reticulum (core glycosylation) and the Golgi apparatus (full glycosylation), followed by insertion in the cell membrane. Channel degradation occurs by ubiquitination at the ER and subsequent proteasomal degradation. Caveolin-3 acts through the ubiquitin-ligase Nedd4-2 to regulate the amount of mature hERG channels, increasing ubiquitination and subsequent degradation.
19,20
Besides, rififylin, the RING finger ubiquitin ligase, can participate in the process of hERG channel-ubiquitination.
21
Mutations in the KCNH2-hERG gene may produce a mutated channel, which cannot be appropriately trafficked to the cell membrane. The reduced number of functional channels resultant from the gene mutation, clinically known as LQTS2, is characterized by the presence of ventricular arrhythmias and reports of SCD.
22
IKs is known as the slowly activating delayed rectifier repolarizing current. This current moves through the channel-alpha subunit, Kv7.1, which, in turn, is encoded by the KCNQ1 gene. The IKs current plays a role in cardiac repolarization. Gene mutations can affect the function of the channel decreasing Ikscurrent, delaying repolarization. Such mutation presents as the LQTS1. Drugs, although less frequently and often at doses higher than needed to inhibit IKr, may inhibit the IKs current.
4
IK1 is an inwardly rectifying current that develops during diastole, keeping negative the resting membrane potential. Kir2.1. is the alpha-pore-forming subunit of the channel, which is encoded by the KCNJ2 gene. Bidirectional polymorphic ventricular tachycardia, associated with repolarization abnormalities (long QTc, large U waves, frequent PVCs), are reported in patients harboring loss of function mutations in the KCNJ2 gene. Inhibition of IK1 by hypokalemia and stimulation of beta-receptors inhibit IK1 currents, increasing the risk of arrhythmias.
23,24
In summary, inhibition of IKr, IKs, IK1, and/or Ito currents interfere with membrane repolarization, lead to long QT, and favor the development of after-depolarization, thereby increasing the risk of triggering ventricular tachycardia, TdP, and SCD. Direct inhibition of hERG channels and reduced number of hERG channels (Kv11.1,), are the commonest cause of drug-induced and congenital LQTS, respectively, and severe ventricular arrhythmias (Fig. 1).
Pro-arrhythmic potential of drugs used for the treatment of COVID-19 disease
Antivirals, long QT and risk of TdP and SCD
Protease inhibitors
Protease inhibitors, which inhibit viral protein synthesis, have been employed to treat patients with COVID-19.
24
By inhibiting 3-chymotrypsin-like protease, the combination of lopinavir and ritonavir has been shown to exert in vitro activity against novel coronaviruses.
25
Despite initial expectations, a recent randomized control trial on the use of lopinavir/ritonavir, failed to show survival benefits.
25
The use of protease inhibitors was associated with QTc prolongation, and/or TdP.
26
However, in HIV patients treated with protease inhibitors, the presence of long QTc was unrelated to drug use, and was related to the length of HIV infection and to the presence of known factors that lead to long QT.
27,28
Further, in patients with severe COVID, the lopinavir-ritonavir combination did not induce QT prolongation or serious arrhythmic events.
29
Studies in HEK293 cells and cardiac myocytes showed that saquinavir, lopinavir, ritonavir, and nelfinavir have inhibitory actions on IKr.
26
Atazanavir inhibits hERG channels and Ikrcurrents, likely by binding to the S6 channel-domain (residues Y652 and F656).
30
In addition, with longer times of exposure, atazanavir interferes with channel glycosylation, reduces the quantity of membrane hERG that is fully glycosylated. Such dual-action, channel inhibition, and reduced number of mature channels in the membrane may account for the potent inhibitory actions of atazanavir on Ikrcurrent.
9,31
Besides, protease inhibitors can increase the levels of reactive oxygen species (ROS), which may exert inhibitory and stimulatory actions on several ion channels and transporters.
31-33
In cardiac myocytes, protease inhibitors induce depolarizing chloride outward currents due to increases in mitochondrial ROS, which may play a pro-arrhythmic role.
31
In summary, more evidence suggests that protease inhibitors can inhibit the hERG channel, leading to long QT, and increase the risk of patients to TdP (Table 1). Monitoring the QT interval is recommended in patients with COVID-19 treated with protease inhibitors, in particular when combined with azithromycin, CQ, or OHCQ, or with any QT-prolonging drugs.
26
RNA polymerase Inhibitors
Remdesivir and Favipiravir inhibit viral RNA polymerase interfering with RNA Synthesis. Both drugs are broad-spectrum antivirals. Remdesivir is an adenosine nucleotide analog, RdRp inhibitor, and Favipiravir, a guanosine nucleotide analog, RdRp inhibitor.
33,34
In particular, remdesivir is employed for the treatment of COVID-19.
25,34-36
In vitro studies demonstrated that remdesivir is a potent inhibitor of coronaviruses. Remdesivir inhibits SARS-CoV-2 with EC50 and EC90 values of 0.77 μM and 1.76 μM, respectively.
37,38
Concentrations 5-10 times higher than remdesivir’ s EC90 for COVID-19 are reached during the initial 200 mg IV infusion over 30 minutes.
No reports of QT prolongation, TdP, or cardiac arrhythmias were found for remdesivir and favipiravir (Table 1). A QTc study in Japanese adults found no effects of favipiravir.
39
Two cases of QTc prolongation were reported, one during favipiravir therapy in one Ebolavirus-infected patient,
40
and another in one patient infected with COVID-19 during remdesivir therapy
41
; however, this patient was also treated with azithromycin. In summary, we found no studies other than these two case reports, to suggest that RNA polymerase inhibitors prolong the QTc interval, or that these drugs interfere with cardiac ion channels. Dedicated studies on the potential effects of remdesivir and favipiravir on the QT interval are needed.
Chloroquine and hydroxychloroquine
Chloroquine (CQ) and hydroxychloroquine (OHCQ) have been used to treat COVID-19 infection.
37
OHCQ and CQ inhibit the synthesis of sialic acids, affecting protein glycosylation and disrupting SARS-CoV-2 attachment and entry into cells.
37,38,42
Initially, these drugs were extensively used; however, recent evidence suggests that CQ and OHCQ are infective in the patients that are hospitalized with COVID-19. The US FDA revoked its authorization for emergency use to treat hospitalized patients with COVID-19.
43
SARS-CoV-2 entry into lung epithelial cells seems not to require low-pH processing in endosomes, which may account for the poor efficacy of CQ and OHCQ against COVID-19 disease in clinical trials.
The cardiovascular adverse events reported with CQ and OHCQ are TdP, long QT, hypotension, and ventricular tachycardia
37
(Table 1). Patients experiencing an overdose may present cardiovascular collapse, cardiac arrest, and hypokalemia. Cardiovascular side effects occurring less than 0.1%, include hypotension, cardiomyopathy, T wave inversion, or depression with QRS widening.
44
The effects of CQ and OHCQ on cardiac ion channels could account for the reported cardiac adverse events. CQ inhibits the IK1 and the hERG channel IKr currents (Table 1). At higher and toxic concentrations,CQ can also block sodium and calcium channels. Contrary to most of the hERG blockers and ion-channels blockers that block the currents acting at the transmembrane site, CQ inhibits the potassium channel acting on the cytoplasmic side.
