Bioimpacts. 2025;15:30127.
doi: 10.34172/bi.30127
Review
Intraocular drug delivery systems for Diabetic retinopathy: Current and future prospective
Sayed Latif Taheri Data curation, Investigation, Writing – original draft, 
Safoora Poorirani Data curation, Investigation, Writing – original draft,
Sayed Abolfazl Mostafavi Conceptualization, Project administration, Resources, Supervision, Visualization, Writing – review & editing, , * 
Author information:
Department of Pharmaceutics, School of Pharmacy and Pharmaceutical Sciences and Isfahan Pharmaceutical Sciences Research Center, Isfahan University of Medical Sciences, Isfahan, Iran
Abstract
In pharmaceutical research and development, novel drug delivery systems represent a significant advancement aimed at enhancing the efficacy of therapeutic agents through innovative delivery mechanisms. The primary objective of these systems is to transport therapeutic compounds to specific target sites, such as tumors and afflicted tissues, with the dual purpose of mitigating side effects and toxicity associated with the drugs while concurrently augmenting therapeutic effectiveness. Numerous innovative drug delivery strategies have been scrutinized for their applicability in the context of targeted ocular drug delivery. Diverse novel carriers, including but not limited to implants, hydrogels, metal nanoparticles, Nano-liposomes, micelles, solid lipid nanoparticles (SLN), emulsions, and biodegradable nanoparticles, have been harnessed to facilitate the controlled release of pharmaceutical agents to the retina and vitreous. These carriers offer distinct advantages, such as enhanced intraocular drug delivery, precise control over drug release kinetics, heightened stability, and superior entrapment efficiency. This comprehensive review seeks to elucidate the current strides made in the realm of carriers and their contemporary applications in treating diabetic retinopathy (DR). Furthermore, it underscores these carriers' pivotal role in achieving efficacious intraocular drug delivery. Additionally, this article explores the various administration routes, potential future advancements, and the multifaceted challenges confronting the domain of novel carriers in treating DR. In conclusion, novel formulations are introduced to surmount the challenges associated with intraocular drug delivery.
Keywords: Retinopathy, Intraocular, Nanocarriers, Hydrogels, Implants
Copyright and License Information
© 2025 The Author(s).
This work is published by BioImpacts as an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (
http://creativecommons.org/licenses/by-nc/4.0/). Non-commercial uses of the work are permitted, provided the original work is properly cited.
Funding Statement
This research received grant from Isfahan University of Medical Sciences for supporting this work, Grant Number 140124.
Introduction
Novel drug delivery systems represent a contemporary scientific frontier that has catalyzed advancements across various scientific domains, particularly in pharmaceuticals. These systems have effectively surmounted challenges associated with drug delivery, sparking considerable interest among pharmaceutical researchers seeking innovative technologies for precision drug targeting.1-3 The fundamental objective of this paradigm is to transport therapeutic agents in quantities sufficient to target specific sites, such as tumors and afflicted tissues, while simultaneously mitigating undesirable side effects and toxicity, thus enhancing therapeutic efficacy.4-6
Diabetes mellitus, characterized by inadequate insulin production or responsiveness leading to elevated blood glucose levels, is one of the most significant health epidemics of the 21st century.7 Diabetes can be broadly categorized into type 1 (insulin-dependent) and type 2 (insulin-independent). Diabetic retinopathy (DR) emerges as a prevalent microvascular complication, affecting virtually all type 1 diabetes patients and over 60% of type 2 diabetes individuals during the first two decades of the disease.8 Clinically, DR manifests in two primary forms: non-proliferative diabetic retinopathy (NPDR) and proliferative diabetic retinopathy (PDR). NPDR can be further stratified into mild, moderate, and severe stages, characterized by microaneurysms, hemorrhages, hard exudates (lipid deposits), cotton wool spots, intraretinal microvascular abnormalities, venous beading, and loop formation (Fig. 1).
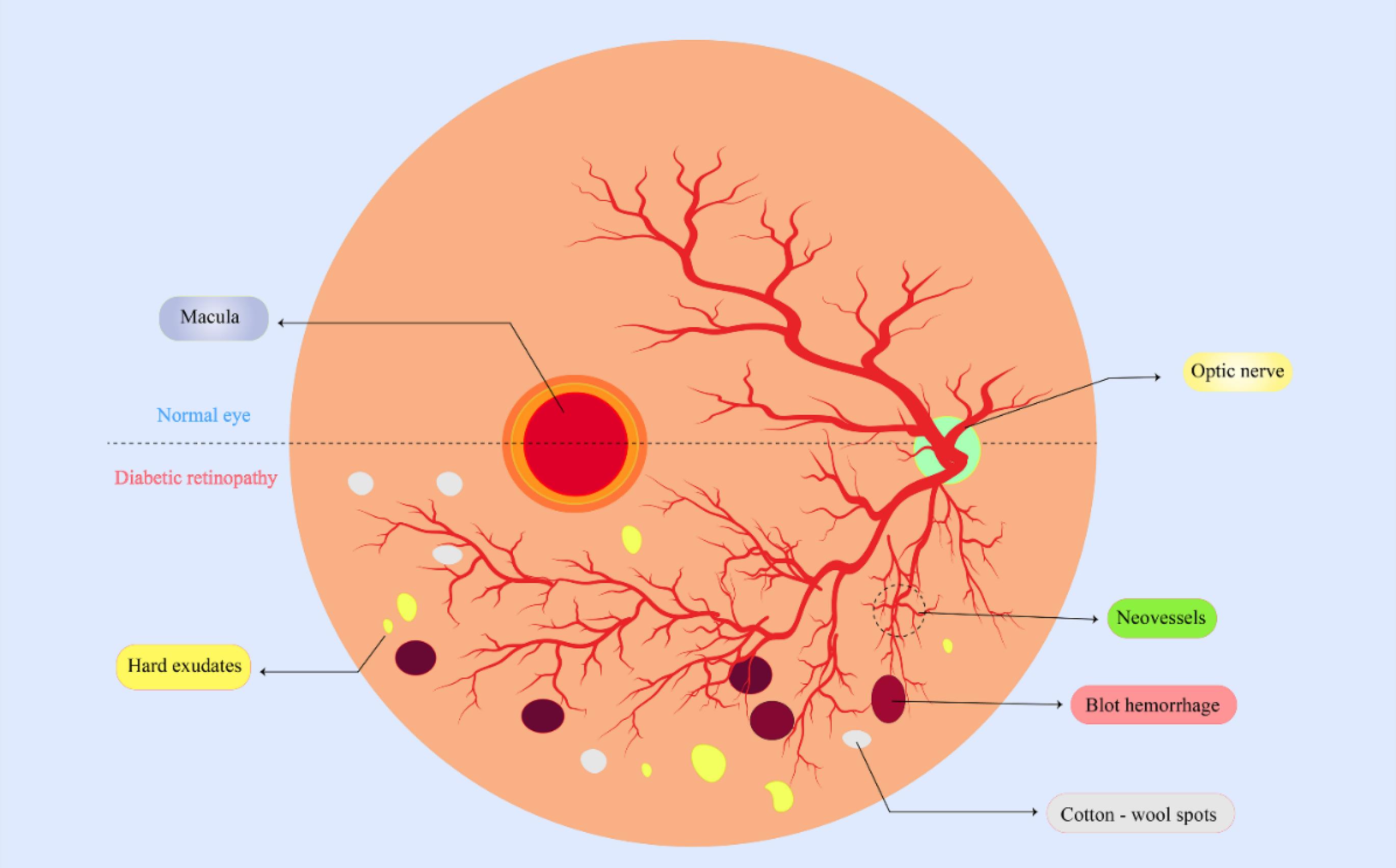
Fig. 1.
Schematic of diabetic retinopathy.
.
Schematic of diabetic retinopathy.
NPDR can progress to PDR, marked by retinal neovascularization and vitreous hemorrhage.8 This condition disrupts the blood-retinal barrier (BRB) and increases vascular permeability, leading to leakage and diabetic macular edema (DME).9 Elevated vascular permeability exacerbates capillary occlusion, causing retinal ischemia and upregulating vascular endothelial growth factor (VEGF) levels.10 Recently, VEGF inhibitors like pegaptanib sodium, ranibizumab, and bevacizumab have demonstrated their effectiveness in suppressing ocular neovascularization, significantly reducing neovascular activity and vascular permeability in various ocular tissues.11 Table 1 provides an overview of different drug types for retinopathy treatment.
Table 1.
A summary of the different types of drugs that are currently available in the market for diabetic retinopathy
|
Drug |
Dosage forms
|
Mechanism
|
Drug delivery system
|
References
|
| Triamcinolone acetonide |
4 mg/0.1 mL (injection suspension) |
Anti-inflammatory effects |
intravitreal |
12
|
| Fluocinolone acetonide |
0.59 mg (implant) |
Anti-inflammatory effects |
intravitreal implant |
13
|
| Dexamethasone |
0.7 mg (implant) |
Anti-inflammatory effects |
Extended-release implant |
14
|
| Etanercept |
2.5 mg/0.1 mL (Injection Suspension) |
Anti-inflammatory effects |
intravitreal |
15
|
| Infliximab |
5 mg/kg (injection suspension) |
Anti-inflammatory effects |
Intravenous injection |
15
|
| Pegaptanib |
0.3 mg/0.09 mL (Injection Suspension) |
VEGF inhibitors |
intravitreal |
16
|
| Bevacizumab |
1.25 mg/0.05 mL (Injection Suspension) |
VEGF inhibitors |
intravitreal |
17
|
| Ranibizumab |
0.5 mg/0.05 mL (Injection Suspension) |
VEGF inhibitors |
intravitreal |
18
|
| Ruboxistaurin |
32 mg/day |
inhibitor |
oral |
19,20
|
| Hyaluronidase |
75 IU/0.05 mL saline |
Clearance of vitreous hemorrhage |
intravitreal |
20
|
VEGF, vascular endothelial growth factor; PKC β, Protein kinase C β.
Bevacizumab, also known as Avastin, stands out as the preferred anti-VEGF antibody fragment for PDR treatment. Although generally well-tolerated, bevacizumab is associated with common side effects such as hypertension, proteinuria, impaired wound healing, and thrombosis, raising concerns.21 Consequently, intraocular injection is the prevailing method of bevacizumab administration. However, a significant drawback of intravitreal anti-VEGF treatment lies in the need for repetitive injections due to the short half-life of these drugs within the vitreous. This approach incurs challenges and costs, along with potential adverse effects, including intravitreal hemorrhage, endophthalmitis, cataract formation, and retinal detachment.11,21
Conventional drug formulations in treatment confront several hurdles, such as short half-lives, low solubility, efficacy at high doses, aggregation, and susceptibility to degradation.22 A limited repertoire of technologies has been available for addressing intraocular disorders and diseases. While traditional delivery methods, such as subconjunctival injection, intravitreal injection, and topical eye drops, exist, numerous biological and physiological barriers pose formidable challenges that therapeutic payloads must surmount. Clinical studies employing these delivery vehicles have raised concerns regarding safety, including immunogenicity, broad tissue tropism, and genomic insertional mutagenesis.23 Addressing these limitations necessitates the development of innovative intraocular drug delivery systems grounded in biodegradable carriers that offer heightened effectiveness and durability within the intraocular environment. A diverse array of drug delivery systems, including implants, hydrogels, nanoliposomes, micelles, solid lipid nanoparticles (SLNs), emulsions, and biodegradable nanoparticles, have been deployed for precise and controlled drug delivery to the retina and vitreous.24-30 Several review articles covering ocular disorders have been published.22,31,32
Within drug delivery and therapeutics, nanocarriers are specialized vehicles for transporting drugs to specific anatomical targets within the body, including sites affected by DR. These nanocarriers can encapsulate drugs within their structures, serving as protective capsules that shield the drug from degradation, metabolism, or elimination, prolonging circulation time and enhancing stability.33 Many drugs employed in DR treatment exhibit poor solubility or limited bioavailability. Nanoparticles address this issue by enhancing the solubility of hydrophobic drugs, leading to improved drug absorption and distribution. Additionally, nanoparticles offer protection against enzymatic degradation, augmenting drug bioavailability and ensuring a more pronounced therapeutic effect.34 Notable nanocarrier advantages include targeted drug delivery, controlled and sustained drug release, enhanced drug stability and protection, prolonged drug circulation, and improved solubility and bioavailability, thus facilitating enhanced drug absorption and distribution.
However, nanocarriers have certain limitations, including intricate formulation and manufacturing processes, potential immunogenic responses or toxicities associated with nanoparticle carrier materials, challenges in scaling up production for large-scale clinical applications, and variable clearance from the body contingent upon nanoparticle characteristics and size.35
This review represents a comprehensive examination of emerging intraocular drug delivery systems for treating DR, a subject not comprehensively covered in prior reviews.
Tonicity, referring to the osmotic pressure or solute concentration in a solution, emerges as a pivotal factor in drug delivery systems for DR treatment. Maintaining the structural integrity of ocular tissues necessitates a tolerable tonicity range of 0.5% to 2% NaCl solution. Common tonicity modifiers include 1.9% boric acid and sodium acid phosphate buffer.36,37
Sterile drug delivery systems are pivotal in mitigating infection risks and ensuring patient safety when addressing DR. Given their direct contact with sensitive ocular tissues; microbial contamination can have severe consequences. Therefore, production processes must be executed within a sterile environment. Sterilization techniques such as filtration, autoclaving, or gamma irradiation may be employed to eliminate existing microbiological contaminants. Furthermore, ensuring the sterility of drug delivery systems until administration necessitates appropriate storage conditions, including regulated temperature and aseptic packaging.36
The meticulous engineering of drug delivery systems is imperative to ensure biocompatibility with ocular tissues and to maintain their integrity during ocular administration, thereby optimizing therapeutic efficacy while minimizing side effects. Biodegradable implantable technologies, including hydrogels and polymeric microspheres, hold promise as platforms for long-term drug delivery. Ideally, these systems should undergo gradual biodegradation, yielding non-irritating and non-toxic degradation byproducts, thus obviating surgical removal.38
The biodegradation of drug delivery systems significantly impacts their efficacy and safety in treating DR. Long-term drug release, reduced frequency of administration, and avoidance of complications associated with extended implantation are all contingent upon the capacity of a delivery system to biodegrade over time. Biodegradable materials such as polymeric microspheres, nanoparticles, and hydrogels are conventionally employed to encapsulate and release therapeutic agents.39
This comprehensive review delves into recent advancements in intraocular drug delivery systems for DR. Drawing from multiple articles, cutting-edge delivery methods for intraocular applications are introduced. The investigation encompasses nanocarriers and implants, each further segmented into subclasses, providing detailed insights. Additionally, current research developments and challenges linked to the use of carriers are addressed, along with specifics about DR treatments and regeneration incorporating carrier-based approaches.