45
CQ binds to the acidic residues (Glu224, Glu299, Asp255, Asp259, and Phe 254) located in the cytoplasmic side of the channel pore.
45
Thus, the long QTc and TdP reported with these drugs likely result from inhibition of potassium currents.
46
HERG inhibition is not limited to CQ and OHCQ, since it is observed with most antimalarial drugs.
3,47
Azithromycin
Azithromycin, as well as other macrolide antibiotics, has been reported to induce long QTc and to increase the risk of ventricular arrhythmias due to inhibition of the hERG (Kv11.1) channel and the Ikrcurrent
48,49
(Table 1). Besides, azithromycin increases the dispersion of repolarization, favoring the development of TdP.
50
Adverse event reports from 2004 to 2011 revealed a total of 203 events, long QT-TdP (63 cases), and ventricular tachycardia or SCD (140 cases), likely leading to 65 deaths that were associated with azithromycin treatment.
51
Prolongation of the QRS interval and complete heart block were also reported as adverse events.
52
Retrospective, observational studies revealed that use of azithromycin, moxifloxacin, and levofloxacin was connected with an increase in the risk of cardiovascular events, including a ventricular arrhythmias.
53,54
However, in a collection of 12 case reports of possible azithromycin-induced/associated QTc interval prolongation and TdP, there was no association between the duration of the QTc and the dose of azithromycin employed.
53
Moreover, causality was not proven, since all patients had additional risk factors for long QTc.
54
An increased risk in cardiovascular deaths associated with azithromycin use was reported in a retrospective study in young adults; however, the study design did not allow to determine whether the events were due to the azithromycin, the comorbid conditions associated with its use, or combination of both factors.
55
In summary, the increased risk in cardiovascular events and mortality reported with azithromycin use is greater in patients with multiple preexisting cardiovascular comorbidities, electrolyte abnormalities, and complex medication regimens. Consequently, a higher risk is expected when azithromycin is used in COVID-19 patients. These patients requiring close ECG monitoring, mainly in the presence of heart failure, ischemic heart disease, electrolyte abnormalities, and in those treated with anti-COVID-19 long QT drugs, such as CQ, OHCQ, protease inhibitors such as lopinavir/ritonavir, TKIs, and the Bruton’s tyrosine kinase inhibitors (BTKI) (Table 1).
Combination of OHCQ, azithromycin, and protease inhibitors
A combination of long QT drugs should be avoided since it is associated with a greater risk of triggering TdP.
4
In a randomized trial in subjects treated with OHCQ (1000 mg/qd), the addition of azithromycin prolonged the QTc interval in a dose-dependent manner, from 10 to 14 milliseconds.
56
Reports of long QT and/or ventricular tachycardia including TdP were more common with azithromycin monotherapy than with OHCQ monotherapy and increased with combination treatment.
57
The need for patient monitoring is known to prevent TdP. In a retrospective study of COVID-19 patients, Jain and co-authors
58
reported that close monitoring of QT and associated risk factors prevented the development of arrhythmic events. The authors analyzed over 2000 ECGs from 524 patients, most with COVID-19 infection. Patients who developed long QT were managed with electrolyte correction and by stopping nonessential long-QT drugs.
In COVID-19 patients, the use of OHCQ combined with either atazanavir or azithromycin required preventive discontinuation of OHCQ in nearly one-third of the patients. None of their patients developed TdP. However, severe QT prolongation (QTc >500 ms) was seen in 11% of patients treated with OHCQ
51
and in 9%
52
of those treated with azithromycin drugs discontinuation was reported in 3.5% of the patients,
52
and no TdP or arrhythmic deaths were reported.
59,60
In summary, studies in non-COVID-19 and COVID-19 patients ratify that both OH-QC, protease inhibitors, and azithromycin prolong the QT interval and that their combination produces greater QT lengthening, with the associated increased risk of developing TdP (Table 1). Close monitoring of these patients, discontinuation of nonessential drugs, and correction of electrolyte abnormalities is recommended and was successfully shown to prevent the development of severe ventricular arrhythmias.
58
Inflammation and long QT: IL-1 and IL-6 antagonists
COVID-19 hyperinflammatory syndrome
COVID-19 severity and outcomes seem to be determined by the patient’s immune-inflammatory response. Exaggerated production of ILs and tumor necrosis factor α (TNF-α) are involved in the multisystem inflammatory syndrome reported in COVID-19 patients with severe disease.
3
Increased levels of inflammatory cytokines, ferritin, and D-dimer, associated with lymphocytopenia and thrombocytopenia, are present in these patients.
3
Severely affected COVID-19 patients may present features suggestive of the cytokine storm syndrome, and occasionally features of the macrophage activation and secondary hemophagocytic-lymphohistiocytosis syndromes.
3,61-63
These conditions are characterized by very high levels of interleukins, interferons, and of TNF–α.
62
These pathophysiological considerations account for the ongoing clinical trials testing the use of IL-6 and IL-1 antagonists, and of drugs that interfere with JAK kinases in the management of COVID-19 patients with severe disease.
61-63
Long QT in inflammatory syndromes: Effects of interleukin antagonists
Cardiac muscle and conduction system abnormalities are described in animal models of Kawasaki disease. Mice injected with Lactobacillus caseicell-wall extract developed tachycardia and decreased R-wave amplitude, prolonged ventricular repolarization, and increased neural remodeling of the myocardium. Treatment withIL-1 signaling inhibitor, anakinra, for four days, corrected theECG abnormalities and prevented the ventricular remodeling.
64,65
Patients with rheumatoid arthritis (RA) and chronic systemic inflammation often present with long QTc.
66-68
The lengthening of the QTc interval and development of TdP were positively related to the circulating levels of IL6, IL1β, TNFα and C-reactive protein (CRP).
68,69,70
At pretreatment, nearly three-fourth of the RA patients showed QTc prolongation (452.3 ± 35.8 ms). In these patients, tocilizumab (anti-IL-6 therapy) shortened the QTc interval to 428.1 ± 34.3 milliseconds, with all patients having QTc intervals shorter than 440 milliseconds. Shortening the QT interval was related to lowering in the TNFα and CRP levels.
71
Of interest, nearly 10% of patients with SLE had longer QTc than healthy controls, and around 3% of the SLE patients showed marked QTc prolongation (>500 ms). The QTc prolongation was directly associated with inflammatory markers and disease activity.
69,70
Notably, in these patients, anti-IL-6 therapy with tocilizumab resulted in QTc shortening.
72
Cytokines and cardiac channels
The effects of cytokines on cardiac ion channels support the above-described observations of long QT in RA and SLE patients (Table 1). In HEK transfected cells with hERG channels, and ventricular myocytes, IL-6 inhibitedIKr currents, decreased KCNH2- hERG channel mRNA and protein expression, and prolonged the duration of the ventricular action potential.
73
IL-6 receptors and the Janus kinase (JAK) pathways mediated the inhibition of hERG induced by IL-6. Besides the JAK pathway, increases in ROS and ceramide signaling pathways may also mediate the effects of the cytokines on the ionic currents and prolongation of the QTc interval.
TNF-α was also shown to inhibit IKrcurrents in HEK293 cells and ventricular myocytes
73
and to decrease the amount of mature hERG channels on the cell membrane.
74,75
In addition, in rat ventricular myocytes, TNF-α inhibited Ito currents and the amount of Kv4.2 in the cell membrane,
76
and nuclear factor-β decreased Ito,f acting on KChIP2 expression.
77
Besides the cytokines, anti-Ro antibodies (against antigen Ro52/TRIM21), obtained from patients with autoimmune disease, inhibited IKrcurrent acting on KCNH2-hERG channels.
78,79
Anti-Ro/SSA antibodies, of the 52kD subtype, seem to be responsible for an immune-mediated LQTS, acting the hERG-Kv11-1 channel pore
80
(Table 1).
In summary, in vitro studies on ion channels and clinical observations in patients with inflammatory syndromes suggest that the long QTc and the increased risk of arrhythmia observed is associated with high levels of cytokines. These effects are more likely due to the inhibition of hERG function and channel synthesis, and the interference with Ito currents. Consequently, it is not surprising that anti-cytokine therapies may shorten the QTc interval.
71,72
We found no reports of QT prolongation or TdP associated with the use of the anakinra, an antagonist of IL1 receptors, with rinolacept, a soluble decoy receptor, canakinumab, a monoclonal anti-IL-1β antibody, and for the human monoclonal antibodies against IL-6 receptor, tocilizumab, sarilumab, and siltuximab (Table 1).
Tyrosine kinase inhibitors
Tyrosine kinases stimulate the phosphorylation of proteins transferring phosphate groups to their tyrosine residues, regulating cell growth, migration, differentiation, and death; effects that are responsible for the use of TKI in different malignancies. Some of the TKIs exert in vitro inhibitory activity against SARS-CoV-2.
81
An additional important aspect is the use of TKI in cancer patients infected with COVID-19.
Cardiac toxicity has been reported in patients treated with TKI.
3,81,82
Treatment with VEGFR-TKIs (sunitinib, axitinib, vandetanib, sorafenib, pazopanib, cabozantinib, ponatinib, and regorafenib) was associated with a four-time greater risk for QTc prolongation
83-85
(Table 1). Due to the increased risk of arrhythmia and SCD, vandetanib use is restricted via a Risk Evaluations and Mitigation Strategy program.
82
Vandetanib must not be given combined with other long QT drugs. One-third of cancer patients treated within imatinib, nilotinib, or dasatinib developed QT prolongation during drug administration. QTc intervals longer than 500 milliseconds were found in 53 administrations, ventricular tachycardia in nine, TdP in two, and SCD in three administrations.
84
Vandetanib inhibits hERG/KCNH2 channels decreasing IKr current amplitude, leading to a long QT. At higher concentrations, it inhibits the IKs and IK1 potassium currents, interfering with INa and ICa currents at high micromolar concentrations.
86
Similarly, sunitinib was shown to inhibit IKr currents at low micromolar and INa currents at higher micromolar concentrations.
87
Imatinib mesylate inhibited IKr in HEK-293 cells and Xenopus oocytes
88
(Table 1).
In summary, TKI preferentially inhibits the hERG-Kv11.1 channels and the corresponding IKrcurrents, and interferes with other potassium, sodium, and calcium currents at higher concentrations. Treatment with TKI is reported associated with long QTc and increased risk of TdP and SCD. Caution should be exerted and monitoring should be implemented during TKI administration. The combination of TKI with other long QT drugs should be avoided, and patients with comorbidities and electrolyte disturbances associated with long QT should be carefully monitored.
Bruton’s tyrosine kinase inhibitors
BTK is a member of the Tec family of non-receptor tyrosine kinases. BKTs are involved in the B-cell signaling pathways involved in the growth and maturation of the B-cells. Inhibition of B-cell function may theoretically reduce the hyper-inflammatory immune response observed in severely ill patients with COVID-19.
Ibrutinib is being studied in hospitalized patients with COVID-19.
89,90
Ibrutinib is the first human BTK inhibitor approved by the FDA.
91
However, an increased risk of atrial and ventricular arrhythmias, sinoatrial arrest, heart failure, hypertension, and SCD was reported with ibrutinib.
3,92-94
In patients with leukemia, atrial fibrillation was the most common cause of treatment discontinuation.
95,96
A 4-9 fold increase in the incidence of atrial fibrillation was reported with ibrutinib.
97
Noteworthy, the increased incidence of ventricular arrhythmias seemed not associated with QT prolongation.
98
In fact, episodes of polymorphic ventricular tachycardia occurred with a normal QTc interval, and with a short-coupled variant before the arrhythmia.
94
Besides, QT shortening has been described with ibrutinib.
95
It was proposed that this type of ventricular tachycardia is the consequence of disrupted calcium handling in the myocardium, favoring delayed afterdepolarization
99
(Table 1). Further studies are required to determine the mechanism of the pro-arrhythmic action of ibrutinib.
In summary, ibrutinib use is associated with an increased number of cardiac arrhythmias, such as atrial fibrillation, ventricular and supraventricular arrhythmias, and with a greater risk of heart failure and hypertension. Current evidence suggests that the ventricular polymorphic arrhythmias reported with ibrutinib occur in the absence of a long QT, but rather with either a normal or a short QT interval. Patients on ibrutinib therapy must be carefully monitored.
Janus kinase inhibitors (JAKI)
Baricitinib and ruxolitinib are selective inhibitors of the JAK1 and JAK2 enzymes, inhibiting the JAK-STAT signaling pathway. Its main clinical use is in the treatment of adult patients with moderately to severely active RA.
99
Besides its anti-cytokine activity, baricitinib may exert antiviral effects. Baricitinib has an affinity for AP2-associated protein AAK1, interfering with the endocytosis of SARS-CoV-2.
100
In an open pilot study conducted in hospitalized with moderate pneumonia due to COVID-19 pneumonia, baricitinib combined with lopinavir/ritonavir improved clinical and laboratory parameters, no patient required ICU support, and most patients were discharged in good health.
101
Besides, in hospitalized patients with COVID-19, baricitinib in combination with remdesivir was more effective than remdesivir alone in reducing the time to recover from COVID-19 infection.
102
Baricitinib inhibits hERG channels at high concentrations. Its IC50% is in the range of 50 to 60 μg/mL. Steady-state levels obtained with an 8 mg daily dose were 1000 times lower than those required to inhibit hERG.
103
No significant QTc prolongation was reported with a single dose of 40 mg.
103
We found no published reports suggestive of baricitinib-induced QT prolongation, of its effects on potassium channels and cardiac ion channels. In summary, although more studies are needed, baricitinib appears to have a low potential for inducing long QT and associated TdP (Table 1).
Monoclonal antibodies: casirivimab-imdevimab and bamlanivimab-etesevimab
Casirivimab (REGN10933) and imdevimab (REGN10987) are two human monoclonal antibodies. These antibodies were constructed to bind to two different areas of the spike protein of SARS-CoV-2, and are expected to inhibit virus’ binding and entry into cells. When employed in patients with COVID-19 and at high risk for severe disease, the antibody combination reduced the viral load (less virus penetrates the cells, reducing viral replication and infectivity) and the number of hospitalizations and visits to the ED.
104
Bamlavinimab (LY-CoV555) either alone or preferably in combination with etesevimab, are also neutralizing human monoclonal IgG1 antibodies, which to the receptor-binding domain of the spike protein of COVID-19.