The human eye's anatomy
The human eye is an intricate organ crucial for vision, composed of several interconnected structures endowed with distinct functions. In Fig. 2, the primary components of the human eye are depicted. The cornea, a transparent and dome-shaped tissue, envelops the eye's anterior, facilitating light entry. The iris, which showcases the eye's coloration, governs the pupil's size, regulating the influx of light. The pupil, a malleable circular aperture at the iris's center, permits light passage. Positioned behind the iris, the lens concentrates light onto the retina, with its capacity to alter shape to accommodate near and far vision. The retina, a slender, light-sensitive layer at the eye's posterior, teems with specialized photoreceptor cells that convert light into electrical signals dispatched to the brain via the optic nerve. The optic nerve, a bundle of nerve fibers, is the conduit for visual information transmission from the retina to the brain. Filling the eye's central cavity, the vitreous humor is a gel-like substance that maintains the ocular structure and supports its internal components; the white outer layer, known as the scale, protects and sustains stain the eye's form.40
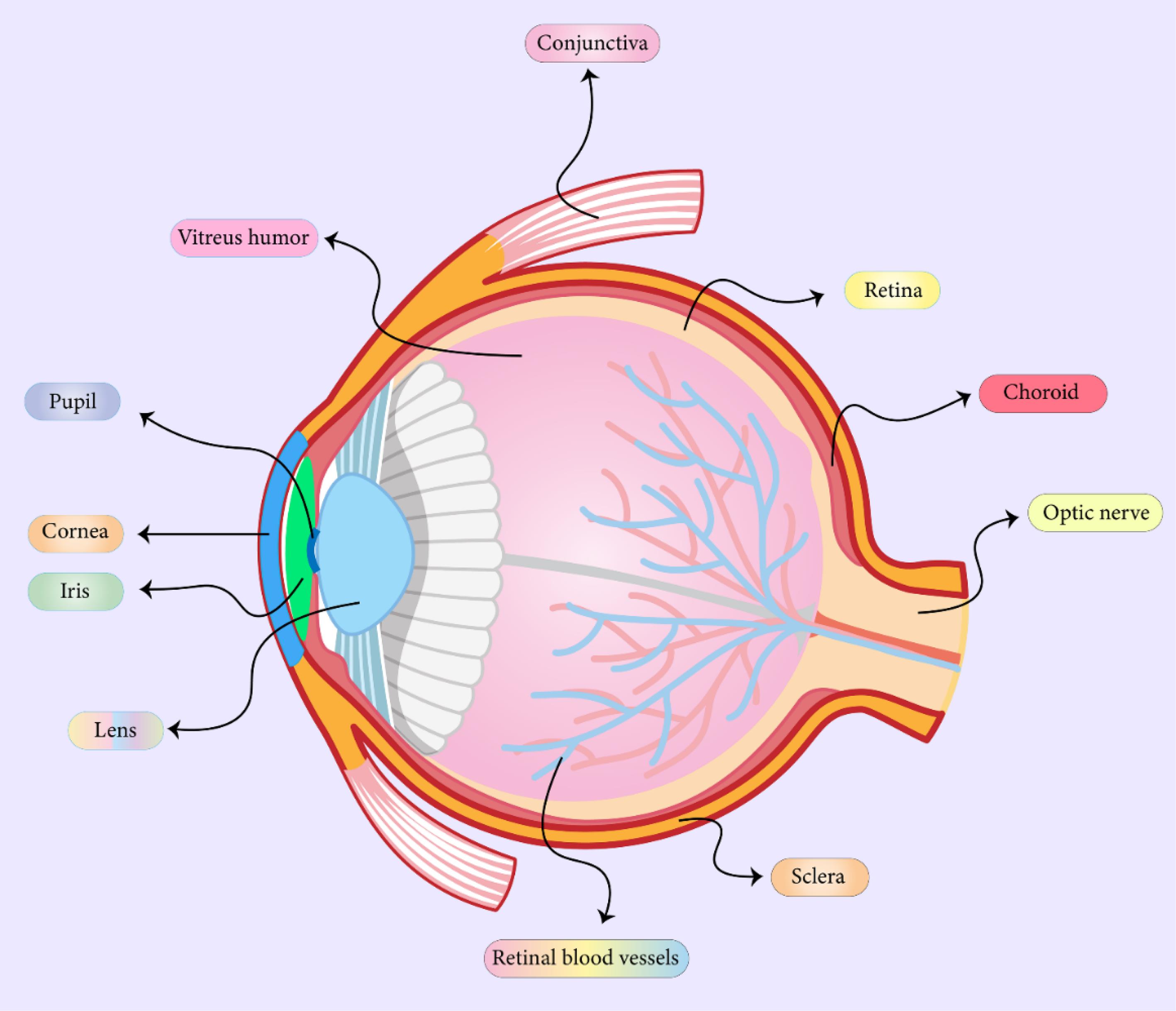
Fig. 2.
Schematic of the human eye.
.
Schematic of the human eye.
Challenges in Retinal Drug Delivery for DR The administration of drugs to the retina for DR treatment poses a formidable challenge due to many barriers, including the BRB, Limited Drug Permeability, Rapid Clearance, Ocular Dynamics, and Patient Compliance.41 The retina's blood vessels are exceptionally selective, forming a formidable barricade called the BRB. This barrier imposes stringent restrictions on the transit of large molecules, including numerous drugs, from the bloodstream into the retina, rendering the delivery of therapeutic agents to the target location challenging. Even if a drug succeeds in traversing the BRB, it may encounter obstacles in permeating the diverse retinal layers to access the intended site of action. The retina's intricate architecture and tight junctions among retinal cells curtail drug permeability. The eye possesses highly efficient mechanisms for expeditiously expelling foreign substances, including drugs. This can curtail the duration and efficacy of drug action prior to its elimination from the eye. The perpetual motion and blinking of the eye pose mechanical hurdles in drug delivery. Tears and blinking can either rinse away or dilute drug formulations, thus diminishing their concentration and efficacy. In cases where drugs necessitate repeated and prolonged administration, patient adherence becomes a formidable obstacle.42 Consistent and timely drug administration is imperative for the successful management of DR, with non-compliance serving as a hindrance to treatment outcomes. Surmounting these obstacles continues to be an area of active exploration within the realm of ocular drug delivery. Scientists are investigating many strategies, encompassing the development of innovative drug delivery systems, nanoparticles, and targeted drug carriers, all aimed at augmenting the efficiency and effectiveness of drug delivery to the retina in the context of DR and other ocular maladies.43
Intraocular delivery for DR
Hydrogels
The utilization of hydrogels in biomedical applications has been an evolutionary journey commencing in the 1960s when hydrogel first found its application as contact lenses.44 Hydrogels represent a distinctive class of polymeric materials renowned for their remarkable capacity to absorb and retain substantial quantities of water within their intricate three-dimensional matrix.45,46 This unique characteristic has paved the way for the effective delivery of biologically active substances through controlled drug release mechanisms.
Significant research efforts have been directed towards developing novel hydrogel structures and chemically cross-linked networks.47-49 Among these, polyhydroxy ethyl methacrylate (PHEMA) has emerged as the most compelling polymer.50
As the 1970s progressed, a newfound fascination with stimuli-responsive hydrogels emerged. These hydrogels exhibit an inherent ability to undergo physical or chemical transitions in response to specific environmental stimuli, such as changes in pH, temperature, light exposure, and pressure.51 Thermosensitive hydrogel is one of the most extensively studied and utilized forms of stimuli-responsive hydrogels.52,53 Thermosensitive hydrogels can be categorized into lower critical solution temperature (LCST) and upper critical solution temperature (UCST) hydrogels. In LCST hydrogels, the system exists as a liquid under critical temperature conditions, whereas in UCST hydrogels, the system assumes a gel state under critical temperature and becomes liquid at temperatures above the critical threshold.54,55
Crucially, to circumvent potential immune reactions, the polymers employed in hydrogel formulations must possess specific essential characteristics, including biodegradability, biocompatibility, and non-cytotoxicity.56 Hydrogels are constructed from cross-linked polymers that can imbibe water, resulting in swelling and the maintenance of an expanded water-rich structure.57,58
Another vital category within the realm of stimuli-responsive hydrogels is the pH-responsive hydrogel. These systems feature ionizable pendant groups within the polymer backbone, allowing them to respond to variations in pH levels. Alterations in environmental pH lead to ionization of the pendant groups, creating electrostatic repulsive forces between ionized groups, thus inducing swelling. pH-responsive hydrogels can be further classified into anionic and cationic hydrogels. Anionic hydrogels incorporate carboxylic or sulfonic acid groups that undergo deprotonation and swell as the pH increases, while cationic hydrogels incorporate amine groups that protonate and swell as the pH decreases.59,60
In the context of temperature-sensitive hydrogels, changes in temperature trigger either swelling or de-swelling within the system, which can be harnessed for drug delivery purposes. The interplay between hydrophobic and hydrophilic regions within the hydrogel structure plays a pivotal role in this physical response.
Shear-thinning hydrogels represent yet another facet of hydrogel engineering. Shear-thinning is a property wherein the material's viscosity decreases with increased shear forces. Such hydrogels can be readily loaded into syringes, extruded when subjected to shear, and swiftly regain their original form when mechanical forces cease, a phenomenon commonly referred to as self-healing. This property is highly advantageous for maintaining material integrity following injection and facilitating in situ gelation processes, with the benefit of minimizing potential embolization into the systemic circulation. However, it is worth noting that physical cross-linking often disrupts self-healing properties and fails to exhibit the mechanical integrity seen in in situ cross-linking covalent systems. Researchers have therefore explored alternative cross-linking methodologies to enhance the stability of mechanically deployed hydrogels after injection.61,62 Shear-thinning hydrogels have been the subject of extensive investigation across various biomedical applications, encompassing drug delivery, tissue regeneration, and intraocular drug delivery.63-65
In summary, developing in situ injectable hydrogels, offering precise control over drug release rates and degradation, holds immense promise as a versatile drug carrier for ocular drug delivery.66 Moreover, preclinical studies have yielded encouraging results in applying hydrogel-based tissue adhesives, vitreous replacements, and intravitreal drug delivery systems.The intraocular administration of hydrogels stands poised to bring about significant advancements in this field, ultimately contributing to the refinement of existing hydrogel technologies and the potential for future clinical approvals. It is imperative to concurrently advance injection systems capable of efficiently and safely handling in situ forming hydrogels in clinical settings.67 Table 2 and Fig. 3 provide a succinct overview of the various types of hydrogels mentioned herein.
Table 2.
Various types of hydrogels used for retinopathy drug delivery
|
Hydrogel type
|
Stimuli-responsive polymer
|
Drug
|
Property
|
Therapeutic outcome
|
Reference
|
| Temperature-sensitive hydrogels |
PLGA-PEG-PLGA |
Bevacizumab |
sol-to-gel transition with change in temperature |
showed the beneficial effects of hydrogels in prolonging the residency of drugs in the vitreous and increasing the drug's efficiency |
68
|
| Poloxamer |
Bevacizumab |
69
|
| ESHU |
Bevacizumab |
70
|
| PLGA-PEG-PLGA |
Dexamethasone acetate |
53
|
| PEG-PCL-PEG |
Bevacizumab |
71
|
| PLGA-PEG-PLGA |
Insulin |
72
|
| Chitosan |
Fluconazole |
|
|
73
|
| Shear sensitive hydrogels |
Tragacanthic acid |
|
Gel-to-sol transition with shear |
-
|
65
|
PLGA-PEG-PLGA; Poly (lactic acid-co-glycolic acid)-poly (ethylene glycol)-poly (lactic acid-co-glycolic acid)), ESHU; poly (ethylene glycol)-poly-(serinol hexamethylene urethane), PEG-PCL-PEG; poly (ethylene glycol)-poly (ɛ-caprolactone)-poly (ethylene glycol)
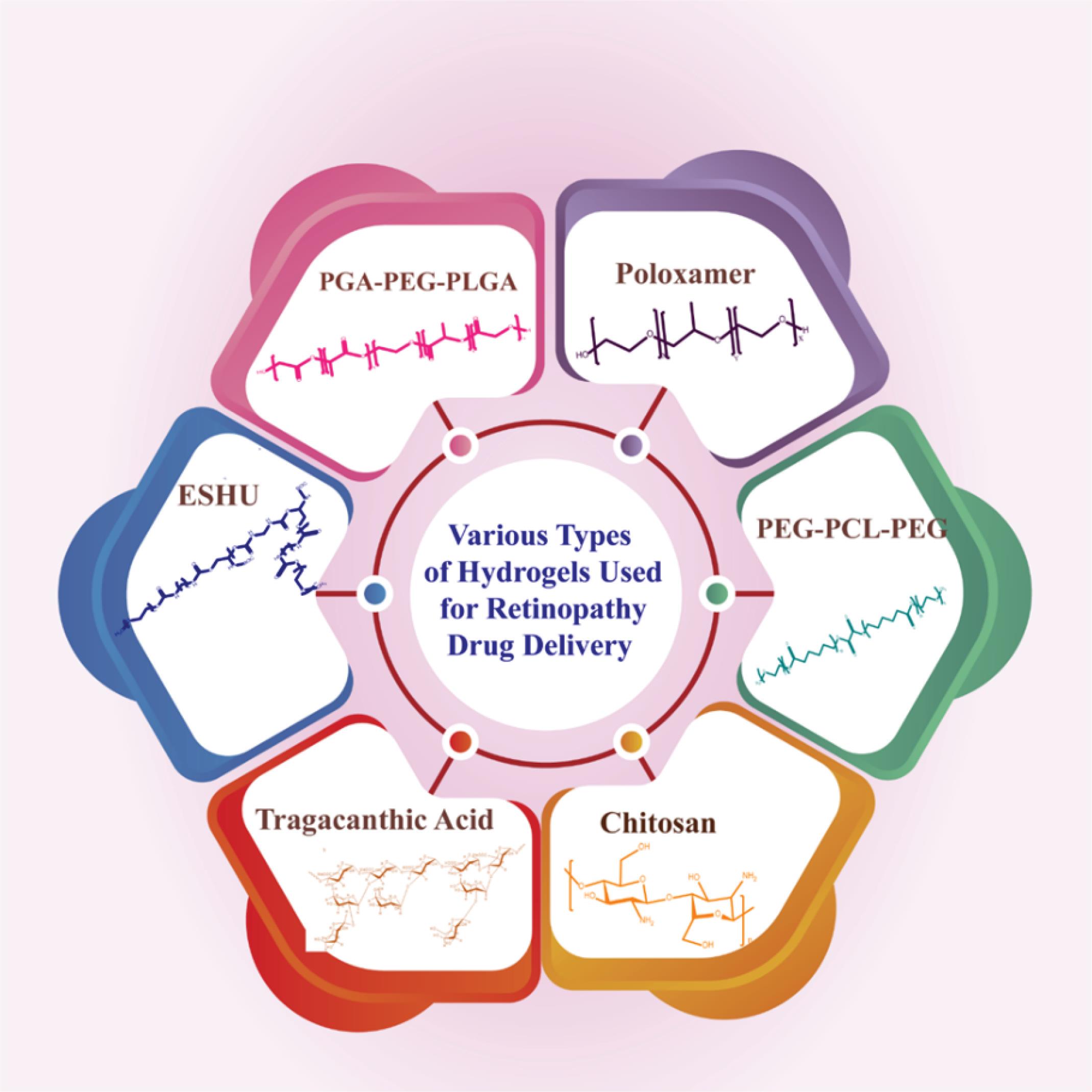
Fig. 3.
Schematic diagram of the hydrogels used for retinopathy drug delivery.
.
Schematic diagram of the hydrogels used for retinopathy drug delivery.
Exemplary instances of hydrogels
Hydrogels with shear sensitivity
In a study by Chegini et al, cross-linked and injectable hydrogels were formulated using tragacanth gum as a base material. This particular hydrogel demonstrated potential applicability for the targeted delivery of therapeutic agents to the posterior segment of the eye in conditions such as DR and macular edema. The formulation involved the utilization of tragacanthic acid (TA), the water-soluble component of tragacanth gum, and three distinct acetate salts.
The evaluations of this study revealed that the hydrogel formulation incorporating TA and sodium acetate (referred to as TA-NaOAc) exhibited biocompatibility, optical transparency, injectability, and adequate structural integrity in its quiescent state post-injection. Importantly, assessments conducted via the MTT assay demonstrated the absence of cytotoxicity toward human umbilical vein endothelial cells (HUVECs). Additionally, in vivo Draize tests and histological examinations of the retinas in rabbits and rats did not reveal any signs of allergic reactions or histopathological alterations.
The amalgamation of these in vitro and in vivo findings augurs well for the potential application of TA-NaOAc hydrogel in ocular drug delivery, holding significant promise in this domain.65
Temperature-sensitive hydrogels
In the sphere of ocular drug delivery, a diverse array of thermosensitive hydrogels has been developed, encompassing both natural polymers such as chitosan, alginate, and hyaluronic acid, as well as synthetic polymers like poloxamer, PEG/PLGA block polymers, PEG/PCL block polymers, and poly(N-isopropyl acrylamide).67,74-77
Among these, poloxamers, recognized as triblock copolymers poly (ethylene glycol-b-propylene glycol-b-ethylene glycol) (PEG-PPG-PEG), have undergone extensive scrutiny due to their unique inverse thermosensitive properties. A significant transformation from a liquid to a gel state in aqueous poloxamer solutions materializes at the physiological temperature of 37 °C. This attribute renders them particularly attractive for applications as injectable agents in controlled-release drug delivery, tissue engineering, and cell therapy.78
Recent developments have also witnessed a growing interest in PEG-based hydrogels, augmented by including hydrophobic components such as PLGA and PCL. Consequently, PEG-based thermosensitive hydrogels have emerged as compelling candidates for exploration as injectable thermosensitive materials. Notably, PEG can be copolymerized alongside PLGA, PLA, and PCL, resulting in copolymers characterized by A-B-A or B-A-B structures.25,79 In parenteral drug delivery, attributes such as biodegradability and biocompatibility assume paramount significance. Notably, PLGA has witnessed substantial investigation in combination with PEG to create block thermoreversible gelling polymers.80,81 One remarkable study employed temperature-sensitive injectable hydrogels, specifically PLGA-PEG-PLGA, to facilitate the sustained release of Avastin into the ocular environment. Impressively, the in vitro drug release kinetics extended over two weeks, diverging significantly from the free drug release profile. Additionally, no toxicity or inflammation was discerned within the hydrogel system.82
Nanocarriers
Nanocarriers represent a pivotal innovation in drug delivery, offering versatile solutions that can be crafted from a variety of inorganic and organic materials, including biodegradable and non-degradable polymers, metals, lipids, and self-assembling amphiphilic molecules.83-85 This remarkable development addresses a longstanding challenge in pharmacotherapy, where bioactive substances, while exhibiting therapeutic benefits, often manifest undesirable side effects that restrict their clinical applicability.