The FDA has granted the EUA for the use of the monoclonal antibodies as a single IV administration, in non-hospitalized patients with mild to moderate COVID-19, at high risk for progressing to severe COVID-19, and as post-exposure prophylaxis.
105
The latter was defined as been 65 or older, and/or with chronic medical conditions. We found no reports of QT prolongation, ventricular tachycardia, or TdP during or following administration of these monoclonal antibodies (Table 1). Additional studies are needed to draw firm conclusions about any adverse cardiac toxicity.
Concluding Remarks
It is of pharmacological and clinical significance the observed lack of apparent selectivity to inhibition of the hERG channels by a large number of structurally different pharmacological agents. Most of the drugs that inhibit hERG channels seem to act at the channel pore tetramer. Channel opening during membrane depolarization seems required for the binding of most channel inhibitors (Table1, Fig. 1). Therefore, it was not unexpected that several of the drugs with purported anti-COVID-19 activity would inhibit the hERG channels, increasing the risk of cardiac arrhythmias. Furthermore, some of these agents, particularly at higher doses, may also interfere with other potassium (IK1, IKs, Ito) currents, as well as withsodium (INa), and even calcium (ICa) currents, increasing their cardiac toxicity. Further studies on the molecular and electrical gating mechanism modulating hERG gating mechanisms are required to further understand the mechanism of hERG channel promiscuity.
Inhibition of the rapid depolarizing IKr current prolongs the repolarization time, which manifests with a long QTc interval on the ECG. In high-risk patients, prolonged repolarization facilitates the development of early afterdepolarizations, which may induce a ventricular arrhythmia that may be severe and even fatal. Antiretroviral drugs, azithromycin, CQ and OHCQ, and some of the TKIs inhibit the IKrcurrent. Inhibition of IKris achieved by direct drug action on the hERG channel, and/or by a reduced amount of channels in the cell membrane. Decreased synthesis, faulty channel trafficking, and glycosylation, and/or increased degradation can reduce the number of mature membrane channels. Of interest, Ibrutinib, a BTKI, increases the risk of atrial fibrillation, and ventricular tachycardia not associated with long QTc, but rather with a short QTc. In addition to the above-mentioned drugs, the cytokine storm-hyper-inflammatory response, described in severe COVID-19 infection, favors arrhythmogenesis. High concentrations of inflammatory cytokines, IL-6, TNF-α, and nuclear factor β can inhibit the IKr and the Ito currents, which are essential for normal repolarization, and may account for the 6-fold greater incidence of arrhythmias reported in the more severely ill COVID-19 patients.
In summary, besides drug therapy, older age, presence of high-risk comorbidities, excessive immune-inflammation, and severe COVID-19 disease can additively inhibit cardiac ion channels, increasing the risk for ventricular arrhythmias. Avoiding therapies with a high risk/ benefit ratio for arrhythmogenesis, strict patient EKG monitoring for QT changes, maintaining K+ and Mg++ concentration within normal range, avoiding long-QT drug combination, and use of effective anti-inflammatory therapies to lower cytokine levels (dexamethasone) are the mainstay for the prevention of triggered arrhythmias in COVID-19 patients.
Funding sources
None.
Ethical statement
Not applicable.
Competing interests
There are no competing interests.
Authors’ contribution
DdlR contributed to the bibliographic search, bibliographic organization, and writing of the manuscript. MA contributed to the safety evaluation of anti-COVID-19 drugs. LXC initiated and organized the study, conducted the data analysis, and contributed to the writing and reviewing of the manuscript.
Review Highlights
What is the current knowledge?
simple
-
√ The severe acute respiratory syndrome, named COVID-19, is caused by CoV-2, and is characterized by high infectiveness, morbidity, and mortality.
-
√ The lack of an effective cure led to compassionate and emergency use approval of therapies with insufficient efficacy and safety data.
-
√ Arrhythmic events are more frequent in severely ill, hospitalized, COVID-19 patients.
-
√ Drug-induced inhibition of hERG potassium channels is the most common cause of long QT, and TdP.
What is new here?
simple
-
√ The following drugs with proposed anti-COVID-19 activity inhibit hERG channels and increase the risk of TdP: azithromycin, OHCQ, CQ, lopinavir/ritonavir/atazanavir, TKI (i.e., vandetanib). The BTKI, Ibrutinib, increases the risk of atrial fibrillation and ventricular arrhythmias. The mechanisms of channel inhibition are discussed.
-
√ In addition to the above-mentioned drugs, endogenous cytokines, IL-6 and TNF-α can inhibit hERG channels interfering with potassium currents. When associated with pro-arrhythmic drugs, high levels of cytokines observed in severe COVID-19 can further increase the risk of severe arrhythmias.
-
√ No evidence of pro-arrhythmic activity was found for remdesivir, dexamethasone, baricitinib, and human monoclonal antibodies (bamlanivimab + etesevimab, and casirivimab + imdevimab).
-
√ In addition to drugs with pro-arrhythmic activity, patients’ old age, cardiac comorbidities, and extensive inflammation can explain the increased arrhythmic events reported in patients with severe COVID-19 disease.
References
-
WHO coronavirus disease (COVID-19) dashboard. World Health Organization; 2021 [updated 2021 January 10; cited 2021 January 11 ]; Available from: https://covid19.who.int/.
- Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020; 584:430-6. doi: 10.1038/s41586-020-2521-4 [Crossref] [ Google Scholar]
- Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 2020; 395:1033-4. doi: 10.1016/S0140-6736(20)30628-0 [Crossref] [ Google Scholar]
-
Simpson TF, Kovacs RJ, Stecker EC. Ventricular arrhythmia risk due to hydroxychloroquine-azithromycin treatment for COVID-19. American College of Cardiology; 2020 March 29 [cited 2020 March 31]; Available from: https://www.acc.org/latest-in-cardiology/articles/2020/03/27/14/00/ventricular-arrhythmia-risk-due-to-hydroxychloroquine-azithromycin-treatment-for-covid-19.
- Malviya A. Ventricular arrhythmia risk due to chloroquine / hydroxychloroquine treatment for COVID-19: should it be given. Indian Heart J 2020; 72:131-2. doi: 10.1016/j.ihj.2020.04.006 [Crossref] [ Google Scholar]
- Roden DM, Harrington RA, Poppas A, Russo AM. Considerations for drug interactions on QTc interval in exploratory COVID-19 treatment. J Am Coll Cardiol 2020; 75:2623-4. doi: 10.1016/j.jacc.2020.04.016 [Crossref] [ Google Scholar]
- Roden DM. Long QT syndrome and tosades de pointes: basic and clinical aspects. In: El-Sherif N, P Samet, editors. Cardiac Pacing and Electrophysiology. 3rd ed. Philadelphia: W.B. Saunders; 1991. p. 265-84.
- Cubeddu LX. Iatrogenic QT abnormalities and fatal arrhythmias: mechanisms and clinical significance. Curr Cardiol Rev 2009; 5:166-76. doi: 10.2174/157340309788970397 [Crossref] [ Google Scholar]
- Cubeddu LX. Drug-induced inhibition and trafficking disruption of ion channels: pathogenesis of QT abnormalities and drug-induced fatal arrhythmias. Curr Cardiol Rev 2016; 12:141-54. doi: 10.2174/1573403X12666160301120217 [Crossref] [ Google Scholar]
- Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 2020; 323:1061-9. doi: 10.1001/jama.2020.1585 [Crossref] [ Google Scholar]
- Tran HT, Chow MS, Kluger J. Amiodarone induced torsades de pointes with excessive QT dispersion following quinidine induced polymorphic ventricular tachycardia. Pacing Clin Electrophysiol 1997; 20:2275-8. doi: 10.1111/j.1540-8159.1997.tb04249.x [Crossref] [ Google Scholar]
- Jeevaratnam K, Chadda KR, Huang CL, Camm AJ. Cardiac potassium channels: physiological insights for targeted therapy. J Cardiovasc Pharmacol Ther 2018; 23:119-29. doi: 10.1177/1074248417729880 [Crossref] [ Google Scholar]
- Grant AO. Cardiac ion channels. Circ Arrhythm Electrophysiol 2009; 2:185-94. doi: 10.1161/circep.108.789081 [Crossref] [ Google Scholar]
- Splawski I, Shen J, Timothy KW, Lehmann MH, Priori S, Robinson JL. Spectrum of mutations in long-QT syndrome genes KVLQT1, HERG, SCN5A, KCNE1, and KCNE2. Circulation 2000; 102:1178-85. doi: 10.1161/01.cir.102.10.1178 [Crossref] [ Google Scholar]
- Yatani A, Wakamori M, Mikala G, Bahinski A. Block of transient outward-type cloned cardiac K+ channel currents by quinidine. Circ Res 1993; 73:351-9. doi: 10.1161/01.res.73.2.351 [Crossref] [ Google Scholar]
- Adler A, Novelli V, Amin AS, Abiusi E, Care M, Nannenberg EA. An international, multicentered, evidence-based reappraisal of genes reported to cause congenital long QT syndrome. Circulation 2020; 141:418-28. doi: 10.1161/circulationaha.119.043132 [Crossref] [ Google Scholar]
- Yang T, Roden DM. Extracellular potassium modulation of drug block of IKr Implications for torsade de pointes and reverse use-dependence. Circulation 1996; 93:407-11. doi: 10.1161/01.cir.93.3.407 [Crossref] [ Google Scholar]
- Ficker E, Dennis AT, Wang L, Brown AM. Role of the cytosolic chaperones Hsp70 and Hsp90 in maturation of the cardiac potassium channel HERG. Circ Res 2003; 92:e87-100. doi: 10.1161/01.Res.0000079028.31393.15 [Crossref] [ Google Scholar]
- Guo J, Wang T, Li X, Shallow H, Yang T, Li W. Cell surface expression of human ether-a-go-go-related gene (hERG) channels is regulated by caveolin-3 protein via the ubiquitin ligase Nedd4-2. J Biol Chem 2012; 287:33132-41. doi: 10.1074/jbc.M112.389643 [Crossref] [ Google Scholar]
- Kang Y, Guo J, Yang T, Li W, Zhang S. Regulation of the human ether-a-go-go-related gene (hERG) potassium channel by Nedd4 family interacting proteins (Ndfips). Biochem J 2015; 472:71-82. doi: 10.1042/bj20141282 [Crossref] [ Google Scholar]
- Roder K, Werdich AA, Li W, Liu M, Kim TY, Organ-Darling LE. RING finger protein RNF207, a novel regulator of cardiac excitation. J Biol Chem 2014; 289:33730-40. doi: 10.1074/jbc.M114.592295 [Crossref] [ Google Scholar]
- Keller DI, Grenier J, Christé G, Dubouloz F, Osswald S, Brink M. Characterization of novel KCNH2 mutations in type 2 long QT syndrome manifesting as seizures. Can J Cardiol 2009; 25:455-62. doi: 10.1016/s0828-282x(09)70117-5 [Crossref] [ Google Scholar]
- Sung RJ, Wu SN, Wu JS, Chang HD, Luo CH. Electrophysiological mechanisms of ventricular arrhythmias in relation to Andersen-Tawil syndrome under conditions of reduced IK1: a simulation study. Am J Physiol Heart Circ Physiol 2006; 291:H2597-605. doi: 10.1152/ajpheart.00393.2006 [Crossref] [ Google Scholar]
- Morita H, Zipes DP, Morita ST, Wu J. Mechanism of U wave and polymorphic ventricular tachycardia in a canine tissue model of Andersen-Tawil syndrome. Cardiovasc Res 2007; 75:510-8. doi: 10.1016/j.cardiores.2007.04.028 [Crossref] [ Google Scholar]
- Şimşek Yavuz S, Ünal S. Antiviral treatment of COVID-19. Turk J Med Sci 2020; 50:611-9. doi: 10.3906/sag-2004-145 [Crossref] [ Google Scholar]
- Anson BD, Weaver JG, Ackerman MJ, Akinsete O, Henry K, January CT. Blockade of HERG channels by HIV protease inhibitors. Lancet 2005; 365:682-6. doi: 10.1016/s0140-6736(05)17950-1 [Crossref] [ Google Scholar]
- Charbit B, Rosier A, Bollens D, Boccara F, Boelle PY, Koubaa A. Relationship between HIV protease inhibitors and QTc interval duration in HIV-infected patients: a cross-sectional study. Br J Clin Pharmacol 2009; 67:76-82. doi: 10.1111/j.1365-2125.2008.03332.x [Crossref] [ Google Scholar]
- Soliman EZ, Lundgren JD, Roediger MP, Duprez DA, Temesgen Z, Bickel M. Boosted protease inhibitors and the electrocardiographic measures of QT and PR durations. AIDS 2011; 25:367-77. doi: 10.1097/QAD.0b013e328341dcc0 [Crossref] [ Google Scholar]
- Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med 2020; 382:1787-99. doi: 10.1056/NEJMoa2001282 [Crossref] [ Google Scholar]
- Han SN, Sun XY, Zhang Z, Zhang LR. The protease inhibitor atazanavir blocks hERG K(+) channels expressed in HEK293 cells and obstructs hERG protein transport to cell membrane. Acta Pharmacol Sin 2015; 36:454-62. doi: 10.1038/aps.2014.165 [Crossref] [ Google Scholar]
- Deng W, Baki L, Yin J, Zhou H, Baumgarten CM. HIV protease inhibitors elicit volume-sensitive Cl- current in cardiac myocytes via mitochondrial ROS. J Mol Cell Cardiol 2010; 49:746-52. doi: 10.1016/j.yjmcc.2010.08.013 [Crossref] [ Google Scholar]
- Song Y, Shryock JC, Wagner S, Maier LS, Belardinelli L. Blocking late sodium current reduces hydrogen peroxide-induced arrhythmogenic activity and contractile dysfunction. J Pharmacol Exp Ther 2006; 318:214-22. doi: 10.1124/jpet.106.101832 [Crossref] [ Google Scholar]