One prominent example is chemotherapy, employed in cancer treatment, where drugs indiscriminately target both cancerous and healthy cells, resulting in adverse effects. The scientific community has sought ways to selectively deliver bioactives to specific anatomical sites within the body to optimize therapeutic potential while minimizing these detrimental outcomes. This endeavor has spurred extensive research into nanocarriers for precise drug and gene delivery, enhancing therapeutic efficacy while mitigating side effects. These nanoparticulate delivery systems offer several crucial advantages, including elevated target-to-non-target concentration ratios, prolonged drug residency at the intended site, and enhanced cellular uptake and intracellular stability.86
Nanocarriers have recently gained significant attention as a preferred drug delivery system due to their remarkable ability to reduce toxicity and enhance therapeutic effectiveness. These systems are characterized by submicron particle sizes, typically less than 500 nm, which results in a high surface area-to-volume ratio. This unique attribute profoundly influences the properties and bioactivity of encapsulated drugs. Critical attributes of nanocarriers include:
-
Prolonged circulation time in the bloodstream.
-
Precise delivery of drugs to the intended target sites.
-
Reduction in the required drug dosage.
-
Controlled drug release.
Various types of nanocarriers have been developed, such as microemulsions, nanosuspensions, liposomes, micelles, solid lipid nanoparticles (SLN), dendrimers, and hydrogels.87-89 The properties of nanocarriers can be tailored through modifications in their composition, shape, size, and surface characteristics. These modifications encompass PEGylation, functional group introduction, surface charge adjustment, and targeting moieties incorporation.90 Table 3 provides an overview of the diverse nanocarrier types.
Table 3.
Summary of the different types of nanocarrier used for drug delivery
|
Nanocarrier type
|
Materials
|
Drug
|
References
|
| Polymeric nanoparticles |
Chitosan |
Bevacizumab |
91
|
| CNAC |
Ranibizumab |
92
|
| Polycaprolactone and Pluronic® F68 |
Triamcinolone acetonide |
93
|
| PGS |
Sunitinib |
94
|
| Generation-4 hydroxyl polyamidoamine dendrimer |
Triamcinolone acetonide |
95
|
| PLGA |
Dexamethasone acetate |
96
|
| PLGA |
bevacizumab |
97
|
| Nanoliposome |
NLC |
Triamcinolone |
98
|
| DPPC (C40H80NO8P) |
Bevacizumab |
99
|
| egg phosphatidylcholine |
Bevacizumab |
100
|
| Albumin nanoparticles |
HSA |
Bevacizumab |
101
|
| Gold nanoparticles |
HAuCl4 |
Resveratrol |
102
|
| zincoxide nanoparticles |
zinc acetate |
Cyperus rotundus leaf extract |
103
|
| magnetic nanoparticles |
iron oxide core and an organic shell exposing carboxylic groups |
Octreotide |
104
|
| Silver nanoparticles |
AgNO3 |
- |
105
|
| silicate nanoparticles |
tetraethoxysilane and Cyclohexane |
- |
106
|
| Fullerene nanoparticles |
C60 fullerene (C60) |
- |
107
|
Chitosan-N-acetyl-L-cysteine (CNAC), Polyglycerol sebacate (PGS), Human serum albumin (HSA), 1, 2-Dipalimitoyl-Sn-glycero-3-phosphocholine (DPPC), nanostructured lipid carrier (NLC; Lipophilic solid lipids used included C16, C18, and a mixture of monoglycerides, diglycerides, and triglyc-erides, Hydrophilic solid lipids used included Ethylene oxide, propylene oxide copolymer, and surfactant)
Utilizing nanocarriers yields many benefits, including enhanced drug bioavailability, improved drug permeation to specific retinal areas, prolonged drug residence time, non-invasive drug delivery, and enhanced ocular tolerability. These advancements represent a substantial leap forward in achieving safer, more efficient, and more convenient medication delivery systems.
Exemplary instances of nanocarriers
Polymeric nanoparticles
Polymeric nanoparticles (PNPs) are polymeric materials comprising nanoscale colloidal organic compounds. These PNPs exhibit configurations resembling nanospheres or nanocapsules.108 Presently, a diverse array of polymers, including chitosan, polycaprolactone, hyaluronic acid, carbopol, eudragit, gelatin, poly butyl polylactic acid, and cyanoacrylate, are harnessed for the fabrication of nanoparticles intended for ocular drug delivery.109-111
Chitosan, notable for its non-toxic, biodegradable, and biocompatible nature, emerges as a polymeric mucoadhesive capable of orchestrating drug release and averting abrupt drug discharges. Various investigations have scrutinized the therapeutic potential of chitosan nanoparticles encapsulating Avastin® for DR treatment, achieving regulated drug dispensation and enhanced intraocular drug transport.112-115
The antiangiogenic prowess of chitosan is ascribed to multiple mechanisms.116,117 It has inhibited tumor invasion and endothelial cell migration. Notably, chitosan has recently demonstrated its capability to mitigate lipopolysaccharide (LPS)-induced interleukin-8 (IL-8) production in endothelial cells, a phenomenon associated with the pathogenesis of vascular disorders.111,118,119 In an endeavor to further augment its efficacy, a chemically modified derivative of chitosan, namely chitosan-N-acetyl-L-cysteine (CNAC), was employed to fabricate nanoparticles for comparative assessment against unaltered chitosan-based nanoparticles in the context of Avastin® delivery via hydrogel.120,121 CNAC distinguishes itself by the presence of N-acetyl-L-cysteine (NAC), a feature absent in unmodified chitosan. This addition introduces thiol groups that forge robust disulfide bonds with cysteine-rich domains in mucus glycoproteins, endowing CNAC with superior mucoadhesive attributes relative to chitosan.120 Given that VEGF comprises cysteine residues, it is conceivable that CNAC may confer additional antiangiogenic activity via binding to VEGF.121 Indeed, an investigation into N-acetylcysteine's antioxidant and antiangiogenic properties in a rat model of DR affirmed NAC's capacity for antiangiogenic and antioxidant actions.122
Nanoliposomes
Nanoliposomes are self-assembling, bilayered, circular particles that share similarities with cell membranes.75 The synthesis of nanoliposome formulations varies depending on their intended application and functionality, with several methods available. These methods encompass thin-film hydration-sonication, ethanol injection, reverse phase evaporation, supercritical fluid technology, heating, and the Mozafari technique.123,124
Liposomes offer numerous advantages, including low toxicity, biodegradability, and non-immunogenicity, rendering them an ideal choice for drug delivery systems.125 Employing liposomes for intravitreal injections enables a controlled and gradual drug release into the vitreous, potentially reducing the frequency of injections.126 Several studies have investigated the use of liposomes for intravitreal administration.127 Abrishami et al. conducted a study examining the impact of nanoliposome encapsulation on bevacizumab following intravitreal injection in rabbits. Their findings revealed that intravitreal nanoliposome injections containing bevacizumab were well-tolerated in rabbits for 42 days. Notably, the clearance rate of this drug from the vitreous when delivered through nanoliposomal formulations was significantly slower than its soluble counterpart. The concentration of bevacizumab following intravitreal injection indicated that this delivery system maintained appropriate therapeutic drug levels for up to six weeks, particularly for diabetic neovascularization and potentially other neovascular eye disorders. These results suggest that the use of nanoliposomes has a beneficial impact in extending the presence of bevacizumab in the vitreous.128
Albumin nanoparticles
Human serum albumin (HSA) is a protein with a molecular weight of 66 kD that is abundantly found in plasma and is widely employed in the pharmaceutical industry as an excipient for various purposes. It boasts remarkable stability in fluid environments and possesses amphiphilic properties. Consequently, HSA proves to be a suitable choice for formulating numerous therapeutic proteins as an additive, serving to mitigate irreversible adsorption to containers or the occurrence of aggregation phenomena.129,130 Standard techniques for generating albumin nanoparticles encompass the desolvation method, thermal-induced aggregation, self-assembly, and albumin-bound technology.131 Owing to its notably high glass transition temperature, HSA can function effectively as a cryoprotectant during freeze-drying processes.132 Moreover, it plays a pivotal role in producing micro- and nanoparticles like Albunex®,133 Abraxane®,134 and bevacizumab-loaded albumin. The latter is created through a desolvation process followed by freeze-drying. Remarkably, these resulting nanoparticles exhibit stability without requiring additional measures, such as cross-linking with glutaraldehyde. This stability is primarily attributed to reinforcing protein-protein interactions between the antibody and albumin. Notably, assessments conducted on ARPE-19 cells revealed these nanoparticles to be non-cytotoxic. Furthermore, when administered as eye drops to laboratory animals, they exhibit a sustained presence on the ocular surface for at least four hours. These collective findings strongly indicate that albumin-based nanoparticles hold significant promise for the ocular delivery of bevacizumab, thereby paving the way for further in vivo evaluations.135
Gold nanoparticles
Gold nanoparticles (GNPs) are agglomerations of particles varying in size from a few to several hundred nanometers, comprising a central gold core enveloped by a surface coating.136 Two fundamental approaches are employed in the synthesis of AuNPs, namely chemical synthesis and biological synthesis. Chemical synthesis methods encompass Turkevich, Brust, seed-mediated growth, and digestive ripening. On the other hand, biological synthesis exploits microorganisms such as bacteria and fungi, as well as plants, algae, and biomolecules.137
The resulting morphology of these nanoparticles can vary, encompassing quasi-spherical, spherical, cubic, triangular, pentagonal, rod-shaped, hexagonal, and plate-like structures, although spherical GNPs have been the most frequently documented.138
Due to their diminutive dimensions and distinct physicochemical attributes, GNPs have garnered considerable attention in drug delivery, bio-imaging, bio-sensing, and nanomedicine.139-141 Investigations have indicated that GNPs can impede VEGF-induced endothelial cell migration through modulation of the Akt pathway, also known as protein kinase B (PKB).142 Generally, GNPs can trigger nanostructural modifications of vascular endothelial growth factor receptor 2 (VEGFR2), thereby hindering VEGFR2 activation and suppressing angiogenesis. Further examinations have suggested that GNPs can inhibit both in vitro and in vivo angiogenesis by inducing autophagy.143,144 The interplay between autophagy and angiogenesis is intricate, as heightened autophagy can either promote or obstruct angiogenesis. Inhibitory autophagy of angiogenesis is often associated with lysosomal dysfunction.145,146 Key proteins involved in autophagosome formation and autophagic cell death,147 such as Autophagy-related protein 5 (ATG5) and Beclin1, displayed increased expression upon GNPs administration, underscoring GNPs' potential as a therapeutic nanomedicine for angiogenesis treatment and offering novel insights into ocular angiogenesis therapy.148
Notably, GNPs have exhibited no detrimental effects on the cellular viability of retinal microvascular endothelial cells, as supported by Jin Hyoung Kim et al. Additionally, intravenous administration of GNPs has not induced any toxicity towards retinal cells, including retinal microvascular cells, retinal neurons, endothelial cells, and astrocytes. Furthermore, when administered at high doses directly into the vitreous cavity, GNPs did not manifest any retinal toxicity. These findings collectively suggest that GNPs may be safely employed in treating various retinopathies without causing harm to the retina or normal retinal vessels.149
Magnetic nanoparticles
Magnetic nanoparticles (MNPs) represent nanoscale materials of magnetic elements, such as nickel, iron, manganese, cobalt, gadolinium, chromium, and various chemical compounds. Owing to their nanoscale dimensions, MNPs exhibit superparamagnetic properties. They hold immense potential across diverse applications in their unmodified form. However, selecting functional groups and applying surface coatings are imperative for specific applications. Among MNPs, ferrite nanoparticles have garnered substantial research attention.150,151 Various synthetic techniques are employed to attain the desired shape, size, stability, and biocompatibility of MNPs. Standard methods for MNP synthesis include ball milling, thermal decomposition, coprecipitation, microemulsion, hydrothermal, sol-gel processes, and biological approaches.152
MNPs hold promise for intraocular drug delivery.153 Intraocular MNPs have demonstrated rapid and sustained penetration into the retina, explicitly targeting the retinal pigment epithelium (RPE) without inducing any tissue damage.154 Furthermore, MNPs possess significant capabilities in loading and delivering specific molecules. Notably, MNPs have been effective carriers for brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF), enhancing their efficacy in preventing retinal oxidative damage.155
Amato R et al. conducted a study to assess the feasibility of employing magnetic nanoparticle-octreotide (MNP-OCT) to treat DR. Their evaluation examined MNP-OCT's ability to suppress VEGF-induced pro-angiogenic responses in human retinal endothelial cells (HRECs) and its effectiveness in safeguarding ex-vivo retinal explants from oxidative stress (OS)-induced apoptosis. Furthermore, they investigated the actual localization of MNP-OCT in retinas in vivo following intraocular injection. The results of their experiments in animal models demonstrated that MNPs exhibit no toxicity to ocular tissues. Furthermore, the biocompatibility of MNPs was corroborated in ex-vivo mouse retinal explants, where no apoptotic activity was observed in the presence of MNPs.104
Silver nanoparticles
Silver nanoparticles (Ag NPs) have emerged as a prominent subject of investigation within the realm of nanoparticles in recent decades. These Ag NPs typically comprise 20 to 15,000 silver atoms and exhibit diameters below the 100 nm threshold.109 Silver, a naturally occurring element, is characterized by its distinctive white appearance, softness, lustrous sheen, and notable thermal and electrical conductivity.