- Zima AV, Blatter LA. Redox regulation of cardiac calcium channels and transporters. Cardiovasc Res 2006; 71:310-21. doi: 10.1016/j.cardiores.2006.02.019 [Crossref] [ Google Scholar]
- Gordon CJ, Tchesnokov EP, Woolner E, Perry JK, Feng JY, Porter DP. Remdesivir is a direct-acting antiviral that inhibits RNA-dependent RNA polymerase from severe acute respiratory syndrome coronavirus 2 with high potency. J Biol Chem 2020; 295:6785-97. doi: 10.1074/jbc.RA120.013679 [Crossref] [ Google Scholar]
- Malin JJ, Suárez I, Priesner V, Fätkenheuer G, Rybniker J. Remdesivir against COVID-19 and other viral diseases. Clin Microbiol Rev 2020; 34. doi: 10.1128/cmr.00162-20 [Crossref]
- Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC. Remdesivir for the treatment of Covid-19 - final report. N Engl J Med 2020; 383:1813-26. doi: 10.1056/NEJMoa2007764 [Crossref] [ Google Scholar]
- Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA 2020; 323:1824-36. doi: 10.1001/jama.2020.6019 [Crossref] [ Google Scholar]
- Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res 2020; 30:269-71. doi: 10.1038/s41422-020-0282-0 [Crossref] [ Google Scholar]
- Kumagai Y, Murakawa Y, Hasunuma T, Aso M, Yuji W, Sakurai T. Lack of effect of favipiravir, a novel antiviral agent, on QT interval in healthy Japanese adults. Int J Clin Pharmacol Ther 2015; 53:866-74. doi: 10.5414/cp202388 [Crossref] [ Google Scholar]
- Chinello P, Petrosillo N, Pittalis S, Biava G, Ippolito G, Nicastri E. QTc interval prolongation during favipiravir therapy in an Ebolavirus-infected patient. PLoS Negl Trop Dis 2017; 11:e0006034. doi: 10.1371/journal.pntd.0006034 [Crossref] [ Google Scholar]
- Gupta AK, Parker BM, Priyadarshi V, Parker J. Cardiac adverse events with remdesivir in COVID-19 infection. Cureus 2020; 12:e11132. doi: 10.7759/cureus.11132 [Crossref] [ Google Scholar]
- Cortegiani A, Ingoglia G, Ippolito M, Giarratano A, Einav S. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care 2020; 57:279-83. doi: 10.1016/j.jcrc.2020.03.005 [Crossref] [ Google Scholar]
-
Coronavirus (COVID-19) update: FDA revokes emergency use authorization for chloroquine and hydroxychloroquine. U.S. Food & Drug Administration; 2020 June 15 [cited 2020 July 15]; Available from: https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-revokes-emergency-use-authorization-chloroquine-and.
-
Chloroquine. DrugBank; 2005 [updated January 7, 2021; cited 2020 November 18]; Available from: https://go.drugbank.com/drugs/DB00608.
- Rodríguez-Menchaca AA, Navarro-Polanco RA, Ferrer-Villada T, Rupp J, Sachse FB, Tristani-Firouzi M. The molecular basis of chloroquine block of the inward rectifier Kir21 channel. Proc Natl Acad Sci U S A 2008; 105:1364-8. doi: 10.1073/pnas.0708153105 [Crossref] [ Google Scholar]
- Cubeddu LX. QT prolongation and fatal arrhythmias: a review of clinical implications and effects of drugs. Am J Ther 2003; 10:452-7. doi: 10.1097/00045391-200311000-00013 [Crossref] [ Google Scholar]
- Luo C, Wang K, Zhang H. Modelling the effects of chloroquine on KCNJ2-linked short QT syndrome. Oncotarget 2017; 8:106511-26. doi: 10.18632/oncotarget.22490 [Crossref] [ Google Scholar]
- Owens RC, Jr Jr. Risk assessment for antimicrobial agent-induced QTc interval prolongation and torsades de pointes. Pharmacotherapy 2001; 21:301-19. doi: 10.1592/phco.21.3.301.34206 [Crossref] [ Google Scholar]
- Albert RK, Schuller JL. Macrolide antibiotics and the risk of cardiac arrhythmias. Am J Respir Crit Care Med 2014; 189:1173-80. doi: 10.1164/rccm.201402-0385CI [Crossref] [ Google Scholar]
- Milberg P, Eckardt L, Bruns HJ, Biertz J, Ramtin S, Reinsch N. Divergent proarrhythmic potential of macrolide antibiotics despite similar QT prolongation: fast phase 3 repolarization prevents early afterdepolarizations and torsade de pointes. J Pharmacol Exp Ther 2002; 303:218-25. doi: 10.1124/jpet.102.037911 [Crossref] [ Google Scholar]
- Raschi E, Poluzzi E, Koci A, Moretti U, Sturkenboom M, Ponti FD. Macrolides and torsadogenic risk: emerging issues from the FDA pharmacovigilance database. J Pharmacovigil 2013; 1:1000104. doi: 10.4172/2329-6887.1000104 [Crossref] [ Google Scholar]
- Chou HW, Wang JL, Chang CH, Lai CL, Lai MS, Chan KA. Risks of cardiac arrhythmia and mortality among patients using new-generation macrolides, fluoroquinolones, and β-lactam/β-lactamase inhibitors: a Taiwanese nationwide study. Clin Infect Dis 2015; 60:566-77. doi: 10.1093/cid/ciu914 [Crossref] [ Google Scholar]
- Ray WA, Murray KT, Hall K, Arbogast PG, Stein CM. Azithromycin and the risk of cardiovascular death. N Engl J Med 2012; 366:1881-90. doi: 10.1056/NEJMoa1003833 [Crossref] [ Google Scholar]
- Hancox JC, Hasnain M, Vieweg WV, Crouse EL, Baranchuk A. Azithromycin, cardiovascular risks, QTc interval prolongation, torsade de pointes, and regulatory issues: a narrative review based on the study of case reports. Ther Adv Infect Dis 2013; 1:155-65. doi: 10.1177/2049936113501816 [Crossref] [ Google Scholar]
- Svanström H, Pasternak B, Hviid A. Use of azithromycin and death from cardiovascular causes. N Engl J Med 2013; 368:1704-12. doi: 10.1056/NEJMoa1300799 [Crossref] [ Google Scholar]
- Chico RM, Chandramohan D. Azithromycin plus chloroquine: combination therapy for protection against malaria and sexually transmitted infections in pregnancy. Expert Opin Drug Metab Toxicol 2011; 7:1153-67. doi: 10.1517/17425255.2011.598506 [Crossref] [ Google Scholar]
- Nguyen LS, Dolladille C, Drici MD, Fenioux C, Alexandre J, Mira JP. Cardiovascular toxicities associated with hydroxychloroquine and azithromycin: an analysis of the World Health Organization pharmacovigilance database. Circulation 2020; 142:303-5. doi: 10.1161/circulationaha.120.048238 [Crossref] [ Google Scholar]
- Jain S, Workman V, Ganeshan R, Obasare ER, Burr A, DeBiasi RM. Enhanced electrocardiographic monitoring of patients with coronavirus disease 2019. Heart Rhythm 2020; 17:1417-22. doi: 10.1016/j.hrthm.2020.04.047 [Crossref] [ Google Scholar]
- Chorin E, Dai M, Shulman E, Wadhwani L, Bar-Cohen R, Barbhaiya C. The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nat Med 2020; 26:808-9. doi: 10.1038/s41591-020-0888-2 [Crossref] [ Google Scholar]
- Crotti L, Arbelo E. COVID-19 treatments, QT interval, and arrhythmic risk: The need for an international registry on arrhythmias. Heart Rhythm 2020; 17:1423-4. doi: 10.1016/j.hrthm.2020.05.024 [Crossref] [ Google Scholar]
- Xu X, Han M, Li T, Sun W, Wang D, Fu B. Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci U S A 2020; 117:10970-5. doi: 10.1073/pnas.2005615117 [Crossref] [ Google Scholar]
- Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395:1054-62. doi: 10.1016/s0140-6736(20)30566-3 [Crossref] [ Google Scholar]
- Canna SW, Behrens EM. Making sense of the cytokine storm: a conceptual framework for understanding, diagnosing, and treating hemophagocytic syndromes. Pediatr Clin North Am 2012; 59:329-44. doi: 10.1016/j.pcl.2012.03.002 [Crossref] [ Google Scholar]
- Gorelik M, Lee Y, Abe M, Andrews T, Davis L, Patterson J. IL-1 receptor antagonist, anakinra, prevents myocardial dysfunction in a mouse model of Kawasaki disease vasculitis and myocarditis. Clin Exp Immunol 2019; 198:101-10. doi: 10.1111/cei.13314 [Crossref] [ Google Scholar]
- Noval Rivas M, Arditi M. Kawasaki disease: pathophysiology and insights from mouse models. Nat Rev Rheumatol 2020; 16:391-405. doi: 10.1038/s41584-020-0426-0 [Crossref] [ Google Scholar]
- Chauhan K, Ackerman MJ, Crowson CS, Matteson EL, Gabriel SE. Population-based study of QT interval prolongation in patients with rheumatoid arthritis. Clin Exp Rheumatol 2015; 33:84-9. [ Google Scholar]
- Panoulas VF, Toms TE, Douglas KM, Sandoo A, Metsios GS, Stavropoulos-Kalinoglou A. Prolonged QTc interval predicts all-cause mortality in patients with rheumatoid arthritis: an association driven by high inflammatory burden. Rheumatology (Oxford) 2014; 53:131-7. doi: 10.1093/rheumatology/ket338 [Crossref] [ Google Scholar]
- Adlan AM, Panoulas VF, Smith JP, Fisher JP, Kitas GD. Association between corrected QT interval and inflammatory cytokines in rheumatoid arthritis. J Rheumatol 2015; 42:421-8. doi: 10.3899/jrheum.140861 [Crossref] [ Google Scholar]
- Lazzerini PE, Capecchi PL, Laghi-Pasini F. Assessing QT interval in patients with autoimmune chronic inflammatory diseases: perils and pitfalls. Lupus Sci Med 2016; 3:e000189. doi: 10.1136/lupus-2016-000189 [Crossref] [ Google Scholar]
- Sham S, Madheshwaran M, Tamilselvam TN, Rajeswari S. Correlation of QT interval with disease activity in newly detected SLE patients at baseline and during flare. Indian J Rheumatol 2015; 10:121-4. doi: 10.1016/j.injr.2015.03.010 [Crossref] [ Google Scholar]
- Aromolaran AS, Srivastava U, Alí A, Chahine M, Lazaro D, El-Sherif N. Interleukin-6 inhibition of hERG underlies risk for acquired long QT in cardiac and systemic inflammation. PLoS One 2018; 13:e0208321. doi: 10.1371/journal.pone.0208321 [Crossref] [ Google Scholar]
- Lazzerini PE, Capecchi PL, El-Sherif N, Laghi-Pasini F, Boutjdir M. Emerging arrhythmic risk of autoimmune and inflammatory cardiac channelopathies. J Am Heart Assoc 2018; 7:e010595. doi: 10.1161/jaha.118.010595 [Crossref] [ Google Scholar]
- Wang J, Wang H, Zhang Y, Gao H, Nattel S, Wang Z. Impairment of HERG K(+) channel function by tumor necrosis factor-alpha: role of reactive oxygen species as a mediator. J Biol Chem 2004; 279:13289-92. doi: 10.1074/jbc.C400025200 [Crossref] [ Google Scholar]
- Petkova-Kirova PS, Gursoy E, Mehdi H, McTiernan CF, London B, Salama G. Electrical remodeling of cardiac myocytes from mice with heart failure due to the overexpression of tumor necrosis factor-alpha. Am J Physiol Heart Circ Physiol 2006; 290:H2098-107. doi: 10.1152/ajpheart.00097.2005 [Crossref] [ Google Scholar]
- Fernández-Velasco M, Ruiz-Hurtado G, Hurtado O, Moro MA, Delgado C. TNF-alpha downregulates transient outward potassium current in rat ventricular myocytes through iNOS overexpression and oxidant species generation. Am J Physiol Heart Circ Physiol 2007; 293:H238-45. doi: 10.1152/ajpheart.01122.2006 [Crossref] [ Google Scholar]
- Kawada H, Niwano S, Niwano H, Yumoto Y, Wakisaka Y, Yuge M. Tumor necrosis factor-alpha downregulates the voltage gated outward K+ current in cultured neonatal rat cardiomyocytes: a possible cause of electrical remodeling in diseased hearts. Circ J 2006; 70:605-9. doi: 10.1253/circj.70.605 [Crossref] [ Google Scholar]
- Panama BK, Latour-Villamil D, Farman GP, Zhao D, Bolz SS, Kirshenbaum LA. Nuclear factor kappaB downregulates the transient outward potassium current I(to,f) through control of KChIP2 expression. Circ Res 2011; 108:537-43. doi: 10.1161/circresaha.110.229112 [Crossref] [ Google Scholar]
- Lazzerini PE, Capecchi PL, Acampa M, Morozzi G, Bellisai F, Bacarelli MR. Anti-Ro/SSA-associated corrected QT interval prolongation in adults: the role of antibody level and specificity. Arthritis Care Res (Hoboken) 2011; 63:1463-70. doi: 10.1002/acr.20540 [Crossref] [ Google Scholar]
- Tufan AN, Sag S, Oksuz MF, Ermurat S, Coskun BN, Gullulu M. Prolonged Tpeak-Tend interval in anti-Ro52 antibody-positive connective tissue diseases. Rheumatol Int 2017; 37:67-73. doi: 10.1007/s00296-016-3488-1 [Crossref] [ Google Scholar]
- Lazzerini PE, Yue Y, Srivastava U, Fabris F, Capecchi PL, Bertolozzi I. Arrhythmogenicity of anti-Ro/SSA antibodies in patients with torsades de pointes. Circ Arrhythm Electrophysiol 2016; 9:e003419. doi: 10.1161/circep.115.003419 [Crossref] [ Google Scholar]
- Coleman CM, Sisk JM, Mingo RM, Nelson EA, White JM, Frieman MB. Abelson kinase inhibitors are potent inhibitors of severe acute respiratory syndrome coronavirus and Middle East respiratory syndrome coronavirus fusion. J Virol 2016; 90:8924-33. doi: 10.1128/jvi.01429-16 [Crossref] [ Google Scholar]