Various methodologies can be employed to synthesize Ag NPs, encompassing physical, chemical, biological, and mechanical approaches. Among these, chemical processes have gained prominence as a standard and straightforward means of Ag NP fabrication.156
Due to their remarkable antimicrobial attributes, Ag NPs find application in many domains, including wound dressings, contraceptive devices, surgical equipment, and bone prostheses. Moreover, Ag NPs are extensively utilized to coat ocular lenses, effectively thwarting microbial activity. Additionally, these nanoparticles have been recognized for their antifungal characteristics, anti-viral efficacy, and anti-inflammatory properties.157-163
Gurunathan et al have presented compelling evidence suggesting that Ag NPs function as potent antiangiogenic agents, effectively suppressing angiogenesis initiated by VEGF via the PI3K/Akt pathway.164
Pathological conditions often manifest with heightened permeability in choroidal and retinal vessels, as seen in age-related macular degeneration and diabetes.165 Sheikpranbabu et al have elucidated that Ag NPs possess the capability to inhibit retinal vascular hyperpermeability induced by advanced glycation end-products-bovine serum albumin (AGE-BSA), a condition characterized by the suppression of intracellular adhesion molecule-1 (ICAM-1) expression and the upregulation of tight junction proteins ZO-1 and occludin166. Additionally, their study unveiled Ag NPs' potential to counteract permeability induced by VEGF and interleukin-1β (IL-1β) via the deactivation of the Src kinase pathway, thus presenting a promising therapeutic avenue for ocular diseases such as DR.167
Kalishwaralal et al have contributed further insights by reporting that Ag NPs exert inhibitory effects on cell survival through the PI3K/Akt-dependent pathway in retinal endothelial cells.168
Silicate nanoparticles
Silica nanoparticles (SiNPs) are non-crystalline, spherical particles in various forms and sizes with easily modifiable surface properties to cater to diverse applications. In their nonporous state, SiNPs exhibit abrasive and absorbent characteristics. However, mesoporous SiNPs, characterized by hexagonal pore architectures, hold significant promise within nanomedicine and therapeutic interventions.124 Several methods exist for synthesizing SiNPs, resulting in a range of sizes from 10 to 500 nm and diverse morphologies and physicochemical attributes. Stober's procedure and microemulsion synthesis are among the widely employed synthesis techniques.169
SiNPs find utility in gene therapy and drug delivery, whether used independently or with other treatment modalities.170-172 A study by Mohammadpour et al posited that the small size of SiNPs allows them to permeate corneal epithelial tight junctions effectively, suggesting their potential efficacy in treating and preventing corneal neovascularization. Further investigations could explore the application of SiNPs in controlling retinal neovascularization associated with vascular disorders and DR.173
Jo et al demonstrated that SiNPs hold promise for the treatment of retinal neovascularization without inducing toxicity. SiNPs effectively inhibited VEGF-induced retinal neovascularization and suppressed the activation of extracellular signal-regulated protein kinase (ERK 1/2) by inhibiting VEGFR-2 phosphorylation. Pathological angiogenesis, driven by angiogenic molecules like VEGF, contributes to angiogenesis-related blindness (ARB), making SiNPs and siRNAs potential candidates for preventing vision loss.174
Observational research has shown that magnesium silicate nanoparticles exhibit low toxicity. Specifically, Emodin–MgSiO3 effectively impedes the expression of both protein and the VEGF gene. This suggests that magnesium silicate hollow spheres hold promise as safe drug carriers.175
Zinc oxide nanoparticles
Zinc oxide (ZnO) is categorized as an n-type semiconducting metal oxide with commendable biocompatibility, safety, and enduring efficacy, rendering it a viable option for addressing various medical conditions. In the context of diabetes mellitus, zinc oxide nanoparticles (ZnO NPs) have garnered significant attention owing to their capability to administer zinc ions, as substantiated by references.176-178 An array of investigations have unequivocally demonstrated that ZnO NPs exhibit negligible toxicity towards human cells, as corroborated.179
ZnO NPs can be synthesized through diverse chemical methodologies encompassing vapor transfer, precipitation, and hydrothermal routes. However, it is noteworthy that the contemporary focus of research lies in the burgeoning domain of biogenic synthesis of ZnO NPs, which entails the utilization of various plant extracts or microorganisms. This biological approach to nanoparticle synthesis offers many advantages when compared to conventional chemical and physical methods, as elaborated.180
In a recent study, DR in rats was targeted for treatment using zinc oxide nanoparticles loaded with extracts from Cyperus rotundus (CR-ZnONPs). The experimental findings demonstrated significant improvements in key parameters, including fasting blood sugar (FBS), retina thickness, insulin levels, and HbA1c, all of which returned to average values. These outcomes suggest that CR-ZnONPs possess remarkable anti-inflammatory and anti-diabetic properties.181
Fullerene nanoparticles
Fullerenes represent the third naturally occurring allotropic variation of carbon.181 The pseudo-aromatic structure of C60 molecules, characterized by the delocalization of π-electrons over its carbon core, is primarily determined by the presence of sp2,5-bonds.182
Numerous scientific publications have detailed various methodologies for synthesizing fullerenes, which can possess varying degrees of hydroxylation while conforming to the general formula C60. These methods predominantly involve the solubilization of fullerenes in water through solvent exchange utilizing diverse organic solvents such as toluene, ethanol, acetone, and THF, often aided by sonication or mechanical stirring. Furthermore, it has been documented that C60 can be directly solubilized in water through prolonged stirring or sonication.183
Fullerene has recently garnered significant attention as an up-and-coming candidate for numerous medical applications.184 Ever since research commenced on C60's antioxidant properties, it has been posited that the extensive electron-conjugation system is a unique characteristic of fullerene molecules. Until recently, fullerene was regarded as a groundbreaking "structural" antioxidant, a notion articulated by Krusic, who referred to it as a "radical sponge".185
The principal mechanism by which C60 benefits astrocytes is through its capacity to neutralize free radicals and safeguard cellular membranes from oxidative damage. This property has significant implications for astrocyte survival. Strikingly, fullerene also exerts a profound influence on signaling pathways associated with the regulation of apoptosis, offering a novel therapeutic approach for addressing cellular dysfunction in DR.107
Concurrently with the progression of DR, there is a noteworthy occurrence of pro-inflammatory and pro-oxidative alterations in retinal cells, including astrocytes and Muller cells. Glial reactivity has been recognized as a pivotal pathogenetic factor in various neural tissue disorders. The anti-inflammatory and antioxidant attributes of fullerene C60 nanoparticles have been empirically substantiated. Nedzvetsky et al investigated the glioprotective efficacy of water-soluble hydrated fullerene C60 (C60HyFn) in a 12-week STZ-diabetes model. Their findings indicated that C60HyFn treatment ameliorated astrocyte reactivity in the STZ-diabetic rat group, as evidenced by reduced S100β and PARP1 overexpression. Additionally, the retinas of STZ-diabetic rats subjected to C60HyFn treatment exhibited diminished TNFα production. These results underscore the glioprotective potential of C60HyFn on retinal cells, suggesting its viability as a prospective nano-strategy for DR therapy.186
Intraocular implants
Ocular drug delivery implants have garnered significant attention as a solution to address the inherent limitations associated with conventional eye therapies.187-190 These implants offer distinct advantages, such as ease of administration, precise drug delivery to ocular tissues, and minimal interference with the normal functioning of the eye.191 Specifically, intravitreal implants represent a class of drug delivery systems designed for either injection or surgical implantation into the vitreous humor, facilitating sustained drug release to the posterior and intermediate regions of the eye.192
Over the past few decades, the medical community has witnessed a burgeoning interest in bioimplants. Bioimplants encompass diverse technologies to enhance the functionality of damaged natural organs. These encompass brain/neural implants,193 sensory implants,194 spinal implants,195 organ stimulation implants,196 subcutaneous implants,197 dental implants,198 cosmetic implants,199 and convenience implants. Additionally, structural implants such as rods, braces, heart valves, pins, bones, hip prostheses, ear implants, ocular implants, skull implants, and knee replacements have played pivotal roles in improving patients' quality of life. Bioimplants have ushered in a new era of drug delivery systems, enhancing therapeutic outcomes while mitigating side effects through targeted and localized drug administration.200 This localized approach has improved drug bioavailability at the desired site, reduced dosing frequency, and eliminated systemic side effects. Furthermore, an essential feature of these implants is their ability to be removed in the event of adverse effects.
Implants can be classified into two primary categories: passive and active implants. Passive implants, in turn, can be further divided into biodegradable and non-biodegradable varieties. Biodegradable implants have been crafted using materials like polycaprolactone (PCL), polylactic acid (PLA), and polylactic-co-glycolic acid (PLGA).201,202 Conversely, non-biodegradable implants are typically manufactured from materials such as silicones, polyurethanes, polyacrylates, and polyethylene vinyl acetate.203-209
One notable application of implants is sustained-release steroids, which have proven effective in reducing inflammation and managing macular edema associated with DR. These implants provide extended therapeutic effects while minimizing the risk of systemic side effects compared to systemic steroid administration. Nevertheless, it is essential to acknowledge potential drawbacks, including the risk of cataract formation, elevated intraocular pressure as a side effect of steroid use, implant dislocation or migration, and the need for meticulous monitoring. Prolonged use may necessitate the management of steroid-related complications.210
Active implants harness two principal mechanisms to control drug release: osmotic pressure gradients and electromechanical drives.211
To exemplify the innovation in ocular implants, one can reference the work of Maulvi et al.211 They devised a novel ocular implant for timolol maleate (TM) delivery, starting with loading ethyl cellulose nanoparticles within hydrogel rings in a multi-step process for controlled drug administration in the treatment of Glaucoma. The initial step involved the synthesis of TM-ethyl cellulose nanoparticles using the double emulsion method. Subsequently, hydrogel implants were crafted through a free radical polymerization process, employing HEMA (hydroxyethyl-methyl acrylate) as monomers and ethylene glycol dimethacrylate as a cross-linker. In the final step, TM-encapsulated ethyl cellulose nanoparticles were dispersed within an acrylate hydrogel. Compared to traditional eye drop therapy, in vivo pharmacokinetic assessments demonstrated an increase in mean residence time (MRT) and area under the curve (AUC) with TM implant contact lenses.212 The success of this innovative approach has ignited interest in developing similar implants for treating retinopathy and other ocular diseases.
Exemplary instances of intraocular implants
Various sustained-release corticosteroid delivery systems have undergone scrutiny for therapeutic purposes. The fluocinolone acetonide intravitreal implant, commercially available as Retisert, is the pioneering device in this domain. This particular implant is the subject of investigation for its efficacy in treating DME and autoimmune retinopathy.213,214 A more compact device, designed for in-clinic administration under the name Iluvien, is also examined for DME treatment.215 Another noteworthy option is the biodegradable, extended-release dexamethasone implant known as Ozurdex, which can be introduced in a clinical setting to address macular edema. Ozurdex comprises a biodegradable copolymer encompassing lactic acid and glycolic acid, laden with an adjustable quantity of dexamethasone. Many in vitro and in vivo studies have firmly established its biocompatibility.216,217 Furthermore, the triamcinolone-eluting intravitreal implant, named I-vation, has also been explored as a therapeutic approach for DME.218
Constraints and complexities of intraocular drug delivery systems
Hydrogels
Despite their broad potential applications in ophthalmology, the commercialization of products based on hydrogels has remained relatively limited. In contrast, hydrogel-based soft contact lenses (SCLs) and foldable intraocular lenses (IOLs) have showcased exceptional efficacy. However, the practical deployment of hydrogel-based vitreous substitutes or intravitreal medication delivery systems currently faces substantial challenges. Several factors contribute to this situation. First and foremost, sterilizing hydrogels poses a formidable challenge.219,220 Conventional thermal sterilization tends to degrade most natural and synthetic polymers, while radiation or chemical sterilization may trigger side reactions that alter the inherent characteristics of the hydrogel. These issues are exacerbated when incorporating proteins or other biopharmaceuticals.221
Aseptic processing offers a viable solution for sterilizing hydrogel drug delivery systems. Maintaining aseptic conditions throughout the manufacturing process, encompassing material handling, formulation, and packaging, minimizes the risk of introducing contaminants while preserving the delicate attributes of the hydrogel and any incorporated biopharmaceuticals. Filtration represents another standard method for sterilizing heat-sensitive materials and solutions in hydrogel drug delivery systems. This method passes the product through sterile filters to eliminate microorganisms and particulate matter.222
Furthermore, pre-formed hydrogels exhibit a finite shelf life, and the reactivity of in situ cross-linkable polymers may diminish during storage. The shelf life of a hydrogel largely hinges on its composition and the cross-linking methodology employed. Hydrogels featuring stable, covalently cross-linked polymer networks exhibit superior long-term stability to those with physically cross-linked structures, which may be more susceptible to degradation over time. Environmental factors, such as temperature, humidity, and light exposure, can significantly influence hydrogel stability. Commercially available hydrogel formulations are typically engineered to withstand various environmental conditions, and packaging considerations may further contribute to overall stability.223
Moreover, the selection and quantity of medications that can be integrated into hydrogels remain constrained.224 While physical cross-linking may enhance the stability of encapsulated therapeutic proteins, it may simultaneously limit control over gel degradation and drug release.225 Finally, antibodies released from hydrogels at a slower rate may display instability over several weeks.226
Nanocarriers
Ophthalmic drug delivery faces numerous challenges attributed to the eyes' distinctive physiological and anatomical features. These intricate organs present several formidable barriers that must be surmounted to target specific ocular tissues.227 Additionally, emerging scenarios, such as the initial burst release from nanoparticles, compound the limitations of ocular drug delivery. Furthermore, investigating toxicity in retinal cells remains incomplete, primarily conducted in vitro, leaving essential gaps in the understanding.228,229 While nanoparticles are generally biocompatible, factors such as their charge, particle size, drug concentration, and duration of exposure significantly influence their potential toxicity to the retina. Typically, the interaction between cationic surfaces of nanoparticles and negatively charged cell membranes serves as the predominant catalyst for toxicity.230 These limitations are not exclusive to nanoparticles; other types of formulations also face similar obstacles when delivering drugs to the specific tissues within the eye. For instance, traditional eye drops encounter difficulties in achieving targeted delivery to the intended ocular tissues. Additionally, challenges exist for ointments, gels, and intravitreal injections in ensuring effective and sustained drug delivery while minimizing potential side effects.231
Intraocular implants: surgical considerations and challenges
The implantation procedure for intraocular implants necessitates a surgical intervention, inherently carrying associated risks. Moreover, post-surgical complications, such as infection, inflammation, elevated intraocular pressure, and cataract formation, may manifest. In certain instances, the extraction of intraocular implants can be a formidable task, mainly when complications arise, or implant replacement is imperative.217
Limitations and complexities in intraocular drug delivery systems
Nanotechnology has brought substantial promise in intraocular drug delivery, encompassing nanocarriers, hydrogels, and implants. Despite promising outcomes in laboratory settings, the practical utilization of these carriers remains encumbered by difficulties. Notably, attempts involving nanoparticles have yielded less sanguine results. The heterogeneity inherent to these carriers, potentially leading to therapeutic instability, is a significant challenge.232 Furthermore, paramount concerns concerning immunological responses and toxicity must be addressed. In sum, using nanocarriers, hydrogels, and implants in intraocular formulations holds excellent potential, necessitating further extensive investigation to surmount the challenges encountered during experimental application. With persistent exploration and advancement, these carriers can revolutionize the treatment of DR.
Beyond traditional dosage forms, ongoing clinical trials and innovative formulations are under development for DR treatment. Some researchers are exploring sustained-release implants, such as Retisert and Ozurdex, designed to administer medications directly into the eye over an extended period. These implants can reduce the required injection frequency and enhance patient compliance.213,216
Researchers are also investigating the potential advantages of combining diverse treatment modalities to address multiple facets of DR. Concurrently; clinical trials are underway to assess the efficacy of combining anti-VEGF therapy with other pharmaceutical agents like Bevacizumab and Triamcinolone, as well as laser therapy or surgical interventions. The aim is to improve visual outcomes and disease progression substantially.233
Future research endeavors will tackle the challenges of scaling up and industrial production, ensuring efficient and cost-effective manufacturing of novel drug delivery systems. This may encompass the development of scalable production processes, optimization of manufacturing technologies, and tailored implementation of quality control measures in line with the specific requirements of DR treatments. As novel drug delivery systems for DR continue to evolve, a comprehensive assessment of cost-benefit considerations will be imperative. Researchers will scrutinize the overall impact on healthcare expenditure, patient outcomes, and resource utilization, offering valuable insights to healthcare decision-makers and stakeholders.
In forthcoming research endeavors, there will be a pronounced emphasis on the rigorous conduct of clinical trials aimed at appraising the safety, efficacy, and relative effectiveness of innovative drug delivery methodologies for treating DR. These trials will not solely gauge the therapeutic advantages inherent in the novel delivery systems but will also delve into ancillary aspects such as patient adherence, quality of life enhancements, and the enduring consequences of treatment.
Long-term safety assessments will emerge as an imperative sphere of concentration in prospective research endeavors. This undertaking encompasses the meticulous surveillance of the cumulative ramifications stemming from the employment of pioneering drug delivery systems for DR across protracted timeframes. It comprehensively scrutinizes potential adversities and the safety profile of these therapeutic modalities.
Significant strides in nanotechnology, sustained-release formulations, targeted drug delivery, and gene therapy are propelling groundbreaking innovations within the ambit of DR treatment. As these innovative approaches continue their evolutionary trajectory, the symbiotic partnership between researchers, pharmaceutical entities, regulatory authorities, and healthcare providers is pivotal in propelling the development, acceptance, and efficacious integration of pioneering drug delivery solutions for managing DR.
Methodology for literature search
The systematic exploration of relevant studies was done through comprehensive searches across Web of Science, Scopus, Google Scholar, and PubMed. This exhaustive investigation spanned from 1993 through the conclusion of 2022. The search queries were centered on the following keywords: nanotechnology, DR, nanoparticles, hydrogels, intraocular implants, and drug delivery. These queries were executed without imposing any language or date restrictions. The screened articles encompassed titles and abstracts, focusing on those elucidating the roles of nanoparticles, hydrogels, intraocular implants, and the effects of drug delivery.
Concluding remarks
The advent of novel intraocular delivery systems has the potential to address the limitations inherent in traditional anti-neovascular therapies while simultaneously forging innovative therapeutic avenues. The realization of efficacious treatments is now on the horizon, thanks to the emergence of novel intraocular delivery devices capable of extending intravitreal drug administration intervals, facilitating the introduction of corrective genes into ocular tissues, or eliminating the necessity for direct ocular injections. In the coming years, there is eager anticipation for translating promising preclinical evidence into successful clinical trials that can unequivocally demonstrate safety and efficacy in the context of human patients.
Furthermore, the newfound antiangiogenic attributes associated with selected intraocular delivery methods may pave the way for integrating intriguing composite materials that synergistically combat neovascular eye diseases. Nanocarriers, hydrogels, and implants have all been meticulously crafted from diverse materials possessing a spectrum of physicochemical characteristics. In addition, concerted efforts have been dedicated to refining the structural attributes of nanocarriers, hydrogels, and implants, primarily enhancing their intraocular delivery efficiency. This optimization involves manipulating their internal and external morphologies, enhancing their stability, and modulating their release kinetics.
Moreover, incorporating biologically responsive components into nanocarriers, hydrogels, and implants has enabled the customization of their responsiveness to various stimuli, including magnetic fields, ultrasound, pH fluctuations, and even dual or multi-stimuli responsiveness. This technological advancement has opened doors to highly specialized therapies characterized by precise site-specific drug release and improved therapeutic outcomes. In summary, the realm of novel intraocular delivery harbors tremendous potential to shape the future landscape of ocular antiangiogenic therapy.
Review Highlights
What is the current knowledge?
√ Various novel drug delivery strategies have been examined for targeted ocular drug delivery.
√ Novel carriers such as implants, hydrogels, and nanocarriers have been employed to deliver drugs in a controlled manner to the retina and vitreous.
What is new here?
√ Current progress and contemporary applications of carriers in treating diabetic retinopathy.
√ Administration routes, prospective future developments, and challenges in the field of novel carriers for treating diabetic retinopathy.
Acknowledgements
The authors wish to thank Isfahan University of Medical Sciences for supporting this work, Grant Number 140124
Competing Interests
The authors declare no competing interests.
Ethical Statement
Not applicable.
References
- Safari J, Zarnegar Z. Advanced drug delivery systems: Nanotechnology of health design A review. J Saudi Chem Soc 2014; 18:85-99. doi: 10.1016/j.jscs.2012.12.009 [Crossref] [ Google Scholar]
- Shatalebi M, Mostafavi SA, Moghaddas A. Niosome as a drug carrier for topical delivery of N-acetyl glucosamine. Res Pharm Sci 2010; 5:107. [ Google Scholar]
- Koo OM, Rubinstein I, Onyuksel H. Role of nanotechnology in targeted drug delivery and imaging: a concise review. Nanotechnol Biol Med 2005; 1:193-212. doi: 10.1016/j.nano.2005.06.004 [Crossref] [ Google Scholar]
- Farokhzad OC, Langer R. Impact of nanotechnology on drug delivery. ACS nano 2009; 3:16-20. doi: 10.1021/nn900002m [Crossref] [ Google Scholar]
- Singh M, Hemant K, Ram M, Shivakumar H. Microencapsulation: A promising technique for controlled drug delivery. Res Pharm Sci 2010; 5:65. [ Google Scholar]
- Poorirani S, Taheri SL, Mostafavi SA. Scaffolds: a biomaterial engineering in targeted drug delivery for osteoporosis. Osteoporos Int 2023; 34:255-67. doi: 10.1007/s00198-022-06543-3 [Crossref] [ Google Scholar]
- Standl E, Khunti K, Hansen TB, Schnell O. The global epidemics of diabetes in the 21st century: Current situation and perspectives. Eur J Prev Cardiol 2019; 26:7-14. doi: 10.1177/2047487319881021 [Crossref] [ Google Scholar]
- Fong DS, Aiello L, Gardner TW, King GL, Blankenship G, Cavallerano JD. Retinopathy in diabetes. Diabetes Care 2004; 27:s84-s7. doi: 10.2337/diacare.27.2007.s84 [Crossref] [ Google Scholar]
- Falcão M, Falcão-Reis F, Rocha-Sousa A. Diabetic retinopathy: understanding pathologic angiogenesis and exploring its treatment options. The Open Circulation and Vascular Journal 2010; 3:30-42. doi: 10.2174/1874382601003010030 [Crossref] [ Google Scholar]
- Lai AKW, Lo AC. Animal models of diabetic retinopathy: summary and comparison. J Diabetes Res 2013; 2013:106594. doi: 10.1155/2013/106594 [Crossref] [ Google Scholar]
- Ciulla TA, Amador AG, Zinman B. Diabetic retinopathy and diabetic macular edema: pathophysiology, screening, and novel therapies. Diabetes Care 2003; 26:2653-64. doi: 10.2337/diacare.26.9.2653 [Crossref] [ Google Scholar]
- Bandello F, Polito A, Pognuz DR, Monaco P, Dimastrogiovanni A, Paissios J. Triamcinolone as adjunctive treatment to laser panretinal photocoagulation for proliferative diabetic retinopathy. Arch Ophthalmol 2006; 124:643-50. doi: 10.1001/archopht.124.5.643 [Crossref] [ Google Scholar]
- Jaffe GJ, Martin D, Callanan D, Pearson PA, Levy B, Comstock T. Fluocinolone acetonide implant (Retisert) for noninfectious posterior uveitis: thirty-four–week results of a multicenter randomized clinical study. Ophthalmology 2006; 113:1020-7. doi: 10.1016/j.ophtha.2006.02.021 [Crossref] [ Google Scholar]
- Kuppermann BD, Blumenkranz MS, Haller JA, Williams GA, Weinberg DV, Chou C. Randomized controlled study of an intravitreous dexamethasone drug delivery system in patients with persistent macular edema. Arch Ophthalmol 2007; 125:309-17. doi: 10.1001/archopht.125.3.309 [Crossref] [ Google Scholar]
- Elakkiya M, Selvaraj K, Kuppuswamy G. Current and Emerging Therapies for the Management of Diabetic Retinopathy. J Appl Pharm Sci 2017; 7:243-51. doi: 10.7324/JAPS.2017.70933 [Crossref] [ Google Scholar]
- Gonzalez VH, Giuliari GP, Banda RM, Guel DA. Intravitreal injection of pegaptanib sodium for proliferative diabetic retinopathy. Br J Ophthalmol 2009; 93:1474-8. doi: 10.1136/bjo.2008.155663 [Crossref] [ Google Scholar]
- Arevalo JF, Lasave AF, Wu L, Maia M, Diaz-Llopis M, Alezzandrini AA. Intravitreal bevacizumab for proliferative diabetic retinopathy. Retina 2017; 37:334-43. doi: 10.1097/IAE.0000000000001181 [Crossref] [ Google Scholar]
- Stewart MW. A review of ranibizumab for the treatment of diabetic retinopathy. Ophthalmol Ther 2017; 6:33-47. doi: 10.1007/s40123-017-0083-9 [Crossref] [ Google Scholar]
- Deissler HL, Lang GE. The protein kinase C inhibitor: ruboxistaurin. Retinal Pharmacotherapeutics. Karger Publishers; 2016. p. 295-301. 10.1159/000431204.
- Schwartz SG, Flynn HW. Pharmacotherapies for diabetic retinopathy: present and future. Exp Diabetes Res 2007; 2007:52487. doi: 10.1155/2007/52487 [Crossref] [ Google Scholar]
- Shord SS, Bressler LR, Tierney LA, Cuellar S, George A. Understanding and managing the possible adverse effects associated with bevacizumab. Am J Health-Syst Pharm 2009. 66: 999-1013. 10.2146/ajhp080455.
- Sharma DS, Wadhwa S, Gulati M, Kadukkattil Ramanunny A, Awasthi A, Singh SK, et al. Recent advances in intraocular and novel drug delivery systems for the treatment of diabetic retinopathy. Expert Opin Drug Deliv 2021. 18: 553-76. 10.1080/17425247.2021.1846518.
- Sontyana AG, Mathew AP, Cho K-H, Uthaman S, Park I-K. Biopolymeric in situ hydrogels for tissue engineering and bioimaging applications. Tissue Eng Regen Med 2018; 15:575-90. doi: 10.1016/j.jconrel.2017.01.012 [Crossref] [ Google Scholar]
- Mandal A, Bisht R, Rupenthal ID, Mitra AK. Polymeric micelles for ocular drug delivery: from structural frameworks to recent preclinical studies. J Control Release 2017; 248:96-116. doi: 10.1016/j.jconrel.2017.01.012 [Crossref] [ Google Scholar]
- He C, Kim SW, Lee DS. In situ gelling stimuli-sensitive block copolymer hydrogels for drug delivery. J Control Release 2008; 127:189-207. doi: 10.1016/j.jconrel.2008.01.005 [Crossref] [ Google Scholar]
- Mehnert W, Mäder K. Solid lipid nanoparticles: production, characterization and applications.Adv Drug Deliv Rev2012. 64: 83-101. 10.1016/j.addr.2012.09.021.
- Lin C-C, Metters AT. Hydrogels in controlled release formulations: network design and mathematical modeling. Adv Drug Deliv Rev 2006; 58:1379-408. doi: 10.1016/j.addr.2006.09.004 [Crossref] [ Google Scholar]
- Zhu Y, Sheng Y. Sustained delivery of epalrestat to the retina using PEGylated solid lipid nanoparticles laden contact lens. Int J Pharm 2020; 587:119688. doi: 10.1016/j.ijpharm.2020.119688 [Crossref] [ Google Scholar]
- Yazdani M, Tavakoli O, Khoobi M, Wu YS, Faramarzi MA, Gholibegloo E. Beta-carotene/cyclodextrin-based inclusion complex: improved loading, solubility, stability, and cytotoxicity. J Incl Phenom Macrocycl Chem 2021; 102:55-64. doi: 10.1007/s10847-021-01100-7 [Crossref] [ Google Scholar]
- Meza-Rios A, Navarro-Partida J, Armendariz-Borunda J, Santos A. Therapies based on nanoparticles for eye drug delivery. Ophthalmol Ther 2020; 9:1-14. doi: 10.1007/s40123-020-00257-7 [Crossref] [ Google Scholar]
- Janoria KG, Gunda S, Boddu SH, Mitra AK. Novel approaches to retinal drug delivery. Expert Opin Drug Deliv 2007; 4:371-88. doi: 10.1517/17425247.4.4.371 [Crossref] [ Google Scholar]
- Lee SS, Robinson MR. Novel drug delivery systems for retinal diseases. Ophthalmic Res2009. 41: 124-35. 10.1159/000209665.
- Donahue ND, Acar H, Wilhelm S. Concepts of nanoparticle cellular uptake, intracellular trafficking, and kinetics in nanomedicine. Adv Drug Deliv Rev 2019; 143:68-96. doi: 10.1016/j.addr.2019.04.008 [Crossref] [ Google Scholar]
- Dhiman N, Awasthi R, Sharma B, Kharkwal H, Kulkarni GT. Lipid nanoparticles as carriers for bioactive delivery. Fron Chem 2021; 9:580118. doi: 10.3389/fchem.2021.580118 [Crossref] [ Google Scholar]
- Zorkina Y, Abramova O, Ushakova V, Morozova A, Zubkov E, Valikhov M. Nano carrier drug delivery systems for the treatment of neuropsychiatric disorders: Advantages and limitations. Molecules 2020; 25:5294. doi: 10.3390/molecules25225294 [Crossref] [ Google Scholar]
- Nayak K, Choudhari MV, Bagul S, Chavan TA, Misra M. Ocular drug delivery systems. In: Drug Delivery Devices and Therapeutic Systems. Elsevier; 2021. p. 515-66.
- Agrahari V, Mandal A, Agrahari V, Trinh HM, Joseph M, Ray A. A comprehensive insight on ocular pharmacokinetics. Drug Deliv Transl Res 2016; 6:735-54. [ Google Scholar]
- Ahmed S, Amin MM, Sayed S. Ocular drug delivery: a comprehensive review. AAPS PharmSciTech 2023; 24:66. [ Google Scholar]
- Tsung T-H, Tsai Y-C, Lee H-P, Chen Y-H, Lu D-W. Biodegradable Polymer-Based Drug-Delivery Systems for Ocular Diseases. Int Jo Mol Sci 2023; 24:12976. [ Google Scholar]
- Ahmed S, Amin MM, Sayed S. Ocular drug delivery: a comprehensive review. AAPS PharmSciTech 2023; 24:66. doi: 10.1208/s12249-023-02516-9 [Crossref] [ Google Scholar]
- Silva M, Peng T, Zhao X, Li S, Farhan M, Zheng W. Recent trends in drug-delivery systems for the treatment of diabetic retinopathy and associated fibrosis. Adv Drug Deliv Rev 2021; 173:439-60. doi: 10.1016/j.addr.2021.04.007 [Crossref] [ Google Scholar]
- Piyasena MMPN, Murthy GVS, Yip JL, Gilbert C, Zuurmond M, Peto T. Systematic review on barriers and enablers for access to diabetic retinopathy screening services in different income settings. PloS One 2019; 14:e0198979. doi: 10.1371/journal.pone.0198979 [Crossref] [ Google Scholar]
- Bisht R, Mandal A, Jaiswal JK, Rupenthal ID. Nanocarrier mediated retinal drug delivery: overcoming ocular barriers to treat posterior eye diseases. Nanomedicine and Nanobiotechnology 2018; 10:e1473. doi: 10.1002/wnan.1473 [Crossref] [ Google Scholar]
- Wichterle O, Lim D. Hydrophilic gels for biological use. Nature 1960; 185:117. doi: 10.1038/185117a0 [Crossref] [ Google Scholar]
- Peppas NA, Khare AR. Preparation, structure and diffusional behavior of hydrogels in controlled release. Adv Drug Deliv Rev 1993; 11:1-35. doi: 10.1016/0169-409x(93)90025-y [Crossref] [ Google Scholar]
- Zheng L, Han Y, Ye E, Zhu Q, Loh XJ, Li Z. Recent Advances in Ocular Therapy by Hydrogel Biomaterials. World Scientific Annual Review of Functional Materials 2023; 1:2230002. doi: 10.1142/s2810922822300021 [Crossref] [ Google Scholar]
- Arslan M, Sanyal R, Sanyal A. Cyclodextrin embedded covalently crosslinked networks: synthesis and applications of hydrogels with nano-containers. Polym Chem 2020; 11:615-29. doi: 10.1039/c9py01679a [Crossref] [ Google Scholar]
- Chimene D, Kaunas R, Gaharwar AK. Hydrogel bioink reinforcement for additive manufacturing: a focused review of emerging strategies. Adv Mater 2020; 32:1902026. doi: 10.1002/adma.201902026 [Crossref] [ Google Scholar]
- Xu X, Jerca VV, Hoogenboom R. Bioinspired double network hydrogels: from covalent double network hydrogels via hybrid double network hydrogels to physical double network hydrogelsMater. Horiz 2021; 8:1173-88. doi: 10.1039/d0mh01514h [Crossref] [ Google Scholar]
- Acet Ö, Aksoy NH, Erdönmez D, Odabaşı M. Determination of some adsorption and kinetic parameters of α-amylase onto Cu + 2-PHEMA beads embedded column. Artif Cells Nanomed Biotechnol 2018; 46:S538-S45. doi: 10.1080/21691401.2018.1501378 [Crossref] [ Google Scholar]
- Qiu Y, Park K. Environment-sensitive hydrogels for drug delivery. Adv Drug Deliv Rev 2001; 53:321-39. doi: 10.1016/j.addr.2012.09.024 [Crossref] [ Google Scholar]
- Tan H, Marra KG. Injectable, biodegradable hydrogels for tissue engineering applications. Mater 2010; 3:1746-67. doi: 10.3390/ma3031746 [Crossref] [ Google Scholar]
- Gao Y, Sun Y, Ren F, Gao S. PLGA–PEG–PLGA hydrogel for ocular drug delivery of dexamethasone acetate. Drug Dev Ind Pharm 2010; 36:1131-8. doi: 10.3109/03639041003680826 [Crossref] [ Google Scholar]
- Huang H, Qi X, Chen Y, Wu Z. Thermo-sensitive hydrogels for delivering biotherapeutic molecules: A review. Saudi Pharm J 2019; 27:990-9. doi: 10.1016/j.jsps.2019.08.001 [Crossref] [ Google Scholar]
- Zarrintaj P, Jouyandeh M, Ganjali MR, Hadavand BS, Mozafari M, Sheiko SS. Thermo-sensitive polymers in medicineA review. Eur Polym J 2019; 117:402-23. doi: 10.1016/j.eurpolymj.2019.05.024 [Crossref] [ Google Scholar]
- Oh JK, Lee DI, Park JM. Biopolymer-based microgels/nanogels for drug delivery applications. Prog Polym Sci 2009; 34:1261-82. doi: 10.1016/j.progpolymsci.2009.08.001 [Crossref] [ Google Scholar]
- Maitra J, Shukla VK. Cross-linking in hydrogels-a review. Am J Polym Sci 2014; 4:25-31. doi: 10.5923/j.ajps.20140402.01 [Crossref] [ Google Scholar]
- Ahmed EM. Hydrogel: Preparation, characterization, and applications: A review. J Adv Res 2015; 6:105-21. doi: 10.1016/j.jare.2013.07.006 [Crossref] [ Google Scholar]