- Hartmann JT, Haap M, Kopp HG, Lipp HP. Tyrosine kinase inhibitors - a review on pharmacology, metabolism and side effects. Curr Drug Metab 2009; 10:470-81. doi: 10.2174/138920009788897975 [Crossref] [ Google Scholar]
- Ghatalia P, Je Y, Kaymakcalan MD, Sonpavde G, Choueiri TK. QTc interval prolongation with vascular endothelial growth factor receptor tyrosine kinase inhibitors. Br J Cancer 2015; 112:296-305. doi: 10.1038/bjc.2014.564 [Crossref] [ Google Scholar]
- Abu Rmilah AA, Lin G, Begna KH, Friedman PA, Herrmann J. Risk of QTc prolongation among cancer patients treated with tyrosine kinase inhibitors. Int J Cancer 2020; 147:3160-7. doi: 10.1002/ijc.33119 [Crossref] [ Google Scholar]
- Kloth JS, Pagani A, Verboom MC, Malovini A, Napolitano C, Kruit WH. Incidence and relevance of QTc-interval prolongation caused by tyrosine kinase inhibitors. Br J Cancer 2015; 112:1011-6. doi: 10.1038/bjc.2015.82 [Crossref] [ Google Scholar]
- Lee HA, Hyun SA, Byun B, Chae JH, Kim KS. Electrophysiological mechanisms of vandetanib-induced cardiotoxicity: comparison of action potentials in rabbit Purkinje fibers and pluripotent stem cell-derived cardiomyocytes. PLoS One 2018; 13:e0195577. doi: 10.1371/journal.pone.0195577 [Crossref] [ Google Scholar]
- Cohen JD, Babiarz JE, Abrams RM, Guo L, Kameoka S, Chiao E. Use of human stem cell derived cardiomyocytes to examine sunitinib mediated cardiotoxicity and electrophysiological alterations. Toxicol Appl Pharmacol 2011; 257:74-83. doi: 10.1016/j.taap.2011.08.020 [Crossref] [ Google Scholar]
- Dong Q, Fu XX, Du LL, Zhao N, Xia CK, Yu KW. Blocking of the human ether-à-go-go-related gene channel by imatinib mesylate. Biol Pharm Bull 2013; 36:268-75. doi: 10.1248/bpb.b12-00778 [Crossref] [ Google Scholar]
- Küppers R. Mechanisms of B-cell lymphoma pathogenesis. Nat Rev Cancer 2005; 5:251-62. doi: 10.1038/nrc1589 [Crossref] [ Google Scholar]
- Nakken B, Munthe LA, Konttinen YT, Sandberg AK, Szekanecz Z, Alex P. B-cells and their targeting in rheumatoid arthritis--current concepts and future perspectives. Autoimmun Rev 2011; 11:28-34. doi: 10.1016/j.autrev.2011.06.010 [Crossref] [ Google Scholar]
- Pan Z, Scheerens H, Li SJ, Schultz BE, Sprengeler PA, Burrill LC. Discovery of selective irreversible inhibitors for Bruton's tyrosine kinase. ChemMedChem 2007; 2:58-61. doi: 10.1002/cmdc.200600221 [Crossref] [ Google Scholar]
- Guha A, Derbala MH, Zhao Q, Wiczer TE, Woyach JA, Byrd JC. Ventricular arrhythmias following ibrutinib initiation for lymphoid malignancies. J Am Coll Cardiol 2018; 72:697-8. doi: 10.1016/j.jacc.2018.06.002 [Crossref] [ Google Scholar]
- Lampson BL, Yu L, Glynn RJ, Barrientos JC, Jacobsen ED, Banerji V. Ventricular arrhythmias and sudden death in patients taking ibrutinib. Blood 2017; 129:2581-4. doi: 10.1182/blood-2016-10-742437 [Crossref] [ Google Scholar]
- Salem JE, Manouchehri A, Bretagne M, Lebrun-Vignes B, Groarke JD, Johnson DB. Cardiovascular toxicities associated with ibrutinib. J Am Coll Cardiol 2019; 74:1667-78. doi: 10.1016/j.jacc.2019.07.056 [Crossref] [ Google Scholar]
- Leong DP, Caron F, Hillis C, Duan A, Healey JS, Fraser G. The risk of atrial fibrillation with ibrutinib use: a systematic review and meta-analysis. Blood 2016; 128:138-40. doi: 10.1182/blood-2016-05-712828 [Crossref] [ Google Scholar]
- Jain P, Thompson PA, Keating M, Estrov Z, Ferrajoli A, Jain N. Long-term outcomes for patients with chronic lymphocytic leukemia who discontinue ibrutinib. Cancer 2017; 123:2268-73. doi: 10.1002/cncr.30596 [Crossref] [ Google Scholar]
- Tuomi JM, Xenocostas A, Jones DL. Increased susceptibility for atrial and ventricular cardiac arrhythmias in mice treated with a single high dose of ibrutinib. Can J Cardiol 2018; 34:337-41. doi: 10.1016/j.cjca.2017.12.001 [Crossref] [ Google Scholar]
- de Jong J, Hellemans P, Jiao JJ, Huang Y, Mesens S, Sukbuntherng J. Ibrutinib does not prolong the corrected QT interval in healthy subjects: results from a thorough QT study. Cancer Chemother Pharmacol 2017; 80:1227-37. doi: 10.1007/s00280-017-3471-x [Crossref] [ Google Scholar]
- Bechman K, Subesinghe S, Norton S, Atzeni F, Galli M, Cope AP. A systematic review and meta-analysis of infection risk with small molecule JAK inhibitors in rheumatoid arthritis. Rheumatology (Oxford) 2019; 58:1755-66. doi: 10.1093/rheumatology/kez087 [Crossref] [ Google Scholar]
- Conner SD, Schmid SL. Identification of an adaptor-associated kinase, AAK1, as a regulator of clathrin-mediated endocytosis. J Cell Biol 2002; 156:921-9. doi: 10.1083/jcb.200108123 [Crossref] [ Google Scholar]
- Cantini F, Niccoli L, Matarrese D, Nicastri E, Stobbione P, Goletti D. Baricitinib therapy in COVID-19: a pilot study on safety and clinical impact. J Infect 2020; 81:318-56. doi: 10.1016/j.jinf.2020.04.017 [Crossref] [ Google Scholar]
- Kalil AC, Patterson TF, Mehta AK, Tomashek KM, Wolfe CR, Ghazaryan V. Baricitinib plus remdesivir for hospitalized adults with Covid-19. N Engl J Med 2020. doi: 10.1056/NEJMoa2031994 [Crossref]
-
FDA briefing document Arthritis Advisory Committee meeting April 23, 2018 NDA 207924 baricitinib Janus kinase (JAK) inhibitor for RA Eli Lilly and Company (Lilly). U.S. Food & Drug Administration; 2018 [cited 2020 December 27]; Available from: https://www.fda.gov/media/112372/download.
-
Coronavirus (COVID-19) update: FDA authorizes monoclonal antibodies for treatment of COVID-19. U.S. Food & Drug Administration; 2020 November 21 [cited 2020 November 22]; Available from: https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-monoclonal-antibodies-treatment-covid-19.
-
Coronavirus (COVID-19) update: FDA authorizes monoclonal antibody for treatment of COVID-19. U.S. Food & Drug Administration; 2020 November 9 [cited 2020 November 11]; Available from: https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-monoclonal-antibody-treatment-covid-19.