- Koetting MC, Peters JT, Steichen SD, Peppas NA. Stimulus-responsive hydrogels: Theory, modern advances, and applications. Mater Sci Eng R Rep MAT SCI ENG R 2015; 93:1-49. doi: 10.1016/j.mser.2015.04.001 [Crossref] [ Google Scholar]
- Ullah F, Othman MBH, Javed F, Ahmad Z, Akil HM. Classification, processing and application of hydrogels: A review. Mater Sci Eng C 2015; 57:414-33. doi: 10.1016/j.msec.2015.07.053 [Crossref] [ Google Scholar]
- Rodell CB, MacArthur Jr JW, Dorsey SM, Wade RJ, Wang LL, Woo YJ. Shear‐thinning supramolecular hydrogels with secondary autonomous covalent crosslinking to modulate viscoelastic properties in vivo. Adv Funct Mater 2015; 25:636-44. doi: 10.1002/adfm.201403550 [Crossref] [ Google Scholar]
- Lu HD, Soranno DE, Rodell CB, Kim IL, Burdick JA. Secondary photocrosslinking of injectable shear‐thinning dock‐and‐lock hydrogels. Adv Healthc Mater 2013; 2:1028-36. doi: 10.1002/adhm.201200343 [Crossref] [ Google Scholar]
- Wang LL, Sloand JN, Gaffey AC, Venkataraman CM, Wang Z, Trubelja A. Injectable, guest–host assembled polyethylenimine hydrogel for siRNA delivery. Biomacromolecules 2017; 18:77-86. doi: 10.1021/acs.biomac.6b01378.s001 [Crossref] [ Google Scholar]
- Li Y, Fan L, Liu S, Liu W, Zhang H, Zhou T. The promotion of bone regeneration through positive regulation of angiogenic–osteogenic coupling using microRNA-26a. Biomater 2013; 34:5048-58. doi: 10.1016/j.biomaterials.2013.03.052 [Crossref] [ Google Scholar]
- Chegini SP, Varshosaz J, Sadeghi HM, Dehghani A, Minaiyan M. Shear sensitive injectable hydrogels of cross-linked tragacanthic acid for ocular drug delivery: Rheological and biological evaluation. Int J Biol Macromol 2020; 165:2789-804. doi: 10.1016/j.ijbiomac.2020.10.164 [Crossref] [ Google Scholar]
- Lynch CR, Kondiah PP, Choonara YE, Du Toit LC, Ally N, Pillay V. Hydrogel biomaterials for application in ocular drug delivery. Front Bioeng Biotechnol 2020; 8:228. doi: 10.3389/fbioe.2020.00228 [Crossref] [ Google Scholar]
- Chen Q, Liu C, Gu Q, Qu D, Shi X, Zhang J. An injectable thermosensitive hydrogel encapsulating tetramethylpyrazine nanocrystals alleviates angiogenesis and apoptosis in a choroidal neovascularization mouse model. Appl Mater Today 2023; 33:101867. doi: 10.1016/j.apmt.2023.101867 [Crossref] [ Google Scholar]
- Xie B, Jin L, Luo Z, Yu J, Shi S, Zhang Z. An injectable thermosensitive polymeric hydrogel for sustained release of Avastin® to treat posterior segment disease. Int J Pharm 2015; 490:375-83. doi: 10.1016/j.ijpharm.2015.05.071 [Crossref] [ Google Scholar]
- Alejandra Quinteros D, Sebastian Lopez E, Lucio Silva Couto J, Angelica Maletto B, Alberto Allemandi D, Daniel Palma S. Evaluation of the performance of an ophthalmic thermosensitive hydrogel containing combination of suramin and bevacizumab. Curr Pharm Des 2016; 22:6587-94. [ Google Scholar]
- Rauck BM, Friberg TR, Mendez CAM, Park D, Shah V, Bilonick RA. Biocompatible reverse thermal gel sustains the release of intravitreal bevacizumab in vivoInvestig Ophthalmol. Vis Sci 2014; 55:469-76. doi: 10.1167/iovs.13-13120 [Crossref] [ Google Scholar]
- Han Q, Wang Y, Li X, Peng R, Li A, Qian Z. Effects of bevacizumab loaded PEG-PCL-PEG hydrogel intracameral application on intraocular pressure after glaucoma filtration surgery. J Mater Sci Mater Med 2015; 26:225. doi: 10.1007/s10856-015-5556-6 [Crossref] [ Google Scholar]
- Rong X, Ji Y, Zhu X, Yang J, Qian D, Mo X. Neuroprotective effect of insulin-loaded chitosan nanoparticles/PLGA-PEG-PLGA hydrogel on diabetic retinopathy in rats. Int J Nanomedicine 2019; 14:45. doi: 10.2147/IJN.S184574 [Crossref] [ Google Scholar]
- Mut AM, Vlaia L, Coneac G, Olariu I, Vlaia V, Stănciulescu C. Chitosan/HPMC-based hydrogels containing essential oils for topical delivery of fluconazole: Preliminary studies. Farmacia 2018; 66:248-56. [ Google Scholar]
- Abdollahi A, Malek-Khatabi A, Razavi MS, Sheikhi M, Abbaspour K, Rezagholi Z. The recent advancement in the chitosan-based thermosensitive hydrogel for tissue regeneration. J Drug Deliv Technol 2023; 86:104627. doi: 10.1016/j.jddst.2023.104627 [Crossref] [ Google Scholar]
- Jin X, Fu Q, Gu Z, Zhang Z, Lv H. Injectable corilagin/low molecular weight chitosan/PLGA-PEG-PLGA thermosensitive hydrogels for localized cancer therapy and promoting drug infiltration by modulation of tumor microenvironment. Int J Pharm 2020; 589:119772. [ Google Scholar]
- Makvandi P, Ali GW, Della Sala F, Abdel-Fattah WI, Borzacchiello A. Biosynthesis and characterization of antibacterial thermosensitive hydrogels based on corn silk extract, hyaluronic acid and nanosilver for potential wound healing. Carbohydr Polym 2019; 223:115023. [ Google Scholar]
- Deng H, Dong A, Song J, Chen X. Injectable thermosensitive hydrogel systems based on functional PEG/PCL block polymer for local drug delivery. Journal of Controlled Release 2019; 297:60-70. [ Google Scholar]
- Chung CK, García-Couce J, Campos Y, Kralisch D, Bierau K, Chan A. Doxorubicin loaded poloxamer thermosensitive hydrogels: chemical, pharmacological and biological evaluation. Molecules 2020; 25(9):2219. [ Google Scholar]
- Cui S, Yu L, Ding J. Thermogelling of amphiphilic block copolymers in water: ABA type versus AB or BAB type. Macromolecules 2019; 52(10):3697-715. [ Google Scholar]
- Rahmani F, Atabaki R, Behrouzi S, Mohamadpour F, Kamali H. The recent advancement in the PLGA-based thermo-sensitive hydrogel for smart drug delivery. Int J Pharm 2022; 631:122484. doi: 10.1016/j.ijpharm.2022.122484 [Crossref] [ Google Scholar]
- Xu K, An N, Zhang H, Zhang Q, Zhang K, Hu X. Sustained-release of PDGF from PLGA microsphere embedded thermo-sensitive hydrogel promoting wound healing by inhibiting autophagy. J Drug Deliv Sci Technol 2020; 55:101405. [ Google Scholar]
- Marques SM, Kumar L. PKPD of PLGA-PEG-PLGA Copolymeric Micelles. Pharmacokinetics and Pharmacodynamics of Nanoparticulate Drug Delivery Systems. Springer; 2022. p. 273-92.
- Xi Y, Wang W, Ma L, Xu N, Shi C, Xu G. Alendronate modified mPEG-PLGA nano-micelle drug delivery system loaded with astragaloside has anti-osteoporotic effect in rats. Drug Deliv 2022; 29:2386-402. doi: 10.1080/10717544.2022.2086942 [Crossref] [ Google Scholar]
- Torchilin VP. Targeted pharmaceutical nanocarriers for cancer therapy and imaging. AAPS J 2007; 9:E128-E47. doi: 10.1208/aapsj0902015 [Crossref] [ Google Scholar]
- Torchilin VP. Nanocarriers. Pharm Res 2007; 24:2333-4. doi: 10.1007/s11095-006-9132-0 [Crossref] [ Google Scholar]
- Wadhwa S, Paliwal R, Paliwal SR, Vyas S. Nanocarriers in ocular drug delivery: an update review. Curr Pharm Des 2009; 15:2724-50. doi: 10.2174/138161209788923886 [Crossref] [ Google Scholar]
- Salehipour M, Rezaei S, Rezaei M, Yazdani M, Mogharabi-Manzari M. Opportunities and Challenges in Biomedical Applications of Metal–Organic Frameworks. J Inorg Organomet Polym Mater 2021; 1:1-20. doi: 10.1007/s10904-021-02118-7 [Crossref] [ Google Scholar]
- Sun T, Zhang YS, Pang B, Hyun DC, Yang M, Xia Y. Engineered nanoparticles for drug delivery in cancer therapy. Angew Chem Int Ed 2014; 53:12320-64. doi: 10.1002/anie.201403036 [Crossref] [ Google Scholar]
- Bhasarkar JB, Bal DK. Nanomaterial-based advanced oxidation processes for degradation of waste pollutants. Handbook of Nanomaterials for Wastewater Treatment. Elsevier; 2021. p. 811-31. 10.1016/b978-0-12-821496-1.00003-9.
- Kimura H, Ogura Y. Biodegradable polymers for ocular drug delivery. Ophthalmologica 2001; 215:143-55. doi: 10.1159/000050849 [Crossref] [ Google Scholar]
- Lu Y, Zhou N, Huang X, Cheng J-W, Li F-Q, Wei R-L. Effect of intravitreal injection of bevacizumab-chitosan nanoparticles on retina of diabetic rats. Int J Ophthalmol 2014; 7:1. doi: 10.1111/j.1442-9071.2010.02370.x [Crossref] [ Google Scholar]
- Elsaid N, Jackson TL, Elsaid Z, Alqathama A, Somavarapu S. PLGA microparticles entrapping chitosan-based nanoparticles for the ocular delivery of ranibizumab. Mol Pharmaceutics 2016; 13:2923-40. doi: 10.1021/acs.molpharmaceut.6b00335 [Crossref] [ Google Scholar]
- Mahaling B, Srinivasarao DA, Raghu G, Kasam RK, Reddy GB, Katti DS. A non-invasive nanoparticle mediated delivery of triamcinolone acetonide ameliorates diabetic retinopathy in rats. Nanoscale 2018; 10:16485-98. doi: 10.1039/C8NR00058A [Crossref] [ Google Scholar]
- Chegini SP, Varshosaz J, Sadeghi HM, Dehghani A, Minayian M. Poly (glycerol sebacate) nanoparticles for ocular delivery of sunitinib: physicochemical, cytotoxic and allergic studies. IET Nanobiotechnol 2019; 13:974-82. doi: 10.1049/iet-nbt.2019.0002 [Crossref] [ Google Scholar]
- Cho H, Kambhampati SP, Lai MJ, Zhou L, Lee G, Xie Y. Dendrimer‐Triamcinolone Acetonide Reduces Neuroinflammation, Pathological Angiogenesis, and Neuroretinal Dysfunction in Ischemic Retinopathy. Adv Ther 2021; 4:2000181. doi: 10.1002/adtp.202000181 [Crossref] [ Google Scholar]
- Xu J, Wang Y, Li Y, Yang X, Zhang P, Hou H. Inhibitory efficacy of intravitreal dexamethasone acetate-loaded PLGA nanoparticles on choroidal neovascularization in a laser-induced rat model. J Ocul Pharmacol Ther 2007; 23:527-40. doi: 10.1089/jop.2007.0002 [Crossref] [ Google Scholar]
- Du S, Wang H, Jiang F, Wang Y. Diabetic Retinopathy Analysis—Effects of Nanoparticle-Based Triamcinolone. J Nanosci Nanotechnol 2020; 20:6111-5. doi: 10.1166/jnn.2020.18569 [Crossref] [ Google Scholar]
- Du S, Wang H, Jiang F, Wang Y. Diabetic Retinopathy Analysis Effects of Nanoparticle-Based Triamcinolone. J Nanosci Nanotechnol 2020; 20:6111-5. doi: 10.1166/jnn.2020.18569 [Crossref] [ Google Scholar]
- Malakouti-Nejad M, Bardania H, Aliakbari F, Baradaran-Rafii A, Elahi E, Monti D. Formulation of nanoliposome-encapsulated bevacizumab (Avastin): Statistical optimization for enhanced drug encapsulation and properties evaluation. Int J Pharm 2020; 590:119895. doi: 10.1016/j.ijpharm.2020.119895 [Crossref] [ Google Scholar]
- Abrishami M, Ganavati SZ, Soroush D, Rouhbakhsh M, Jaafari MR, Malaekeh-Nikouei B. Preparation, characterization, and in vivo evaluation of nanoliposomes-encapsulated bevacizumab (avastin) for intravitreal administration. Retina 2009; 29:699-703. doi: 10.1097/iae.0b013e3181a2f42a [Crossref] [ Google Scholar]
- Varshochian R, Riazi‐Esfahani M, Jeddi‐Tehrani M, Mahmoudi AR, Aghazadeh S, Mahbod M. Albuminated PLGA nanoparticles containing bevacizumab intended for ocular neovascularization treatment. J Biomed Mater Res A 2015; 103:3148-56. doi: 10.1002/jbm.a.35446 [Crossref] [ Google Scholar]
- Dong Y, Wan G, Yan P, Qian C, Li F, Peng G. Fabrication of resveratrol coated gold nanoparticles and investigation of their effect on diabetic retinopathy in streptozotocin induced diabetic rats. J Photochem Photobiol B Biol 2019; 195:51-7. doi: 10.1016/j.jphotobiol.2019.04.012 [Crossref] [ Google Scholar]
- Zhang L, Chu W, Zheng L, Li J, Ren Y, Xue L, et al. Zinc oxide nanoparticles from Cyperus rotundus attenuates diabetic retinopathy by inhibiting NLRP3 inflammasome activation in STZ‐induced diabetic rats. J Biochem Mol Toxicol 2020; 34; e22583. 10.1002/jbt.22583.
- Amato R, Giannaccini M, Dal Monte M, Cammalleri M, Pini A, Raffa V. Association of the Somatostatin Analog Octreotide With Magnetic Nanoparticles for Intraocular Delivery: A Possible Approach for the Treatment of Diabetic Retinopathy. Front Bioeng Biotechnol 2020; 8:144. doi: 10.3389/fbioe.2020.00144.eCollection2020 [Crossref] [ Google Scholar]
- Islam MA, Jacob MV, Antunes E. A critical review on silver nanoparticles: From synthesis and applications to its mitigation through low-cost adsorption by biochar. J Environ Manage 2021; 281:111918. doi: 10.1016/j.jenvman.2020.111918 [Crossref] [ Google Scholar]
- Mohammadpour M, Jabbarvand M, Delrish E, Khoshzaban A. Antiangiogenic effect of silicate nanoparticles on corneal neo-vascularisation induced by vascular endothelial growth factor. J Medical Hypotheses Ideas 2014; 8:14-20. doi: 10.1016/j.jmhi.2013.06.003 [Crossref] [ Google Scholar]
- Nedzvetsky V, Prischepe I, Agca G, Tykhomyrov A, Kyrychenko S. C60 fullerene nanoparticles prevent diabetic retinopathy complications. Bulletin of Lviv University Biological series. 2016; 171-6.
- Kumari A, Yadav SK, Yadav SC. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf B 2010; 75:1-18. doi: 10.1016/j.colsurfb.2009.09.001 [Crossref] [ Google Scholar]
- Nekolova J, Kremlacek J, Lukavsky J, Sikl R, Sin M, Langrova J. The intraocular implant and visual rehabilitation improve the quality of life of elderly patients with geographic atrophy secondary to age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 2023; 261:263-72. doi: 10.1007/s00417-022-05803-6 [Crossref] [ Google Scholar]
- Jiang H, Xu Z. Hyaluronic acid-based nanoparticles to deliver drugs to the ocular posterior segment. Drug Deliv 2023; 30:2204206. doi: 10.1080/10717544.2023.2204206 [Crossref] [ Google Scholar]
- Taheri SL, Rezazadeh M, Hassanzadeh F, Akbari V, Dehghani A, Talebi A. Preparation, physicochemical, and retinal anti-angiogenic evaluation of poloxamer hydrogel containing dexamethasone/avastin-loaded chitosan-N-acetyl-L-cysteine nanoparticles. Int J Biol Macromol 2022; 220:1605-18. doi: 10.1016/j.ijbiomac.2022.09.101 [Crossref] [ Google Scholar]
- Paolicelli P, de la Fuente M, Sánchez A, Seijo B, Alonso MJ. Chitosan nanoparticles for drug delivery to the eye. Expert Opin Drug Deliv 2009; 6:239-53. doi: 10.1517/17425240902762818 [Crossref] [ Google Scholar]
- De Campos AM, Diebold Y, Carvalho EL, Sánchez A, Alonso MJ. Chitosan nanoparticles as new ocular drug delivery systems: in vitro stability, in vivo fate, and cellular toxicity. Pharm Res 2004; 21:803-10. doi: 10.1023/b:pham.0000026432.75781.cb [Crossref] [ Google Scholar]
- Nagarwal RC, Singh PN, Kant S, Maiti P, Pandit JK. Chitosan nanoparticles of 5-fluorouracil for ophthalmic delivery: characterization, in-vitro and in-vivo study. Chem Pharm Bull 2011; 59:272-8. doi: 10.1248/cpb.59.272 [Crossref] [ Google Scholar]
- Prashanth KH, Tharanathan R. Depolymerized products of chitosan as potent inhibitors of tumor-induced angiogenesis. Biochim Biophys Acta Gen Subj 2005; 1722:22-9. doi: 10.1016/j.bbagen.2004.11.009 [Crossref] [ Google Scholar]
- Xu Y, Wen Z, Xu Z. Chitosan nanoparticles inhibit the growth of human hepatocellular carcinoma xenografts through an antiangiogenic mechanism. Anticancer Res 2009; 29:5103-9. [ Google Scholar]
- Okamoto H, Umeda S, Obazawa M, Minami M, Noda T, Mizota A. Complement factor H polymorphisms in Japanese population with age-related macular degeneration. Mol Vis 2006; 12:156-8. doi: 10.1016/j.molimm.2008.08.095 [Crossref] [ Google Scholar]
- Kim M-M, Kim S-K. Chitooligosaccharides inhibit activation and expression of matrix metalloproteinase‐2 in human dermal fibroblasts. FEBS Lett 2006; 580:2661-6. doi: 10.1016/j.febslet.2006.04.015 [Crossref] [ Google Scholar]
- Liu H-t, Huang P, Ma P, Liu Q-s, Yu C, Du Y-g. Chitosan oligosaccharides suppress LPS-induced IL-8 expression in human umbilical vein endothelial cells through blockade of p38 and Akt protein kinases. Acta Pharmacol Sin 2011; 32:478. doi: 10.1038/aps.2011.10 [Crossref] [ Google Scholar]
- Bernkop-Schnürch A, Hornof M, Guggi D. Thiolated chitosans. Eur J Pharm Biopharm 2004; 57:9-17. doi: 10.1016/s0939-6411(03)00147-4 [Crossref] [ Google Scholar]
- Aluigi MG, De SF, D'Agostini F, Albini A, Fassina G. Antiapoptotic and antigenotoxic effects of N-acetylcysteine in human cells of endothelial origin. Anticancer Res 2000; 20:3183-7. [ Google Scholar]
- Zhu Y, Zhang X-L, Zhu B-F, Ding Y-N. Effect of antioxidant N-acetylcysteine on diabetic retinopathy and expression of VEGF and ICAM-1 from retinal blood vessels of diabetic rats. Mol Biol Rep 2012; 39:3727-35. doi: 10.1007/s11033-011-1148-9 [Crossref] [ Google Scholar]
- Bhattacharjee A, Das PJ, Adhikari P, Marbaniang D, Pal P, Ray S. Novel drug delivery systems for ocular therapy: With special reference to liposomal ocular delivery. Eur J Ophthalmol 2019; 29:113-26. doi: 10.1177/1120672118769776 [Crossref] [ Google Scholar]
- Aguilar-Pérez K, Avilés-Castrillo J, Medina DI, Parra-Saldivar R, Iqbal H. Insight into nanoliposomes as smart nanocarriers for greening the twenty-first century biomedical settings. Front Bioeng Biotechnol 2020; 8:1441. doi: 10.3389/fbioe.2020.579536 [Crossref] [ Google Scholar]
- Taheri SL, Varshosaz J. Enhancement of the anti-microbial activity of Mentha spicata essential oil on storage by glycerosomes. Food Sci Biotechnol 2023; 32:2145-2152. doi: 10.1007/s10068-023-01301-5 [Crossref] [ Google Scholar]
- Al-Amin M, Mastrotto F, Subrizi A, Sen M, Turunen T, Arango-Gonzalez B. Tailoring surface properties of liposomes for dexamethasone intraocular administration. J Control Release 2023; 354:323-36. doi: 10.1016/j.jconrel.2023.01.027 [Crossref] [ Google Scholar]
- Christensen G, Urimi D, Lorenzo‐Soler L, Schipper N, Paquet-Durand F. Ocular permeability, intraocular biodistribution of lipid nanocapsule formulation intended for retinal drug delivery. Eur J Pharm Biopharm 2023; 187:175-83. doi: 10.1016/j.ejpb.2023.04.012 [Crossref] [ Google Scholar]
- Bouyer E, Mekhloufi G, Rosilio V, Grossiord J-L, Agnely F. Proteins, polysaccharides, and their complexes used as stabilizers for emulsions: alternatives to synthetic surfactants in the pharmaceutical field? IntJ. Pharm 2012; 436:359-78. doi: 10.1016/j.ijpharm.2012.06.052 [Crossref] [ Google Scholar]
- Matsushita S, Isima Y, Chuang VTG, Watanabe H, Tanase S, Maruyama T. Functional analysis of recombinant human serum albumin domains for pharmaceutical applications. Pharm Res 2004; 21:1924-32. doi: 10.1023/b:pham.0000045248.03337.0e [Crossref] [ Google Scholar]
- Hassanin I, Elzoghby A. Albumin-based nanoparticles: a promising strategy to overcome cancer drug resistance. Cancer Drug Resist 2020; 3:930-46. doi: 10.20517/cdr.2020.68 [Crossref] [ Google Scholar]
- Hawe A, Friess W. Physicohemical characterization of the freezing behavior of mannitol-human serum albumin formulations. AAPS PharmSciTech 2006; 7:E85-E94. doi: 10.1208/pt070494 [Crossref] [ Google Scholar]
- Christiansen C, Kryvi H, Sontum P, Skotland T. Physical and biochemical characterization of Albunex, a new ultrasound contrast agent consisting of air‐filled albumin microspheres suspended in a solution of human albumin. Biotechnol Appl Biochem 1994; 19:307-20. [ Google Scholar]
- Ibrahim NK, Desai N, Legha S, Soon-Shiong P, Theriault RL, Rivera E. Phase I and pharmacokinetic study of ABI-007, a Cremophor-free, protein-stabilized, nanoparticle formulation of paclitaxel. Clin Cancer Res 2002; 8:1038-44. [ Google Scholar]
- de Redín IL, Boiero C, Martínez-Ohárriz MC, Agüeros M, Ramos R, Peñuelas I. Human serum albumin nanoparticles for ocular delivery of bevacizumab. Int J Pharm 2018; 541:214-23. doi: 10.1016/j.ijpharm.2018.02.003 [Crossref] [ Google Scholar]
- Gopinath SC. Nanoparticles in Analytical and Medical Devices. Elsevier; 2020.
- Amina SJ, Guo B. A review on the synthesis and functionalization of gold nanoparticles as a drug delivery vehicle. Int J Nanomedicine 2020; 15:9823. doi: 10.2147/IJN.S279094 [Crossref] [ Google Scholar]
- Santhoshkumar J, Rajeshkumar S, Kumar SV. Phyto-assisted synthesis, characterization and applications of gold nanoparticles–A review. Biochem Biophys Rep 2017; 11:46-57. doi: 10.1016/j.bbrep.2017.06.004 [Crossref] [ Google Scholar]
- Li N, Zhao P, Astruc D. Anisotropic gold nanoparticles: synthesis, properties, applications, and toxicity. Angew Chem Int Ed 2014; 53:1756-89. doi: 10.1002/anie.201300441 [Crossref] [ Google Scholar]
- Daraee H, Eatemadi A, Abbasi E, Fekri Aval S, Kouhi M, Akbarzadeh A. Application of gold nanoparticles in biomedical and drug delivery. Artif Cells Nanomed Biotechnol 2016; 44:410-22. doi: 10.3109/21691401.2014.955107 [Crossref] [ Google Scholar]
- Eissa NG, Eldehna WM, Abdelazim EB, Eissa RA, Mohamed HH, Diab NH. Morphologic Design of Nanogold Carriers for a Carbonic Anhydrase Inhibitor: Effect on Ocular Retention and Intraocular Pressure. Int J Pharm 2023; 642:123161. doi: 10.1016/j.ijpharm.2023.123161 [Crossref] [ Google Scholar]
- Pan Y, Wu Q, Qin L, Cai J, Du B. Gold nanoparticles inhibit VEGF165-induced migration and tube formation of endothelial cells via the Akt pathway. Biomed Res Int 2014; 2014:418624. doi: 10.1155/2014/418624 [Crossref] [ Google Scholar]
- Pan Y, Ding H, Qin L, Zhao X, Cai J, Du B. Gold nanoparticles induce nanostructural reorganization of VEGFR2 to repress angiogenesis. J Biomed Nanotech 2013; 9:1746-56. doi: 10.1166/jbn.2013.1678 [Crossref] [ Google Scholar]
- Shen N, Zhang R, Zhang H-R, Luo H-Y, Shen W, Gao X. Inhibition of retinal angiogenesis by gold nanoparticles via inducing autophagy. Int J Ophthalmol 2018; 11:1269. doi: 10.18240/ijo.2018.08.04 [Crossref] [ Google Scholar]
- Du J, Teng R-J, Guan T, Eis A, Kaul S, Konduri GG. Role of autophagy in angiogenesis in aortic endothelial cellsAmJPhysiol. Cell Physiol 2012; 302:C383-C91. doi: 10.1152/ajpcell.00164.2011 [Crossref] [ Google Scholar]
- Duan J, Yu Y, Yu Y, Li Y, Huang P, Zhou X. Silica nanoparticles enhance autophagic activity, disturb endothelial cell homeostasis and impair angiogenesis. Part Fibre Toxicol 2014; 11:1-13. doi: 10.1186/s12989-014-0050-8 [Crossref] [ Google Scholar]
- Wu Q, Jin R, Feng T, Liu L, Yang L, Tao Y. Iron oxide nanoparticles and induced autophagy in human monocytes. Int J Nanomedicine 2017; 12:3993. doi: 10.2147/IJN.S135189 [Crossref] [ Google Scholar]
- Mizushima N, Levine B, Cuervo AM, Klionsky DJ. Autophagy fights disease through cellular self-digestion. Nature 2008; 451:1069-75. doi: 10.1038/nature06639 [Crossref] [ Google Scholar]
- Kim JH, Kim MH, Jo DH, Yu YS, Lee TG, Kim JH. The inhibition of retinal neovascularization by gold nanoparticles via suppression of VEGFR-2 activation. Biomaterials 2011; 32:1865-71. doi: 10.1016/j.biomaterials.2010.11.030 [Crossref] [ Google Scholar]
- Buzea C, Pacheco II, Robbie K. Nanomaterials and nanoparticles: sources and toxicity. Biointerphases 2007; 2:MR17-MR71. doi: 10.1116/1.2815690 [Crossref] [ Google Scholar]
- Rikken RS, Nolte RJ, Maan JC, van Hest JC, Wilson DA, Christianen PC. Manipulation of micro-and nanostructure motion with magnetic fields. Soft Matter 2014; 10:1295-308. doi: 10.1039/C3SM52294F [Crossref] [ Google Scholar]
- Ali A, Shah T, Ullah R, Zhou P, Guo M, Ovais M. Review on recent progress in magnetic nanoparticles: Synthesis, characterization, and diverse applications. Front Chem 2021; 9:629054. doi: 10.3389/fchem.2021.629054 [Crossref] [ Google Scholar]
- Giannaccini M, Calatayud MP, Poggetti A, Corbianco S, Novelli M, Paoli M. Magnetic nanoparticles for efficient delivery of growth factors: stimulation of peripheral nerve regeneration. Adv Healthc Mater 2017; 6:1601429. doi: 10.1002/adhm.201601429 [Crossref] [ Google Scholar]
- Giannaccini M, Giannini M, Calatayud MP, Goya GF, Cuschieri A, Dente L. Magnetic nanoparticles as intraocular drug delivery system to target retinal pigmented epithelium (RPE). Int J Mol Sci 2014; 15:1590-605. doi: 10.3390/ijms15011590 [Crossref] [ Google Scholar]
- Raju HB, Hu Y, Vedula A, Dubovy SR, Goldberg JL. Evaluation of magnetic micro-and nanoparticle toxicity to ocular tissues. PLoS One 2011; 6:e17452. doi: 10.1371/journal.pone.0017452 [Crossref] [ Google Scholar]
- Oves M, Aslam M, Rauf MA, Qayyum S, Qari HA, Khan MS. Antimicrobial and anticancer activities of silver nanoparticles synthesized from the root hair extract of Phoenix dactylifera. Mater Sci Eng C 2018; 89:429-43. doi: 10.1016/j.msec.2018.03.035 [Crossref] [ Google Scholar]
- Kowshik M, Ashtaputre S, Kharrazi S, Vogel W, Urban J, Kulkarni SK. Extracellular synthesis of silver nanoparticles by a silver-tolerant yeast strain MKY3. Nanotechnology 2002; 14:95. doi: 10.1088/0957-4484/14/1/321 [Crossref] [ Google Scholar]
- Cho K-H, Park J-E, Osaka T, Park S-G. The study of antimicrobial activity and preservative effects of nanosilver ingredient. Electrochim Acta 2005; 51:956-60. doi: 10.1016/j.electacta.2005.04.071 [Crossref] [ Google Scholar]
- Mukherjee P, Ahmad A, Mandal D, Senapati S, Sainkar SR, Khan MI. Fungus-mediated synthesis of silver nanoparticles and their immobilization in the mycelial matrix: a novel biological approach to nanoparticle synthesis. Nano Lett 2001; 1:515-9. doi: 10.1021/nl0155274 [Crossref] [ Google Scholar]
- Durán N, Marcato PD, Alves OL, De Souza GI, Esposito E. Mechanistic aspects of biosynthesis of silver nanoparticles by several Fusarium oxysporum strains. J Nanobiotechnology 2005; 3:1-7. doi: 10.1186/1477-3155-3-8 [Crossref] [ Google Scholar]
- Alt V, Bechert T, Steinrücke P, Wagener M, Seidel P, Dingeldein E. An in vitro assessment of the antibacterial properties and cytotoxicity of nanoparticulate silver bone cement. Biomaterials 2004; 25:4383-91. doi: 10.1016/j.biomaterials.2003.10.078 [Crossref] [ Google Scholar]
- Kim K-J, Sung WS, Suh BK, Moon S-K, Choi J-S, Kim JG. Antifungal activity and mode of action of silver nano-particles on Candida albicans. Biometals 2009; 22:235-42. doi: 10.1007/s10534-008-9159-2 [Crossref] [ Google Scholar]
- Nadworny PL, Wang J, Tredget EE, Burrell RE. Anti-inflammatory activity of nanocrystalline silver in a porcine contact dermatitis model. Nanomed Nanotechnol Biol Med 2008; 4:241-51. doi: 10.1016/j.nano.2008.04.006 [Crossref] [ Google Scholar]
- Chen Y, Su F, Su B. Application of silver nanoparticles in the treatment of central serous chorioretinopathy. Mater Express 2023; 13:274-82. doi: 10.3390/molecules28124701 [Crossref] [ Google Scholar]
- Gurunathan S, Lee K-J, Kalishwaralal K, Sheikpranbabu S, Vaidyanathan R, Eom SH. Antiangiogenic properties of silver nanoparticles. Biomaterials 2009; 30:6341-50. doi: 10.1016/j.biomaterials.2009.08.008 [Crossref] [ Google Scholar]
- Erickson KK, Sundstrom JM, Antonetti DA. Vascular permeability in ocular disease and the role of tight junctions. Angiogenesis 2007; 10:103. doi: 10.1007/s10456-007-9067-z [Crossref] [ Google Scholar]
- Sheikpranbabu S, Kalishwaralal K, Lee K-j, Vaidyanathan R, Eom SH, Gurunathan S. The inhibition of advanced glycation end-products-induced retinal vascular permeability by silver nanoparticles. Biomaterials 2010; 31:2260-71. doi: 10.1016/j.biomaterials.2009.11.076 [Crossref] [ Google Scholar]
- Sheikpranbabu S, Kalishwaralal K, Venkataraman D, Eom SH, Park J, Gurunathan S. Silver nanoparticles inhibit VEGF-and IL-1β-induced vascular permeability via Src dependent pathway in porcine retinal endothelial cells. J Nanobiotechnology 2009; 7:8. doi: 10.1186/1477-3155-7-8 [Crossref] [ Google Scholar]
- Kalishwaralal K, Banumathi E, Pandian SRK, Deepak V, Muniyandi J, Eom SH. Silver nanoparticles inhibit VEGF induced cell proliferation and migration in bovine retinal endothelial cells. Colloids Surf B 2009; 73:51-7. doi: 10.1016/j.colsurfb.2009.04.025 [Crossref] [ Google Scholar]
- Abbasi M, Ghoran SH, Niakan MH, Jamali K, Moeini Z, Jangjou A, et al. Mesoporous silica nanoparticle: Heralding a brighter future in cancer nanomedicine. Microporous Mesoporous Mater 2021; 110967. 10.1016/j.micromeso.2021.110967.
- Selvarajan V, Obuobi S, Ee PLR. Silica Nanoparticles—A Versatile Tool for the Treatment of Bacterial Infections. Front Chem 2020; 8:602. doi: 10.3389/fchem.2020.00602 [Crossref] [ Google Scholar]
- Sekhon BS, Kamboj SR. Inorganic nanomedicine—part 2. Nanomed Nanotechnol Biol Med 2010; 6:612-8. doi: 10.1016/j.nano.2010.04.003 [Crossref] [ Google Scholar]
- Couleaud P, Morosini V, Frochot C, Richeter S, Raehm L, Durand J-O. Silica-based nanoparticles for photodynamic therapy applications. Nanoscale 2010; 2:1083-95. doi: 10.1039/c0nr00096e [Crossref] [ Google Scholar]
- Hong S-S, Lee MS, Park SS, Lee G-D. Synthesis of nanosized TiO2/SiO2 particles in the microemulsion and their photocatalytic activity on the decomposition of p-nitrophenol. Catal Today 2003; 87:99-105. doi: 10.1016/j.cattod.2003.10.012 [Crossref] [ Google Scholar]
- Jo DH, Kim JH, Yu YS, Lee TG, Kim JH. Antiangiogenic effect of silicate nanoparticle on retinal neovascularization induced by vascular endothelial growth factor. Nanomed Nanotechnol Biol Med 2012; 8:784-91. doi: 10.1016/j.nano.2011.09.003 [Crossref] [ Google Scholar]
- Ren H, Zhu C, Li Z, Yang W, Song E. Emodin-loaded magnesium silicate hollow nanocarriers for anti-angiogenesis treatment through inhibiting VEGF. Int J Mol Sci 2014; 15:16936-48. doi: 10.3390/ijms150916936 [Crossref] [ Google Scholar]
- Mishra PK, Mishra H, Ekielski A, Talegaonkar S, Vaidya B. Zinc oxide nanoparticles: a promising nanomaterial for biomedical applications. Drug Discov Today 2017; 22:1825-34. doi: 10.1016/j.drudis.2017.08.006 [Crossref] [ Google Scholar]
- San Tang K. San Tang KThe current and future perspectives of zinc oxide nanoparticles in the treatment of diabetes mellitus. Life Sci 2019; 239:117011. doi: 10.1016/j.lfs.2019.117011 [Crossref] [ Google Scholar]
- Rajesh K, Umar A, Kumar G. Antimicrobial properties of ZnO nanomaterialsA review. Ceramics International 2017; 43:3940-61. doi: 10.1016/j.ceramint.2016.12.062 [Crossref] [ Google Scholar]
- Colon G, Ward BC, Webster TJ. Increased osteoblast and decreased Staphylococcus epidermidis functions on nanophase ZnO and TiO2. J Biomed Mater Res A 2006; 78:595-604. doi: 10.1002/jbm.a.30789 [Crossref] [ Google Scholar]
- Singh A, Singh Ná, Afzal S, Singh T, Hussain I. Zinc oxide nanoparticles: a review of their biological synthesis, antimicrobial activity, uptake, translocation and biotransformation in plants. J Mater Sci 2018; 53:185-201. doi: 10.1007/s10853-017-1544-1 [Crossref] [ Google Scholar]
- Kroto HW, Heath JR, O’Brien SC, Curl RF, Smalley RE. C 60: buckminsterfullerene. Nature 1985; 318:162-3. [ Google Scholar]
- Baati T, Bourasset F, Gharbi N, Njim L, Abderrabba M, Kerkeni A. The prolongation of the lifespan of rats by repeated oral administration of [60] fullerene. Biomaterials 2012; 33:4936-46. doi: 10.1016/j.biomaterials.2012.03.036 [Crossref] [ Google Scholar]
- Astefanei A, Núñez O, Galceran MT. Characterisation and determination of fullerenes: a critical review. Anal Chim Acta 2015; 882:1-21. doi: 10.1016/j.aca.2015.03.025 [Crossref] [ Google Scholar]
- Kerna N, Flores J, Pruitt K, Nwokorie U, Holets H. The Application of Fullerene Derivatives in Medicine and Specific Endocri-nological Conditions. EC Endocrinology and Metabolic Research 2020; 5:56-66. doi: 10.31080/ecemr.2020.05.00229 [Crossref] [ Google Scholar]
- Krusic P, Wasserman E, Keizer P, Morton J, Preston K. Radical reactions of C60. Science 1991; 254:1183-5. doi: 10.1126/science.254.5035.1183 [Crossref] [ Google Scholar]
- Nedzvetsky V, Sukharenko E, Baydas G, Andrievsky G. Water-soluble C60 fullerene ameliorates astroglial reactivity and TNFa production in retina of diabetic rats. Regulatory Mechanisms in Biosystems 2019; 10:513-9. [ Google Scholar]
- Alvarez-Lorenzo C, Hiratani H, Concheiro A. Contact lenses for drug delivery. Am J Drug Deliv Ther 2006; 4:131-51. doi: 10.2165/00137696-200604030-00002 [Crossref] [ Google Scholar]
- McAvoy K, Jones D, Thakur RRS. Synthesis and characterisation of photocrosslinked poly (ethylene glycol) diacrylate implants for sustained ocular drug delivery. Pharm Res 2018; 35:1-17. doi: 10.1007/s11095-017-2298-9 [Crossref] [ Google Scholar]
- Sapino S, Chirio D, Peira E, Abellán Rubio E, Brunella V, Jadhav SA. Ocular drug delivery: a special focus on the thermosensitive approach. Nanomaterials 2019; 9:884. doi: 10.3390/nano9060884 [Crossref] [ Google Scholar]
- Kim HM, Woo SJ. Ocular Drug Delivery to the Retina: Current Innovations and Future Perspectives. Pharmaceutics 2021; 13:108. doi: 10.3390/pharmaceutics13010108 [Crossref] [ Google Scholar]
- González-Chomón C, Concheiro A, Alvarez-Lorenzo C. Soft contact lenses for controlled ocular delivery: 50 years in the making. Ther Deliv 2013; 4:1141-61. doi: 10.4155/tde.13.81 [Crossref] [ Google Scholar]
- Dugel PU, Bandello F, Loewenstein A. Dexamethasone intravitreal implant in the treatment of diabetic macular edema. Clin Ophthalmol 2015; 9:1321. doi: 10.2147/OPTH.S79948 [Crossref] [ Google Scholar]
- Takmakov P, Ruda K, Phillips KS, Isayeva IS, Krauthamer V, Welle CG. Rapid evaluation of the durability of cortical neural implants using accelerated aging with reactive oxygen species. J Neural Eng 2015; 12:026003. doi: 10.1088/1741-2560/12/2/026003 [Crossref] [ Google Scholar]
- Huang Y, Van Dessel J, Martens W, Lambrichts I, Zhong W-J, Ma G-W. Sensory innervation around immediately vsdelayed loaded implants: a pilot study. Int J Oral Sci 2015; 7:49-55. doi: 10.1038/ijos.2014.53 [Crossref] [ Google Scholar]
- Bagga C, Erbe EM, Murphy JP, Freid JM, Pomrink GJ. Bioactive spinal implants and method of manufacture thereof. Google Patents; 2014.
- Oka Y, Ye M, Zuker CS. Thirst driving and suppressing signals encoded by distinct neural populations in the brain. Nature 2015; 520:349-52. doi: 10.1038/nature14108 [Crossref] [ Google Scholar]
- Guo R, Merkel A, Sterling J, Davidson J, Guelcher S. Substrate modulus of 3D-printed scaffolds regulates the regenerative response in subcutaneous implants through the macrophage phenotype and Wnt signaling. Biomaterials 2015; 73:85-95. doi: 10.1016/j.biomaterials.2015.09.005 [Crossref] [ Google Scholar]
- Klein MO, Schiegnitz E, Al-Nawas B. Systematic review on success of narrow-diameter dental implants. Int J Oral Maxillofac Implants 2014; 43:29. doi: 10.11607/jomi.2014suppl.g1.3 [Crossref] [ Google Scholar]
- Yahyavi-Firouz-Abadi N, Menias CO, Bhalla S, Siegel C, Gayer G, Katz DS. Imaging of cosmetic plastic procedures and implants in the body and their potential complications. Am J Roentgenol 2015; 204:707-15. doi: 10.2214/AJR.14.13516 [Crossref] [ Google Scholar]
- Prakasam M, Locs J, Salma-Ancane K, Loca D, Largeteau A, Berzina-Cimdina L. Biodegradable materials and metallic implants—a review. J Funct Biomater 2017; 8:44. doi: 10.3390/jfb8040044 [Crossref] [ Google Scholar]
- Fialho SL, Silva Cunha Ad. Manufacturing techniques of biodegradable implants intended for intraocular application. Drug Deliv 2005; 12:109-16. doi: 10.1080/10717540590921432 [Crossref] [ Google Scholar]
- Durgun ME, Güngör S, Özsoy Y. Micelles: promising ocular drug carriers for anterior and posterior segment diseases. J Ocul Pharmacol Ther 2020; 36:323-41. doi: 10.1089/jop.2019.0109 [Crossref] [ Google Scholar]
- Stewart SA, Domínguez-Robles J, Donnelly RF, Larrañeta E. Implantable polymeric drug delivery devices: classification, manufacture, materials, and clinical applications. Polymers 2018; 10:1379. doi: 10.3390/polym10121379 [Crossref] [ Google Scholar]
- Tamaddon L, Dorkoosh SMF, Karkhaneh R, Riazi-Esfahani M, Rafiee-Tehrani M. A novel design of a biodegradable intravitreal implant for the sustained release of clindamycin. Res Pharm Sci 2012; 7:308. [ Google Scholar]
- Shastri VP. Non-degradable biocompatible polymers in medicine: past, present and future. Curr Pharm Biotechnol 2003; 4:331-7. doi: 10.2174/1389201033489694 [Crossref] [ Google Scholar]
- Colaris MJ, de Boer M, van der Hulst RR, Tervaert JWC. Two hundreds cases of ASIA syndrome following silicone implants: a comparative study of 30 years and a review of current literature. Immunol Res 2017; 65:120-8. doi: 10.1007/s12026-016-8821-y [Crossref] [ Google Scholar]
- Tamaddon L, Mostafavi SA, Karkhane R, Riazi-Esfahani M, Dorkoosh FA, Rafiee-Tehrani M. Design and development of intraocular polymeric implant systems for long-term controlled-release of clindamycin phosphate for toxoplasmic retinochoroiditis. Adv Biomed Res 2015; 4:32. doi: 10.4103/2277-9175.150426 [Crossref] [ Google Scholar]
- Tamaddon L, Mostafavi A, Riazi-Esfahani M, Karkhane R, Aghazadeh S, Rafiee-Tehrani M, et al. Development, characterizations and biocompatibility evaluations of intravitreal lipid implants. Jundishapur J Nat Pharm Prod 2014; 9(2). 10.17795/jjnpp-16414.
- Tamaddon L, Mostafavi SA, Karkhane R, Riazi-Esfahani M, Dorkoosh FA, Rafiee-Tehrani M. Thermoanalytical characterization of clindamycin-loaded intravitreal implants prepared by hot melt extrusion. Adv Biomed Res 2015; 4:147. doi: 10.4103/2277-9175.161563 [Crossref] [ Google Scholar]
- Abdulla D, Ali Y, Menezo V, Taylor SR. The use of sustained release intravitreal steroid implants in non-infectious uveitis affecting the posterior segment of the eye. Ophthalmology and Therapy 2022; 11:479-87. doi: 10.1007/s40123-022-00456-4 [Crossref] [ Google Scholar]
- Maulvi FA, Lakdawala DH, Shaikh AA, Desai AR, Choksi HH, Vaidya RJ. In vitro and in vivo evaluation of novel implantation technology in hydrogel contact lenses for controlled drug delivery. J Control Release 2016; 226:47-56. doi: 10.1016/j.jconrel.2016.02.012 [Crossref] [ Google Scholar]
- Alegre E, Sammamed M, Fernandez-Landazuri S, Zubiri L, Gonzalez A. Circulating biomarkers in malignant melanoma. Adv Clin Chem 2015; 69:47-89. doi: 10.1016/bs.acc.2014.12.002 [Crossref] [ Google Scholar]
- Idrees S, Williams Z, DiLoreto Jr D. Autoimmune retinopathy treated with fluocinolone acetate intravitreal implant (Retisert). Investig Ophthalmol Vis Sci 2020; 61:314. [ Google Scholar]
- Jaffe GJ, Martin D, Callanan D, Pearson PA, Levy B, Comstock T. Fluocinolone Acetonide Uveitis Study GroupFluocinolone acetonide implant (Retisert) for noninfectious posterior uveitis: thirty-four-week results of a multicenter randomized clinical study. Ophthalmology 2006; 113:1020-7. doi: 10.1016/j.ophtha.2006.02.021 [Crossref] [ Google Scholar]
- Kane FE, Burdan J, Cutino A, Green KE. IluvienTM: a new sustained delivery technology for posterior eye disease. Expert Opin Drug Deliv 2008; 5:1039-46. [ Google Scholar]
- Medeiros MD, Postorino M, Navarro R, Garcia-Arumí J, Mateo C, Corcóstegui B. Dexamethasone intravitreal implant for treatment of patients with persistent diabetic macular edema. Ophthalmologica 2014; 231:141-6. doi: 10.1159/000356413 [Crossref] [ Google Scholar]
- Chan A, Leung L-S, Blumenkranz MS. Critical appraisal of the clinical utility of the dexamethasone intravitreal implant (Ozurdex®) for the treatment of macular edema related to branch retinal vein occlusion or central retinal vein occlusion. Clin Ophthalmol (Auckland, NZ) 2011; 5:1043. doi: 10.2147/OPTH.S13775 [Crossref] [ Google Scholar]
- Dugel P, Cantrill H, Eliott D, Mahmoud T, Avery R, Varner S. Clinical safety and preliminary efficacy of an intravitreal triamcinolone implant (I-vationTM TA) in DME. Investig Ophthalmol Vis Sci 2007; 48:1413. doi: 10.1001/archophthalmol.2009.234 [Crossref] [ Google Scholar]
- Kanjickal D, Lopina S, Evancho‐Chapman MM, Schmidt S, Donovan D. Effects of sterilization on poly (ethylene glycol) hydrogels. J Biomed Mater Res A 2008; 87:608-17. doi: 10.1002/jbm.a.31811 [Crossref] [ Google Scholar]
- Karajanagi SS, Yoganathan R, Mammucari R, Park H, Cox J, Zeitels SM. Application of a dense gas technique for sterilizing soft biomaterials. Biotechnol Bioeng 2011; 108:1716-25. doi: 10.1002/bit.23105 [Crossref] [ Google Scholar]
- Ebinuma VdCS, de Oliveira F, Veríssimo NV, Pereira JFB, Pessoa A. Sterilization in pharmaceutical biotechnology. In: Pharmaceutical Biotechnology. CRC Press; 2021. p. 133-45.
- Galante R, Pinto TJ, Colaço R, Serro AP. Sterilization of hydrogels for biomedical applications: A review. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2018; 106:2472-92. doi: 10.1002/jbm.b.34048 [Crossref] [ Google Scholar]
- Zhang K, Feng Q, Fang Z, Gu L, Bian L. Structurally dynamic hydrogels for biomedical applications: pursuing a fine balance between macroscopic stability and microscopic dynamics. Chem Rev 2021; 121:11149-93. doi: 10.1021/acs.chemrev.1c00071 [Crossref] [ Google Scholar]
- Hoare TR, Kohane DS. Hydrogels in drug delivery: Progress and challenges. Polymer 2008; 49:1993-2007. doi: 10.1016/j.polymer.2008.01.027 [Crossref] [ Google Scholar]
- Lin C-C, Anseth KS. Lin C-C, Anseth KSPEG hydrogels for the controlled release of biomolecules in regenerative medicine. Pharm Res 2009; 26:631-43. doi: 10.1007/s11095-008-9801-2 [Crossref] [ Google Scholar]
- Wang W, Singh S, Zeng DL, King K, Nema S. Antibody structure, instability, and formulation. J Pharm Sci 2007; 96:1-26. doi: 10.1002/jps.20727 [Crossref] [ Google Scholar]
- Sridhar MS. Anatomy of cornea and ocular surface. Indian J Ophthalmol 2018; 66:190. doi: 10.4103/ijo.IJO_646_17 [Crossref] [ Google Scholar]
- Sánchez-López E, Espina M, Doktorovova S, Souto E, García M. Lipid nanoparticles (SLN, NLC): Overcoming the anatomical and physiological barriers of the eye–Part I–Barriers and determining factors in ocular delivery. Eur J Pharm Biopharm 2017; 110:70-5. doi: 10.1016/j.ejpb.2016.10.013 [Crossref] [ Google Scholar]
- Sánchez-López E, Espina M, Doktorovova S, Souto E, García M. Lipid nanoparticles (SLN, NLC): Overcoming the anatomical and physiological barriers of the eye–Part II-Ocular drug-loaded lipid nanoparticles. Eur J Pharm Biopharm 2017; 110:58-69. doi: 10.1016/j.ejpb.2016.10.013 [Crossref] [ Google Scholar]
- Ghasemiyeh P, Mohammadi-Samani S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: applications, advantages and disadvantages. Res Pharm Sci 2018; 13:288. doi: 10.4103/1735-5362.235156 [Crossref] [ Google Scholar]
- Del Amo EM, Rimpelä A-K, Heikkinen E, Kari OK, Ramsay E, Lajunen T. Pharmacokinetic aspects of retinal drug delivery. Progress in Retinal and Eye Research 2017; 57:134-85. doi: 10.1016/j.preteyeres.2016.12.001 [Crossref] [ Google Scholar]
- Ragelle H, Danhier F, Préat V, Langer R, Anderson DG. Nanoparticle-based drug delivery systems: a commercial and regulatory outlook as the field matures. Expert Opin Drug Deliv 2017; 14:851-64. doi: 10.1080/17425247.2016.1244187 [Crossref] [ Google Scholar]
- Lim JW, Lee HK, Shin MC. Comparison of intravitreal bevacizumab alone or combined with triamcinolone versus triamcinolone in diabetic macular edema: a randomized clinical trial. Ophthalmologica 2012; 227:100-6. doi: 10.1159/000331935 [Crossref] [ Google Scholar]